An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
- My Bibliography
- Collections
- Citation manager

Save citation to file
Email citation, add to collections.
- Create a new collection
- Add to an existing collection
Add to My Bibliography
Your saved search, create a file for external citation management software, your rss feed.
- Search in PubMed
- Search in NLM Catalog
- Add to Search
Transforming the fermented fish landscape: Microbiota enable novel, safe, flavorful, and healthy products for modern consumers
Affiliations.
- 1 Key Laboratory of Aquatic Product Processing, Ministry of Agriculture and Rural Affairs of The People's Republic of China, National R&D Center for Aquatic Product Processing, South China Fisheries Research Institute, Chinese Academy of Fishery Sciences, Guangzhou, China.
- 2 Co-Innovation Center of Jiangsu Marine Bio-industry Technology, Jiangsu Ocean University, Lianyungang, China.
- 3 Collaborative Innovation Center of Seafood Deep Processing, Dalian Polytechnic University, Dalian, China.
- 4 School of Chemical Sciences, The University of Auckland, Auckland, New Zealand.
- PMID: 37458317
- DOI: 10.1111/1541-4337.13208
Regular consumption of fish promotes sustainable health while reducing negative environmental impacts. Fermentation has long been used for preserving perishable foods, including fish. Fermented fish products are popular consumer foods of historical and cultural significance owing to their abundant essential nutrients and distinct flavor. This review discusses the recent scientific progress on fermented fish, especially the involved flavor formation processes, microbial metabolic activities, and interconnected biochemical pathways (e.g., enzymatic/non-enzymatic reactions associated with lipids, proteins, and their interactions). The multiple roles of fermentation in preservation of fish, development of desirable flavors, and production of health-promoting nutrients and bioactive substances are also discussed. Finally, prospects for further studies on fermented fish are proposed, including the need of monitoring microorganisms, along with the precise control of a fermentation process to transform the traditional fermented fish to novel, flavorful, healthy, and affordable products for modern consumers. Microbial-enabled innovative fermented fish products that consider both flavor and health benefits are expected to become a significant segment in global food markets. The integration of multi-omics technologies, biotechnology-based approaches (including synthetic biology and metabolic engineering) and sensory and consumer sciences, is crucial for technological innovations related to fermented fish. The findings of this review will provide guidance on future development of new or improved fermented fish products through regulating microbial metabolic processes and enzymatic activities.
Keywords: fermentation; fish product; flavor; health benefits; metabolic pathway; microbial community.
© 2023 Institute of Food Technologists®.
PubMed Disclaimer
Similar articles
- Synthetic microbial communities: Novel strategies to enhance the quality of traditional fermented foods. Jin R, Song J, Liu C, Lin R, Liang D, Aweya JJ, Weng W, Zhu L, Shang J, Yang S. Jin R, et al. Compr Rev Food Sci Food Saf. 2024 Jul;23(4):e13388. doi: 10.1111/1541-4337.13388. Compr Rev Food Sci Food Saf. 2024. PMID: 38865218 Review.
- A systematic review on the flavor of soy-based fermented foods: Core fermentation microbiome, multisensory flavor substances, key enzymes, and metabolic pathways. An F, Wu J, Feng Y, Pan G, Ma Y, Jiang J, Yang X, Xue R, Wu R, Zhao M. An F, et al. Compr Rev Food Sci Food Saf. 2023 Jul;22(4):2773-2801. doi: 10.1111/1541-4337.13162. Epub 2023 Apr 20. Compr Rev Food Sci Food Saf. 2023. PMID: 37082778
- Advances in fermented foods revealed by multi-omics: A new direction toward precisely clarifying the roles of microorganisms. Shi H, An F, Lin H, Li M, Wu J, Wu R. Shi H, et al. Front Microbiol. 2022 Dec 14;13:1044820. doi: 10.3389/fmicb.2022.1044820. eCollection 2022. Front Microbiol. 2022. PMID: 36590428 Free PMC article. Review.
- Regulation of microbial metabolism on the formation of characteristic flavor and quality formation in the traditional fish sauce during fermentation: a review. Han J, Kong T, Wang Q, Jiang J, Zhou Q, Li P, Zhu B, Gu Q. Han J, et al. Crit Rev Food Sci Nutr. 2023;63(25):7564-7583. doi: 10.1080/10408398.2022.2047884. Epub 2022 Mar 7. Crit Rev Food Sci Nutr. 2023. PMID: 35253552 Review.
- The relationship between flavor formation, lipid metabolism, and microorganisms in fermented fish products. Feng L, Tang N, Liu R, Gong M, Wang Z, Guo Y, Wang Y, Zhang Y, Chang M. Feng L, et al. Food Funct. 2021 Jul 7;12(13):5685-5702. doi: 10.1039/d1fo00692d. Epub 2021 May 25. Food Funct. 2021. PMID: 34037049 Review.
- Aarti, C., & Khusro, A. (2019). Functional and technological properties of exopolysaccharide producing autochthonous Lactobacillus plantarum strain AAS3 from dry fish based fermented food. LWT, 114(16), 108387. https://doi.org/10.1016/j.lwt.2019.108387
- Aarti, C., Khusro, A., Varghese, R., Arasu, M. V., Agastian, P., Al-Dhabi, N. A., & Choi, K. C. (2017). In vitro studies on probiotic and antioxidant properties of Lactobacillus brevis strain LAP2 isolated from Hentak, a fermented fish product of North-East India. LWT, 86(14), 438-446. https://doi.org/10.1016/j.lwt.2017.07.055
- Alak, G., Hisar, S. A., Hisar, O., & Genççelep, H. (2011). Biogenic amines formation in Atlantic bonito (Sarda sarda) fillets packaged with modified atmosphere and vacuum, wrapped in chitosan and cling film at 4°C. European Food Research and Technology, 232(1), 23-28. https://doi.org/10.1007/s00217-010-1354-z
- Alvarez, M. A., & Moreno-Arribas, M. V. (2014). The problem of biogenic amines in fermented foods and the use of potential biogenic amine-degrading microorganisms as a solution. Trends in Food Science & Technology, 39(2), 146-155. https://doi.org/10.1016/j.tifs.2014.07.007
- An, C., Takahashi, H., Kimura, B., & Kuda, T. (2010). Comparison of PCR-DGGE and PCR-SSCP analysis for bacterial flora of Japanese traditional fermented fish products, aji-narezushi and iwashi-nukazuke. Journal of the Science of Food and Agriculture, 90(11), 1796-1801. https://doi.org/10.1002/jsfa.4015
Publication types
- Search in MeSH
Related information
Linkout - more resources, full text sources.

- Citation Manager
NCBI Literature Resources
MeSH PMC Bookshelf Disclaimer
The PubMed wordmark and PubMed logo are registered trademarks of the U.S. Department of Health and Human Services (HHS). Unauthorized use of these marks is strictly prohibited.
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
Preview improvements coming to the PMC website in October 2024. Learn More or Try it out now .
- Advanced Search
- Journal List

Fish Waste: From Problem to Valuable Resource
Daniela coppola.
1 Department of Marine Biotechnology, Stazione Zoologica Anton Dohrn, Villa Comunale, 80121 Naples, Italy; [email protected] (D.C.); [email protected] (C.L.); [email protected] (F.P.E.); [email protected] (G.R.); [email protected] (C.R.)
Chiara Lauritano
Fortunato palma esposito, gennaro riccio, carmen rizzo, donatella de pascale.
2 Institute of Biochemistry and Cell Biology (IBBC), National Research Council, Via Pietro Castellino 111, 80131 Naples, Italy
Associated Data
Not applicable.
Following the growth of the global population and the subsequent rapid increase in urbanization and industrialization, the fisheries and aquaculture production has seen a massive increase driven mainly by the development of fishing technologies. Accordingly, a remarkable increase in the amount of fish waste has been produced around the world; it has been estimated that about two-thirds of the total amount of fish is discarded as waste, creating huge economic and environmental concerns. For this reason, the disposal and recycling of these wastes has become a key issue to be resolved. With the growing attention of the circular economy, the exploitation of underused or discarded marine material can represent a sustainable strategy for the realization of a circular bioeconomy, with the production of materials with high added value. In this study, we underline the enormous role that fish waste can have in the socio-economic sector. This review presents the different compounds with high commercial value obtained by fish byproducts, including collagen, enzymes, and bioactive peptides, and lists their possible applications in different fields.
1. Introduction
Global use of natural resources has been increasing substantially in recent years, reaching 92.1 billion tons in 2017 and resulting in an increase of 254% from 27 billion in 1970, with the rapid build-up in the annual extraction since 2000 [ 1 ]. Because of this, the research is improving its efforts in building a circular bioeconomy, which aims to enhance the value of material flows and to achieve sustainable consumption and production. It represents an emerging concept that elicits great attention for the purpose of efficient and sustainable use of resources, energy and infrastructure to ensure the quality of life of humans. In this framework, biomass wastes play a major role in the implementation of circular bioeconomy, based on the reuse and recycling of materials to reduce waste production.
In the last years, fish products consumption has seen a huge increase following its recognition as a key component of a balanced diet and healthy lifestyle. Based on the Food and Agricultural Organization (FAO), United States, the total fisheries and aquaculture production has shown a considerable increase of more than eightfold between 1954 and 2014, driven by advances in fishing technologies and rapid developments in aquaculture; in 2014, the global fishery production was 93.4 million tons [ 2 ]. In the period 1961–2016, the growth in the global fish supply had a notable increase, with an average annual growth rate of 3.2%, higher than the growth rate of world’s population (1.6%), even surpassing that of meat from land animals (2.8%). World fish consumption per capita increased from 9.0 kg in 1961 to 20.2 kg in 2015, and preliminary estimates indicate further growth for 2016 and 2017 to approximately 20.3 kg and 20.5 kg, respectively [ 3 ]. Consequently, also the amount of fish waste has undergone a dramatic increase across the world. Currently, the post-catch fish losses represent a huge economic and environmental concern occurring in most fish distribution chains, with large amounts of landed fish lost or discarded between landing and consumption [ 2 ]. It is important to underline that today the expansion of consumption is driven not only by the increase in production, but also by different factors, including the reduction of wastage. In fact, although the continuous increase in fish consumption in 2016 a small decrease in global capture fisheries production (90.9 million tons) was observed, compared to the previous two years [ 3 ]. In order to promote environmentally, economically, and socially sustainable EU fishing practices, the main recent objectives pursued by the EU Common Fisheries Policy (CFP) are the drastic reduction/prohibition of discards and the use of the captured biomass as best as possible [ 4 ].
To date, fish waste is partly destined for the production of fishmeal, fertilizers, and fish oil with low profitability or utilized as raw material for direct feeding in aquaculture [ 5 , 6 , 7 , 8 ], and partly thrown away [ 9 ].
For this reason, a better fish-waste management is needed to overcome environmental issues and for the fully use of biomass for purposes of high-commercial value, at the same time. In this context, the growing recent attention to alternative uses of fish byproducts plays an important role in the economic growth and sustainable development. Several studies have been reported to analyze their possible uses, as they represent a rich source of value-added compounds, including enzymes, bioactive peptides, and bio-polymers, with many possible uses in several fields [ 10 , 11 ].
In this review, the authors discuss about how circular bioeconomy can be achieved through sustainable fish waste management, examining the global situations of fish waste, the main characteristics of compounds obtained from fish byproducts, and possible uses for production of value-added products.
2. Fish Waste in the Circular Bioeconomy Era
The enormous population growth that has taken place in the last two decades, and the consequent extensive use of nonrenewable resources, has negatively affected the quality of the environment and pushed towards sustainable strategies. In this context, the uses of alternative resources that can replace fossil ones and the development of renewable processes based on sustainability are essential for future generations. The transition from a linear to a circular economy is currently an indispensable aspect for managing resources in an eco-efficient way, since the concept of sustainability is totally based on the circularity of all the necessary materials.
The circular bioeconomy is an integral part of the circular economy and it is fundamental in achieving both sustainability in terms of resources and environmental sustainability. The bioeconomy uses materials of biological origin and imitates or uses processes developed by nature to achieve an efficient use in terms of resources [ 12 ]. According to the European Commission, bioeconomy is defined as “the production of renewable biological resources and the conversion of these resources and waste streams into value-added products, including food, feed, bio-based products and bioenergy” [ 13 ]. In this way, the anthropogenic consumption of raw materials of fossil origin is reduced, the influx of renewable resources is supported, and the environmental impact is reduced to a minimum.
The greatest strengths of the circular bioeconomy are: awareness by people and industry, involvement of stakeholders and policy makers, support of politics, sustainable production and consumption, resources valorization, and zero waste. In this sense, the bio-waste valorization approach plays a fundamental role in bringing circularity to the bioeconomy. In this area huge efforts are underway by the scientific community, together with the support of the government, and are largely directed at the recovery of resources from biological waste.
In this context, a growing awareness and special attention to the development of greener and more sustainable processes have led to a greater interest in the use of unwanted marine resources, such as the huge amount of waste obtained by fishing and aquaculture, which is a very promising source of products with high market value [ 10 , 11 , 14 ]. Each year, a huge amount of biomass is (i) discarded, generally incinerated, increasing the energy consumption, financial cost, and environmental impact of their management process [ 15 ], or (ii) utilized for low-value products; to date, fish waste is used mainly in the fish meal industry, since it contains almost the same amount of proteins as fish meat [ 5 , 6 , 7 ]. Moreover, the nutritional composition of fish waste allows to supply plant nutrients or to enrich a compost. In fact, fish waste is/can be processed to produce several fertilizers [ 8 ], and currently commercial fish-based fertilizers are used for agricultural and horticultural crops [ 16 ]. In addition, fish waste has high concentration of biodegradable organics which could be recycled as attractive co-substrate for waste activated sludge, to improve the methane production during anaerobic co-digestion [ 17 , 18 ].
More than 70% of the total fish caught is subjected to further processing before being placed on the market [ 19 ], resulting in the production of large amounts (approximately 20–80%) of fish waste, depending on the level of processing (e.g., gutting, scaling, filleting) and species, because each species has a specific composition, size, shape and intrinsic chemistry [ 20 , 21 ]. These operations generate discards which mainly include muscle-trimmings (15–20%), skin and fins (1–3%), bones (9–15%), heads (9–12%), viscera (12–18%), and scales (5%) [ 22 ]. Fish processing is an important need for large fish companies both to reduce the costs related to transport of inedible parts of the fish and to increase stability and quality of products, removing parts, such as the viscera, that might contain bacteria and enzymes, which represent a risk for processing and storage of the fish [ 23 ]. Preserving the nutritional quality of products represents one of the main challenges for the industry. The degradation of proteins by enzymes is a key aspect that should be minimized, as a high degree of hydrolysis could produce bitter-tasting peptides [ 23 , 24 ], and together with the lipid peroxidation, lead variability in raw materials [ 24 , 25 ]. Acceptable levels of lipid and protein hydrolysis are enormously dependent on the product, based on its end use [ 26 , 27 ]. Hydrolysis determines structural and conformational changes, with possible negative effects on the physicochemical and functional properties of proteins [ 23 ]. For this reason, the control of the autolysis and auto-oxidation of these products is fundamental for their use [ 20 ]. This represents a real challenge for fishing vessels, which require more advanced equipment and technologies for capture and better handling, necessary to maintain the quality of the byproducts [ 28 , 29 ]. Moreover, to ensure food safety and consumer protection, increasingly stringent hygiene measures have been implemented at the National and International level.
In addition, a substantial amount of by-catch is rejected each year, including both low-value species and tons of commercially valuable but undersized fish. However, the amount of fish byproducts is expected to increase in the next years, due to the implementation of the landing obligation, as part of the new reform of the EU-CFP. It requires the obligation of landing all commercially exploited species under the total allowable catch regulations, including undersized fish which cannot be used for direct human consumption, and endangered species, avoiding the great waste of precious fish biomass through the practice of fish discards [ 9 , 30 ]. In fact, it has been estimated that nearly 25% of all the caught fish never reaches the market; every year, approximately 27 million tons of unwanted fish are discarded into the sea, and much of it does not survive.
The final goals of the new landing obligation are also closely related to two other EU strategies, Blue Growth and the 2020 EU Strategy, involved in developing sustainable socio-economic and environmental growth in the marine and maritime EU region [ 31 ]. In particular, Blue Growth is a long-term strategy based on the exploitation of seas and oceans, which have great potential for innovation and growth, offering new ways to help the EU emerge from its current crisis and drive the economy. Considering all the activities that depend on the sea, the EU blue economy implies 5.4 million jobs and a gross added value of almost €500 billion per year. The Organization for Economic Co-operation and Development (OECD) expects that many ocean-based industries have the potential to outperform the global economy by 2030, in terms of value added and jobs, and the output of the global ocean economy could be more than double [ 32 ].
Part of the Blue Growth strategy is focused on the blue biotechnology that involves the transformation of raw marine materials in order to obtain products with high economic value useful for different biotechnological applications, which could be utilized for the development of innovative markets, contributing to the goals of the EU strategies.
In this context, the valorization strategies for fish discards and fish byproducts could contribute to the economic growth. Likewise, new uses for fish waste could alleviate the costs associated to the landing obligation, and reduce the enormous environmental problems associated to the large amount of waste.
3. Fish Byproducts as Source of High Added-Value Compounds
Due to this massive increase in fish consumption, in 2015 fish accounted for approximately 17% of the global population’s intake of animal protein and 6.7% of all proteins consumed in the world [ 3 ]. A fish-based diet has a huge positive nutritional impact, also playing an important role in correcting unbalanced diets and countering obesity. In fact, besides being a rich source of high-quality protein carrying all essential amino acids, fish supply (i) essential fats, such as long-chain omega-3 fatty acids, (ii) vitamins, such as D, A and B, and (iii) minerals, including calcium, iodine, zinc, iron and selenium. Thanks to these valuable nutritional properties, fish bring health benefits in protecting against cardiovascular disease, and help fetal and infant development of the brain and nervous system [ 2 ]. In fact, marine species, including fish products, are popular for medical use and are considered as an exploitable source of efficacious animal-derived medicinal products (ADMPs) [ 33 ].
Fish byproducts are a nutritionally important source of proteins, fatty acids, and minerals, as their composition is similar to that of fish fillet and other food products used for consumption. Studies conducted on meagre and gilthead sea bream fish species have shown that skin is the most significant protein source, trimmings and bones are rich in calcium, and the head, intestines, and bones are a good source of lipids [ 34 ]. Specifically, the mean value of all fish byproducts, calculated on a dry weight basis, is 49.22–57.92% for protein content, 21.79–30.16% for ash content, and 7.16–19.10% for fat content [ 35 , 36 , 37 ]. The latter value is slightly higher than those found by [ 34 ].
In addition to the share of world fish production utilized for direct human consumption, the use of byproducts is increasingly gaining attention, as they offer a significant and sustainable source of high-value bio-compounds, due to their high content of collagen, peptides, chitin, polyunsaturated fatty acids (PUFAs), enzymes, and minerals, suitable for biotechnological or pharmaceutical applications with high market value [ 10 , 11 , 14 , 38 , 39 , 40 , 41 , 42 , 43 , 44 ].
Many studies have already extensively reported the methods used for the extraction of these compounds [ 14 , 38 , 45 , 46 , 47 ]. For this reason, this paragraph provides an overview of the extraordinary potential of fish byproducts, listing the possible applications found so far for the most interesting compounds extracted from fish byproducts (e.g., collagen, peptides, chitin, oil, enzymes).
3.1. Collagen
The Marine Collagen Market has been estimated to reach USD 983.84 million by 2025, growing at a Compound Annual Growth Rate (CAGR) of 7.4%. The growth of the marine collagen market is due to the use of collagen in the cosmetic, food and beverage industry [ 48 ], and fish waste represents a huge and cheap source of collagen for the industry [ 49 ].
Collagen is a complex and abundant structural protein, that is present only in metazoa and groups approximately 20–30% of animal proteins [ 50 , 51 ]; 28 different types of collagen have been found in higher metazoan [ 52 ]. Collagen types are classified depending on their primary structures and forms of supramolecular organization. Fibrillar collagens (collagen types I, II, III, V, XI, XXIV, XXVII) participate in the formation of cross-striated fibrils. In mature fibrillar collagen molecules, the collagenous domain of each α chain contains the repeated triplets sequence Gly-X-Y. Fibrillar collagen (type I) is the most abundant protein in vertebrates and it plays a key role in the mechanical properties of bones, tendons, and skin [ 53 ]. Other subfamilies correspond to non-fibrillar collagens, which contain different interruptions in their collagenous domains. Non-fibrillar collagens may be classified in several subfamilies: (1) network-forming collagen which includes basement membrane (type IV), beaded filaments forming collagen (type VI), anchoring fibril (type VII), short-chain collagens (types VIII, X), which are hexagonal network-forming collagens; (2) fibril-associated collagens with interrupted triple helices (types IX, XII, XIV, XX); (3) fibril-associated with interrupted triplex helix-like (types XVI, XIX, XXI, XXII); (4) plasma membrane collagens (types XIII, XVII, XXIII, XXV); (5) multiplexin collagens (types XV, XVIII), and other molecules with collagenous domains (types XXVI, XXVIII) [ 14 , 53 ].
Partial hydrolysis and heat denaturation of collagen lead to gelatin production. Generally two types of gelatin, type A and B, are obtained by acid and alkaline hydrolysis, respectively [ 54 ]. During production of gelatin by hydrolysis the inter-molecular and intra-molecular bonds of collagen fibers are broken [ 55 ], and the structural characteristics of gelatin depend on the distribution of molecular weight (MW), structure and composition of each subunit [ 54 ]. Composition of gelatin is closely related to the origin collagen, however it contains many glycine, proline and hydroxyproline residues, approximately 21%, 12%, and 12%, respectively [ 56 ].
Moreover, hydrolysed collagen can be obtained by enzymatic action (alcalase, papain, pepsin, and others) in alkaline or acid media at specific incubation temperature, generally above 40 °C. The obtained peptides have low MW (0.3–8 KDa) and are easy to be absorbed by the intestine and available for tissues [ 57 ]. Their solubility and biological activity are related to the parameters used for the extraction, such as types of used enzymes and type of hydrolyses [ 58 ]. Recently, it has been shown that green and sustainable deep eutectic solvents can also be used to extract collagen peptides [ 59 ].
The main sources of commercial collagen are bovine and pig skin, cattle bones and other mammalian animal waste [ 60 ]. Collagen is also present in marine animals like sponge, jellyfish, molluscs (mussel, squid cuttlefish and octopus), echinoderms, and fish [ 61 ]. Therefore, fish waste could be used to obtain cheapest collagen. In fact, it mainly consists of bones, skin, scales and fins [ 5 ] having high collagen content, thus it may be considered to produce collagen [ 14 ]. Fish collagen has important properties, as such as a more efficient adsorption into the body (up to 1.5 fold) and higher bioavailability with respect to the porcine and bovine collagen [ 62 ]. However, fish collagen possesses disadvantages in melting point, mechanical strength, biomechanical stiffness, and high rate of degradation [ 63 ]. However, the main problem related to the use of fish collagen is the development of a sustainable extraction process for its commercial exploitation [ 64 ].
Different procedures have been proposed to extract collagen from fish waste. Acid extraction procedure is the most common method to extract collagen from fish waste, and the obtained product is referred to as acid-soluble collagen (ASC). Collagen extraction is obtained using acids, such as acetic acid or HCl, which solubilized the collagen chains, improving the extraction yield. Pepsin extraction procedure consists in the treatment of the collagen fibres with the pepsin to cleave the specific regions in order to promote solubilisation and increase collagen extraction yield; in this case, the extracted collagen is named pepsin-soluble collagen (PSC) [ 49 ].
Collagen and gelatin properties are of huge importance for many application fields, in fact recent studies are pointing towards the use of food sources such as fish-derived collagen and gelatin as excellent functional molecules for the cosmetic, pharmaceutical, biomaterial, food, and nutraceutical industries [ 14 , 58 , 65 ]. Thus, in the next section and in Table 1 we discuss the recent advances in the use of collagen and gelatin from fish byproducts.
Collagen/gelatin from fish waste and their possible applications. BW, body weight; PVA, polyvinyl alcohol; FC/PCL, fish collagen/polycaprolactone.
| Compound | Byproduct | Source | Applications | Activity/Concentration Used | Reference |
|---|---|---|---|---|---|
| Collagen | Skin | Tilapia | Cartilage tissue engineering | 2% weight/volume | [ ] |
| Collagen/polycaprolactone | Scale | sp. | Cartilage tissue engineering | 12% weight/volume | [ ] |
| Collagen/polycaprolactone | Skin | Bone regeneration | Not specified | [ ] | |
| Collagen | Scale | sp. | Tissue-engineeredoral mucosa | 1% weight/volume | [ , ] |
| Collagen/polyvinyl alcohol | Not specified | sp. | Human periodontal ligament fibroblasts (HPDLFs), gingival fibroblasts (HGFs) | Not specified | [ ] |
| Collagen | Scales | Bone regeneration | 0.3%, weight/volume | [ ] | |
| Collagen | Skin | Bone tissue engineering | 0.2–2.5 mg/mL | [ ] | |
| Collagen | Cartilage | | Bioscaffold | Not specified | [ ] |
| Collagen/chitosan | Skin | Skin regeneration | Not specified | [ ] | |
| Collagen | Skin | Wound healing | 15 mg/mL | [ ] | |
| Collagen | Scale | Wound healing | 5–15% weight/volume | [ ] | |
| Collagen nanofibers | Skin | sp. | Skin regeneration, antibacterial | 30 mg/kg BW | [ ] |
| Collagen | Skin | sp. | Wound healing | Not specified | [ ] |
| Collagen | Scale | Wound healing | Not specified | [ ] | |
| Collagen | Skin | Antioxidant, wound healing | 1–8 mg/mL4 g/day | [ ] | |
| Collagen | Skin | sp. | Antioxidant | 2–7 mg/mL | [ ] |
| Collagen/chitosan | Skin | Not specified | Antioxidant | 12–100 μg/mL | [ ] |
| Collagen/gelatin | Skin | Antioxidant, antibacterial | DPPH and ABTS 1 mg/mL Antibacterial 200 μg | [ ] | |
| Collagen | Swim bladders | Antioxidant | 12.5–50μg/mL | [ ] | |
| Collagen/gelatin | Cartilage | Antioxidant | 1–5 mg/mL | [ ] | |
| Collagen | Skin | Antioxidant | 2.5–10 mg/mL | [ ] | |
| Collagen/gelatin | Scale | Antioxidant | 0.1–5 mg/mL | [ ] | |
| Collagen | Skin | Food packaging | 2.5% weight/weight | [ ] | |
| Collagen | Skin | Food packaging | 0.1% weight/volume | [ ] |
Fish Collagen Applications
Collagen is the most promising natural biomaterial used to build scaffolds in tissue engineering. In fact, scaffolds play a key role in promoting cell regeneration, as they are involved in cell adhesion and differentiation processes [ 66 ]. Synthetic polymeric scaffolds have been widely used in cartilage regeneration [ 67 ]. However, several limitations, such as low biocompatibility and high rate of immune reactions, reduce their further application [ 68 ]. On the contrary, natural scaffolds, including collagen and gelatin, from a wide range of marine sources may possess several advantages, such as biosafety, higher biocompatibility, and weak antigenicity. Moreover, natural scaffolds contain amino acid residues, that promote cell adhesion and differentiation [ 69 ]. Tilapia skin collagen has been tested as scaffold in cartilage regeneration [ 70 ]. Collagen was found to promote cartilage production in vitro (chondrocytes from rabbit auricula) and in vivo (rabbit). In addition, in situ cartilage repair in rabbit articular defect model collagen was tested. Collagen, also in this case, was found to promote cartilage regeneration. Cartilage repair in situ is a direct evidence used to predict the potential of future clinical applications. Tilapia skin collagen did not exhibit cytotoxic side effects in vitro and inflammatory response in mice model. Similar results were obtained using electrospun nanofibrous membranes of fish collagen/polycaprolactone (FC/PCL) [ 66 ]. Collagen from tilapia scale was used to obtain electrospun membranes. The obtained membranes were able to induce in vitro formation of preliminary cartilage-like tissue displaying typical lacunae structures. FC/PCL membranes have been shown to promote in vivo cartilage regeneration in rabbit model. Oh et al. [ 71 ] also proposed a FC/PCL composite scaffolds for bone regeneration processes. Collagen obtained from Paralichthys olivaceus skin was used to produce FC/PCL scaffolds, able to induce cell differentiation, calcium deposition and mineralization in vitro, in model of mouse mesenchymal stem cells. In addition, in vivo experiments were performed to implant the scaffolds in a rabbit tibia. FC/PLC scaffolds have been found to improve new bone formation and no inflammatory or immune responses were observed after scaffolds implantation [ 71 ]. Suzuki et al. engineered oral mucosa by using collagen extracted from tilapia scale. Primary oral mucosa keratinocytes were grown on collagen scaffold. The authors also reported a fully differentiated and stratified epithelial layer developed on collagen scaffolds [ 71 ]. Moreover, blended hydrogels composed of polyvinyl alcohol (PVA) and fish collagen (FC) were used as scaffold to human periodontal ligament fibroblasts (HPDLFs) and gingival fibroblasts (HGFs) growth. A mixture of PVA/FC 50:50 increased growth of both HPDLFs and HGFs [ 72 ]. Collagen from Nile tilapia scales also promoted matrix mineralization in in vitro models [ 73 ]. These data suggest that the tilapia collagen scaffolds may be applied in the production of tissue-engineered oral mucosa equivalents for clinical use. Mineralized collagen from the skin of Atlantic salmon ( Salmo salar ) has been used to produce scaffold for bone regeneration. It allowed human mesenchymal stem cells to grow and to be responsive to the osteogenic stimuli [ 74 ]. Moreover, collagen from raw cartilage from shark ( Prionace glauca ) and ray ( Zeachara chilensis and Bathyraja brachyurops ) has been shown to possess promising properties and biotechnological potential in order to regenerate damaged cartilaginous tissues [ 75 ]. Cao et al. [ 76 ] produced a scaffold-controlled release system for tissue skin engineering based on collagen/chitosan.
Another interesting aspect is the use of the collagen as biomaterials in wet wound dressing [ 77 ]. Collagen from Oreochromis niloticus skin was used to prepare biomedical hydrogel. It has been proven that collagen hydrogel was able to heal of deep second-degree burn of rat skin [ 78 ]. Collagen accelerates wound healing by promoting the expression of vascular endothelial growth factor (VEGF), transforming growth factor-beta (TGF-ß1), basic fibroblast growth factor (bFGF), and alpha-smooth muscle actin (α-SMA) [ 79 ]. Scale fish collagen was also used to deliver polymyxin B, which displays antibacterial activity against Gram-negative bacteria, such as the multidrug-resistant Pseudomonas aeruginosa [ 80 ] and bacitracin, that displays antibacterial activity on Gram-positive bacteria, such as multidrug-resistant staphylococci [ 81 ], in order to prevent bacterial infection during wound healing processes [ 82 ]. Moreover, collagen-based scaffolds impregnated with sago starch capped silver nanoparticles (AgNPs) were fabricated by using collagen derived from fish scales of Lates calcarifer . In vitro studies indicated high tensile strength values for their use as wound dressing materials and antibacterial activities against both Gram-positive ( Staphylococcus aureus ) and negative ( Escherichia coli ) bacterial strains [ 83 ]. Moreover, collagen/bioactive glass nanofibers prepared with collagen from tilapia skin, promoted wound healing and skin regeneration, and at the same time reduced bacterial infection in rat model, showing antibacterial activity against S. aureus [ 84 ]. In addition, Ibrahim et al. [ 85 ] tested Nile tilapia ( O. niloticus ) skin in wound healing in a donkey model. Fish skin accelerated the wound healing process and efficiently inhibited the local microbial activity. Moreover, sponge scaffolds developed by using porcine skin-derived collagen and Ctenopharyngodon idellus scale-derived collagen were compared. Both scaffolds efficiently promoted skin regeneration in rabbit model, with good wound-healing outcome compared to gauze and Vaseline gauze groups. Results showed that fish-collagen scaffold could be an alternative candidate to bovine-collagen scaffolds in burn wound care applications [ 86 ].
Several studies pointed out the antioxidant activity of collagen from fish, interesting for several applications in different fields, including food preservation, healthcare and cosmetics. The antioxidant capacity was mainly evaluated by using the assays 2,2-diphenyl-1-picrylhydrazyl (DPPH), 2,2′-azinobis-(3-ethylbenzothiazoline-6-sulfonate) (ABTS), oxygen radical absorbance capacity (ORAC), Ferric-reducing antioxidant power (FRAP).
PSC obtained from the skin of Lophius litulon was also tested. It has been shown to possess both antioxidant properties in vitro based on its ability to scavenge different free radicals, such as DPPH, ABTS·, HO·, and O 2 - and increase the content of superoxide dismutase (SOD) and catalase (CAT), and wound healing activity in mice model [ 87 ]. Collagen from red tilapia Oreochromis sp. skin was also tested for its antioxidant properties [ 88 ]. It has been found that ASC, salt soluble collagen, and pepsin-hydrolysed collagen showed both radical scavenger activity and ferric-reducing antioxidant power. Hua and collaborators [ 89 ] prepared a bioactive collagen/chitosan complex using fish skin collagen and chitosan solution as raw materials. The bioactive complex possessed antioxidant activity when tested in vivo in mice model, it reduced malonic dialdehyde content and increased SOD activity in mice serum [ 89 ]. Moreover, collagen/gelatin/chitosan novel porous scaffolds fabricated using blends of collagen and gelatin obtained from the marine big eye snapper Priacanthus hamrur skin showed both antioxidant (maximum activity in both DPPH and ABTS assay at 1mg/mL) and antibacterial (maximum activity at 200 µg by disc diffusion assay) activity against E. coli and S. aureus [ 90 ].
In addition, Nibea japonica swim bladders [ 91 ], yellowfin tuna ( Thunnus albacares ) skin [ 92 ], Scomber japonicus bone and skin [ 93 ], shark cartilage [ 94 ] also showed antioxidant properties.
Collagen from fish waste could also find application in the food industry as a food additive and packaging [ 14 ]. Fish collagen has been used as yogurt additive [ 95 ], collagen affected proteolysis of milk proteins and conferred angiotensin I-converting enzyme (ACE) inhibitory property; ACE has a key role in the regulation of blood pressure in mammals and in development of cardiovascular disease, therefore it represents an important target in the treatment of high blood pressure [ 96 ].
Moreover, PSC extracted from P. glauca skin was used to produce chitosan–collagen composite coating. The composite coating has been found to preserve important properties of Pagrus major fillet quality, such as drip loss, pH, and to reduce microbial growth during storage at 4 °C [ 97 ]. Collagen skin from Mustelus mustelus combined with chitosan has been used to produce a protective film in order to preserve nutraceutical products. In addition, this film had antioxidant activities and could act as anti-UV barriers [ 98 ].
Many other fish byproducts collagen applications have been found in the hydrolyzates/peptides obtained from the digestion of collagen and gelatin, which will be discussed in the next section ( Section 3.2 ).
3.2. Peptides
Fish protein hydrolysates market size was about USD 420 million globally in 2019 and it is supposed to increase of 4.5% CAGR between 2020 and 2026 [ 101 ]. Several studies have shown that marine organisms may be an excellent source of bioactive proteins/peptides [ 102 ]. A good amount of proteins has been isolated from marine processing waste. Hydrolysates and purified peptides have been isolated from several fish species (e.g., pollack, sole, salmon, skate, halibut, tuna, catfish, ray, crocker, turbot and hoki), from whole body fish waste or from specific body part waste, such as frame, scale, bone, head, gonads and viscera (as reviewed by [ 38 , 103 ]). Before they can be effectively used, several extraction methods have been applied in order to get bioactive peptides, such as acid-alkaline hydrolysis (by using acidic or alkaline reagent), enzymatic hydrolysis (by using enzymes to hydrolyze fish waste), and fermentation (by using microorganisms as source of enzymes) [ 38 , 47 , 104 ]. Enzymatic hydrolysis is especially preferred in food and pharmaceutical industries because the process does not leave residual organic solvents or toxic chemicals [ 105 ] and is considered the preferred method to hydrolyze fish skin without losing nutritional value [ 47 ]. Fermentation is considered a more natural procedure for protein hydrolysis, and it has been used for centuries as a traditional preservation method, enhances flavor and taste of food but also increases its nutraceutical value [ 39 ]. After the extraction process, various purification steps could follow. For example, ultrafiltration (UF), nanofiltration (NF), and gel filtration (GF) are used to purify peptides based on their MW [ 106 ], ion exchange chromatography (IEC) is used to purify peptides based on their net charge and reversed-phase HPLC can be used to separate compounds based on their hydrophobicity and hydrophilicity. Peptide sequences are then characterized by mass spectrometry methods such as matrix-assisted laser deionization time-of-flight (MALDI-TOF), electrospray ionization mass (ESI), matrix-assisted laser desorption/ionization mass spectrometry (MALDI-MS) (as reviewed by [ 38 ]). For example, Vázquez and collaborators [ 107 ] have just published a study on the use of the turbot Scophthalmus maximus byproducts. In particular, turbot byproducts (from turbot head, viscera, trimmings and frames) were subjected to alcalase hydrolysis obtaining fish protein with high yield of digestion, remarkable degrees of hydrolysis and high content of soluble protein. In the paragraph below and in Table 2 , we summarize peptides isolated from fish waste and their bioactivities.
Hydrolysates and purified peptides from fish waste and their possible applications. MW, molecular weight; MIC, minimum inhibitory concentration; MEC, minimum effective concentration; BW, body weight.
| Compound | Byproduct | Source | Applications | Activity/ Concentration Used | Reference |
|---|---|---|---|---|---|
| SJGAP | Skin | Skipjack tuna ( ) | Antimicrobial | MECs 3, 26, 4.8, 25, 2.7, 9, 16 μg/mL against | [ ] |
| YFGAP | Skin | Yellowfin tuna ( ) | Antimicrobial | MECs 1.2, 6.5, 17, 8, 3, 3.2 μg/mL against | [ ] |
| GKLNLFLSRLEILKLFVGA | Skin | Yellow catfish ( ) | Antimicrobial | MIC 2, 4, 16, 64 μg/mL against | [ ] |
| SIFIQRFTT, RKSGDPLGR, AKPGDGAGSGPR and GLPGLGPAGPK | Not specified | Antibacterial | 200 μg/mL | [ ] | |
| N-KVEIVAINDPFIDL-C | Not specified | Antibacterial | MIC 0.263, 0.131, 0.131, 0.263, 0.263 mMagainst | [ ] | |
| GWGSFFKKAAHVGKHVGKAALTHYL | Skin | Winter flounder ( ) | Antimicrobial | MIC 1.1–2.2, 4.4–8.8, 17.7–35.0, 2.2–3.3, 8.8–17.7, 17.7–35.0 μM against | [ ] |
| GPL, GPM | Skin | Antihypertensive | IC = 2.6 and 17.1 μM | [ ] | |
| DPALATEPDPMPF | Not specified | Antihypertensive | 1–20 μg/mL | [ ] | |
| FGASTRGA | Frame | Pollack ( ) | Antihypertensive | IC = 14.7 µM | [ ] |
| GPEGPAGAR GETGPAGPAGAAGPAGPR | Skin | Antihypertensive | 5 mg/mL | [ ] | |
| MVGSAPGVL, LGPLGHQ | Skin | Skate | Antihypertensive | IC = 3.09, 4.22 µM | [ ] |
| GASSGMPG, LAYA | Skin | Pacific cod skin | Antihypertensive | IC = 6.9, 14.5 µM | [ ] |
| Collagen peptides | Skin | Antihypertensive | IC = 0.49 mg/mL | [ ] | |
| GIPGAP and APGAP | Skin | Antihypertensive | IC =27.9, 170.2 μM | [ ] | |
| GY, VY, GF, VIY | Scale | Sea Bream | Antihypertensive | IC = 265, 16, 708, 7.5 µM | [ ] |
| PGPLGLTGP, QLGFLGPR | Skin | Skate | Antihypertensive | IC = 95, 148 µM | [ ] |
| GLPLNLP | Skin | Salmon ( ) | Antihypertensive | 18.7 µM | [ ] |
| MIFPGAGGPEL | Frame | Sole ( ) | Antihypertensive | 28.7 μg/ml | [ ] |
| GDLGKTTTVSNWSPPKYKDTP | Frame | Tuna | Antihypertensive | 11.28 μM | [ ] |
| Hydrolysates with MW between 1000 and 10,000 Da | Bone | Yellowtail | Antihypertensive, antioxidant | 1.9 mg/mL (ACE inhib.), ~10 mg/mL (DPPH) | [ ] |
| Hydrolysates with MW ≤ 1000 Da | Bone | Yellowtail | Antihypertensive, antioxidant | 1.5 mg/mL (ACE inhib.), ~35 mg/mL (DPPH) | [ ] |
| Hydrolysates with weight ~ 30 KDa | Frame | Cod | Antioxidant, antihypertensive | ~20% (antiox.), ~40 mg protein/mL × 100 | [ ] |
| Hydrolysates with weight ~ 10 KDa | Frame | Cod | Antioxidant, antihypertensive | ~15% (antiox.), ~35 mg protein/mL × 100 | [ ] |
| Hydrolysates with weight ~ 5 KDa | Frame | Cod | Antioxidant, antihypertensive | ~40% (antiox.), ~20 mg protein/mL × 100 | [ ] |
| Hydrolysates with weight ~ 3 KDa | Frame | Cod | Antioxidant, antihypertensive | ~18% (antiox.), ~8 mg protein/mL × 100 | [ ] |
| PYSFK, GFGPEL, VGGRP. | Skin | Grass carp ( ) | Antioxidant | DPPH radical 2.459, 3.634, 6.063 mM (DPPH), 3.563, 2.606, 4.241 mM (hydroxyl), 0.281, 0.530, 0.960 mM (ABTS) | [ ] |
| AVGAT | Skin | Thornback ray | Antioxidant | 33% of activity at 3 mg/mL (DPPH) | [ ] |
| DPALATEPDMPF | Skin | Nile tilapia ( ) | Antioxidant | 8.82 µM (DPPH), 7.56 µM (Hydroxyl) | [ ] |
| PFGPD, PYGAKG, YGPM | Skin | Spanish mackerel | Antioxidant | 0.80, 3.02, 0.72 mg/mL (DPPH), 0.81, 0.66, 0.88 mg/mL (hydroxyl), 0.91, 0.80, 0.73 (superoxide anion), 0.86, 1.07, 0.82 mg/mL (ABTS) | [ ] |
| GSGGL, GPGGFI, FIGP | Skin | Blue leatherjacket ( ) | Antioxidant | 405 µg/mL (DPPH), 179 µg/mL (Hydroxyl); 194 µg/mL (DPPH), 89 µg/mL (Hydroxyl); 118 µg/mL (DPPH), 73 µg/mL (Hydroxyl) | [ ] |
| GLFGPR GATGPQGPLGPR, VLGPF, QLGLGPV | Skin | Seabass ( ) | Antioxidant | 81.41, 10.4, 2.59, 0.50 mmol Trolox equivalents/µmol peptide (ABTS) | [ ] |
| FDSGPAGVL, DGPLQAGQPGER | Skin | Jumbo squid ( ) | Antioxidant | _ | [ ] |
| PAGT | Skin | Amur sturgeon | Antioxidant | 5380 µg/mL (DPPH), 890 µg/mL (Hydroxyl), 8 µg/mL (ABTS) | [ ] |
| EGL, YGDEY | Skin | Nile tilapia ( ) | Antioxidant | 4.61 µg/mL (Hydroxyl), 6.45 µg/mL (Hydroxyl) | [ ] |
| LSGYGP | Skin | Tilapia ( ) | Antioxidant | 22.47 μg/mL | [ ] |
| Hydrolysates | Skin | Snapper ( ) | Antioxidant | _ | [ ] |
| Hydrolysates | Skin | Snapper ( ) | Antioxidant | _ | [ ] |
| Hydrolysates | Skin | Sole | Antioxidant | _ | [ ] |
| Hydrolysates | WB, Head, Gonads | Herring ( ) | Antioxidant | _ | [ ] |
| TCSP, TGGGNV | Skin | Cod ( ) | Antihypertensive, Antioxidant | 81%, 68% at 500 µg/mL, 75% at 500 µg/mL (scavenging activity) for both | [ ] |
| N-terminal RPDFDLEPPY | Frame | Sole ( ) | Antioxidant | _ | [ ] |
| Collagen/gelatin/peptides | Skin | Antioxidant | 9–700 μg/mL | [ ] | |
| GLFGPR | Skin | Antioxidant | 5, 10 mg/mL | [ ] | |
| HGPLGPL | Skin | Hoki ( ) | Antioxidant | 156.2 µM (DPPH) | [ ] |
| GPRGTIGLVG, GPAGPAG and GFPSG | Scales | Antioxidant | IC (mg/mL): 0.293, 0.240, 0.107, (hydroxyl); 1.271, 0.675, 0.283 (DPPH); 0.463, 0.099, 0.151 (superoxide anion); 0.421, 0.309, 0.210 (ABTS). | [ ] | |
| HGPHGE, DGPKGH and MLGPFGPS | Scales | Antioxidant | EC mg/mL: 1.34, 0.54, 0.67 (DPPH) 1.03, 0.41, 0.74 (hydroxyl) 1.19, 0.71, 1.59 (superoxide anion) | [ ] | |
| GAEGFIF | Bone | Antioxidant | EC mg/mL: 0.57, 0.30 (DPPH); 0.25, 0.32 (hydroxyl) 0.52, 0.48 (superoxide anion) 0.41, 0.21 (ABTS) | [ ] | |
| GPE, GARGPQ and GFTGPPGNG | Cartilage | Antioxidant | EC mg/mL: 2.43, 2.66, 1.99 (DPPH); 0.28, 0.21, 0.15 (hydroxyl) 0.24, 0.18, 0.29 (ABTS); 0.10, 0.14, 0.11 (superoxide anion) | [ ] | |
| YGCC, DSSCSG, NNAEYYK and PAGNVR | Skin | Antioxidant | IC = 7.63 μg/mL | [ ] | |
| NHRYDR | Skin | Horse Mackerel ( ) | Antioxidant | 72.3% (DPPH), 51.2% (Hydroxyl) | [ ] |
| GNRGFACRHA | Skin | Crocker ( ) | Antioxidant | 79.6% (DPPH), 56.8% (Hydroxyl) | [ ] |
| QGYRPLRGPEFL | Skin | Skate ( ) | Neuroprotective | 24.26 μM | [ ] |
| Collagen peptides | Skin | Salmon ( ) | Neuroprotective | 0.33, 1.0, 3.0 g/kg rat body | [ ] |
| Collagen peptides | Skin | Salmon ( ) | Neuroprotective | 0.22%, 0.44% or 1.32% wt/wt diet | [ ] |
| Collagen peptide drink | Not specified | Not specified | Antioxidant | 0.25% weight/volume | [ ] |
| Hydrolyzed collagen | Not specified | Skin elasticity | 10 g daily | [ ] | |
| EIGPSGGRGKPGKDGDAGPK, GFSGLDGAKGD | Skin | Cod | Matrix metalloproteinase inhibitory activity | 0.1 mg/mL | [ ] |
| Collagen tripeptide | Skin | Sutchi catfish ( ) | Matrix metalloproteinase inhibitory, anti-photoaging | 167–333 mg/kg/day | [ ] |
| Collagen hydrolysate | Not specified | Not specified | Osteoclastic differentiation of BMSCs | 0.2 mg/mL | [ ] |
| SWFCP | Skin | Tuna | Adipocite differentiation | 0.5–1 mg/mL | [ ] |
| FIMGLY | Cartilage | Anticancer | IC = 4.81 mg/mL | [ ] | |
| Collagen hydrolysate | Skin | Anticancer/antidiabetic/wound healing | 0.05–1 mg/mL | [ ] | |
| Collagen peptides | Skin | Bone regeneration | 1.125, 2.25 or 4.5 g kg BW | [ ] | |
| Collagen peptide | Skin | Gadiformes species | Chondroprotective | 1 g/day | [ ] |
| VLSGGTTMYASLYAE | Frame | Hoki ) | Calcium binding | - | [ ] |
| VLSGGTTMAMYTLV | Frame | Pollack ( ) | Calcium binding | - | [ ] |
| Phosphopeptide (FBP) | Bone | Hoki ( ) | Calcium binding | - | [ ] |
| GPAGPHGPPGKDGR, AGPHGPPGKDGR, AGPAGPAGAR | Skin | Pacific cod | Iron-chelating | _ | [ ] |
Fish Peptides Applications
Several peptides derived from fish hydrolysates have been sequenced and available sequences are summarized in Table 2 . In addition, hydrolysates and purified peptides have been tested for bioactivities useful for prevention and treatment of several human pathologies. Various authors have reported that amino acid type, position and hydrophobicity have been considered to play a relevant role in peptide bioactivities [ 38 ]. Common bioactivities are antimicrobial, antihypertensive, antioxidant and neuroprotective activities [ 104 ].
Regarding antimicrobial activities, peptides from fish hydrolysates were mainly active against Gram-negative bacteria, such as Aeromonas hydrophila , Klebsiella pneumonia , Salmonella enterica , and Salmonella typhi, and Gram-positive bacteria, as Streptococcus iniae , Micrococcus luteus , S. aureus , and Bacillus cereus. However, antifungal activities have been reported as well, such as against the fungus Candida albicans . Activities were evaluated and measured by reporting the minimum inhibitory concentration (MIC) or the minimum effective concentration (MEC) values. SJGAP isolated from Skipjack tuna ( K. pelamis ) skin had the highest activity against E. coli (MEC 2.7 μg/mL) and B. subtilis (MEC 3 μg/mL) [ 108 ], YFGAP from yellowfin tuna ( T. albacares ) was most active against B. subtilis (MEC 1.2 μg/mL), E. coli (MEC 3 μg/mL), and V. parahaemolytics (MEC 3.2 μg/mL) [ 109 ]. GKLNLFLSRLEILKLFVGA from Yellow catfish ( Pelteobagrus fulvidraco ) skin and GWGSFFKKAAHVGKHVGKAALTHYL from winter flounder Pleuronectes americanus skin also were most active against B. subtilis (MIC 2 μg/mL and 1.1–2.2 μM, respectively) [ 110 , 111 ].
Moreover, fish collagen-derived peptides, SIFIQRFTT, RKSGDPLGR, AKPGDGAGSGPR and GLPGLGPAGPK, isolated from Scomber scombrus showed antibacterial activity. In particular, GLPGLGPAGPK had antibacterial activity against both Gram-positive and Gram-negative bacteria [ 112 ]. In addition, the peptide KVEIVAINDPFIDL from Scomber scombrus exhibited antibacterial activity against Lactobacillus acidophilus , Listeria ivanovii , Listeria monocytogenes, M. luteus and Bacteroides thetaiotaomicron (MIC 0.263, 0.131, 0.131, 0.26 and, 0.263 mM, respectively) [ 113 ].
Protein hydrolysate prepared from tilapia waste showed also resistance against Vibrio anguillarum in silver pompano ( Trachinotus blochii ), as well as improved growth performance, metabolism, and innate immune response. The reduced antioxidant enzyme (SOD) activity indicated the fish hydrolysate scavenging activity, which minimizes the necessity of expression of SOD in different tissues [ 114 ].
Several fish collagen-derived peptides showed anti-hypertensive activity, by mainly acting with ACE inhibitory activity or having antihypertensive effects on spontaneously hypertensive rats; these peptides are listed below: GPL and GPM (IC 50 = 2.6 and 17.1 μM, respectively) [ 115 ] and PGASTRGA (IC 50 = 14.7 μM) [ 116 ] isolated from T. chalcogramma skin, DPALATEPDPMPF purified from O. niloticus (higher activity at 20 μg/mL) [ 117 ], GPEGPAGAR and GETGPAGPAGAAGPAGPR from O. niloticus skin gelatin (inhibition at 5 mg/mL) [ 118 ], MVGSAPGVL and LGPLGHQ from Skate (IC 50 3.09 and 4.22 μM, respectively) [ 119 ], GASSGMPG and LAYA purified from Gadus macrocephalus skin gelatin hydrolysates (IC 50 = 6.9 and 14.5 μM) [ 120 ]. Moreover, tripeptides based on GPL isolated from skin gelatin hydrolysates of Alaska Pollock T. chalcogramma (containing glycine, proline, and leucine), were synthesized and showed high ACE inhibitory activity [ 121 ]. Collagen peptides extracted from T. chalcogramma skin, which were obtained by simulating gastrointestinal digestion possessed antihypertensive properties (IC 50 = 0.49 mg/mL) [ 122 ]. In addition, the synthetic peptides, GIPGAP and APGAP, derived from collagen proteolysis of Raja clavata skin have been also characterized for their ACE-inhibitory properties (IC 50 = 27.9 and 170.2 μM) [ 123 ].
Fahmi et al. [ 124 ] tested hydrolysates by using spontaneously hypertensive rats fed with 300 mg of peptides (kg of body weight) −1 d −1 and also purified and tested ACE inhibitory activities of four peptides (GY, VY, GF, VIY) from sea bream scales (IC 50 265, 16, 708, 7.5 µM, respectively). Peptides were also isolated from Raja kenojei skin protein hydrolysates obtained by treatment with several proteases (i.e., alcalase, a-chymotrypsin, neutrase, pepsin, papain, and trypsin). Peptides obtained by chymotrypsin digestion, PGPLGLTGP and QLGFLGPR, showed the higher ACE-inhibitory activity(IC 50 = 95 and 148 μM, respectively) [ 125 ]. Moreover, GLPLNLP was isolated from salmon O. keta skin trypsin hydrolysate (IC 50 = 18.7 μM). Synthetic peptides based on the sequence of the purified peptide from salmon skin were also tested, and the peptide GLP had the highest ACE inhibitory activity (IC 50 = 9.08 μM) [ 126 ]. Interesting peptides with ACE inhibitory activity were isolated from fish frame protein hydrolysates, including FGASTRGA isolated from Alaska pollack protein hydrolysates, obtained with pepsin (IC 50 = 14.7 μM) [ 117 ], MIFPGAGGPEL obtained from Yellowfin sole ( Limanda aspera ) hydrolysates MW < 5KDa (IC 50 = 28.7 μg/mL) [ 127 ], GDLGKTTTVSNWSPPKYKDTP isolated from tuna (IC 50 = 11.28 μM) [ 128 ].
Ohba et al. [ 129 ] evaluated the ACE inhibitory and antioxidant activities of yellowtail fish bone hydrolysates. In particular, they studied the activity of crude hydrolysates, hydrolysates with MW ≥ 10,000, MW between 1000 and 10,000, or MW ≤ 1000. They found that IC 50 were 2.0, 2.8, 1.9 and 1.5 mg/mL for crude hydrolysates, hydrolysates with MW ≥ 10,000, MW between 1000 and 10,000 and MW ≤ 1000, respectively, showing that hydrolysates with MW ≤ 1000 were the most active. Regarding the antioxidant activity, the most active were hydrolysates with MW ≥ 10,000, followed by crude hydrolysates, MW between 1000 and 10,000, and MW ≤ 1000 (IC 50 were about 5, 8, 10, 35 mg/mL). Similar trend was observed for yellowtail fish scale hydrolysates [ 129 ]. Similarly, Jeon et al. [ 130 ] analyzed ACE inhibitory and antioxidant activity of cod frame protein hydrolysates of 30, 10, 5 and 3 KDa. The 3 KDa hydrolysates had the highest ACE inhibitory activity (IC 50 = 0.08 mg prot/mL), while the 5 KDa hydrolysates had the highest antioxidant capacity (~40% oxidant inhibitory ratio).
As reported by Theodore and colleagues [ 131 ], low MW peptides have generally higher ORAC values while high MW peptides have higher FRAP and DPPH radical scavenging activities [ 131 ]. However, it is sometimes difficult to compare bioactivities between different peptides analysed with different techniques because radical scavenging activities are not always reported with the same units. Recently, Vázquez et al. [ 107 ], in order to valorise farmed fish processing wastes, studied fish protein hydrolysates obtained by alcalase hydrolysis from turbot S. maximus head, viscera, trimming and frames. Antioxidant and antihypertensive possible activities were evaluated and results showed that viscera hydrolysates, containing peptides above 1000 Da and below 200 Da, were the most active with scavenging activity of 65.15% (DPPH), 12.81 µg BHT/mL (ABTS), 8.03 µg Trolox/mL (Crocin) and 81.9% of ACE inhibitory activity.
Other peptides from fish skin showed antioxidant properties, such as PYSFK, GFGPEL and VGGRP from the grass carp Ctenopharyngodon idella [ 132 ], AVGAT from Thornback ray [ 123 ], DPALATEPDMPF, EGL and YGDEY from Nile tilapia O. niloticus [ 133 , 134 ], PFGPD, PYGAKG, YGPM from Spanish mackerel Scomberomorous niphonius [ 134 ], GSGGL, GPGGFI and FIGP from blue leatherjacket Navodon septentrionalis [ 135 ], GATGPQGPLGPR, VLGPF and QLGLGPV from seabass Lates calcarifer [ 136 ], FDSGPAGVL and DGPLQAGQPGER from Jumbo squid Dosidicus gigas [ 137 ], PAGT from Amur sturgeon [ 138 ], P1 and P2 from pollack [ 139 ], and LSGYGP from the tilapia O. niloticus [ 140 ]. Finally, generic hydrolysates from snappers Priacanthus macracanthus and Lutjanus vitta and sole skin [ 141 , 142 , 143 ] and hydrolysates from the herring Clupea harengus whole, body, head and gonads had antioxidant activity [ 144 ]. TCSP and TGGGNV from the cod Gadus microcephalus showed both anti-hypertensive and antioxidant properties [ 145 ].
Fish frame protein hydrolysates are also a rich source of antioxidant peptides, including GSTVPERTHPACPDFN isolated from Hoki ( Johnius belengerii ) [ 146 ], N-terminal RPDFDLEPPY purified from yellowfin sole ( L. aspera ) [ 147 ], LPHSGY isolated from Alaska pollack hydrolysates MW < 1KDa [ 148 ]. Moreover, antioxidant activity was identified in the peptide VKAGFAWTANQQLS isolated from tuna backbone protein, that inhibited the lipid peroxidation in linoleic acid emulsion system and quenched free radicals (DPPH, hydroxyl and superoxide) in a dose-dependent manner [ 149 ].
Several studies pointed out the antioxidant activity of hydrolysates of collagen and its derivatives, obtained from different fish byproducts, including Gadous macrocephaius skin [ 150 ], shark cartilage [ 94 ], yellowfin tuna ( T. albacares ) skin; particularly low-weight peptide (< 3kDa) showed higher radical scavenging properties [ 92 ]. Collagen polypeptide from tilapia skin (MW< 3000Da) had protective effects against injuries to the liver and kidneys of mice induced by d-galactose by reducing oxidative stress [ 151 ]. Moreover, collagen-derived peptides that have shown antioxidant activity were: GLFGPR peptide from L. calcarifer skins [ 152 ], HGPLGPL peptide from J. belengerii skin [ 137 ], GPRGTIGLVG, GPAGPAG, and GFPSG from scales of Pseudosciaena crocea [ 153 ], HGPHGE, DGPKGH, and MLGPFGPS from Katsuwonus pelamis scales [ 100 ], GPDGR, GADIVA, GAPGPQMV, AGPK, and GAEGFIF from K. pelamis bones with GADIVA and GAEGFIF that showed strongest antioxidant activity [ 154 ], GPE, GARGPQ, and GFTGPPGNG from Sphyrna lewini cartilage [ 155 ], YGCC, DSSCSG, NNAEYYK, and PAGNVR purified from Theragra chalcogramma skin [ 156 ]. Moreover, peptides NHRYDR and GNRGFACRHA from Magalapis cordyla skin and Otolithes ruber skin, respectively, reduced PUFAs peroxidation [ 157 ].
Regarding neuroprotective activities, QGYRPLRGPEFL isolated from Skate ( R. kenojei ) skin showed β-Secretase inhibitory activity (IC 50 value of 24.26 μM, [ 158 ]), while collagen hydrolysates isolated from Salmon ( O. keta ) skin had anti-acetylcholinesterase activity, learning and memory effects [ 159 , 160 ]. Xu et al. [ 159 ] investigated the neuroprotective effects of salmon collagen peptides (intragastrically administered at 0.33 g/kg, 1.0 g/kg and 3.0 g/kg body weight) in male rats with perinatal asphyxia. Results showed facilitated early body weight gain, long-term learning and memory, reduced oxidative damage and acetylcholinesterase activity in the brain, and increased hippocampus phosphorylated cAMP-response element binding protein and brain derived neurotrophic factor expression. Salmon learning and memory effects were evaluated also by Pei et al. [ 160 ] in 20-month-old female C57BL/6J mice fed with O. keta collagen peptides (0.22%, 0.44% or 1.32% wt/wt) compared to aged control mice. Memory effects were evaluated by step-down test, Morris water maze, oxidative stress, expression of brain-derived neurotrophic factor, and postsynaptic density protein 95. The positive results suggested the potential use of the salmon collagen peptide for functional foods to relieve aging memory deficits [ 160 ].
Collagen peptides from fish waste are an emerging substance also in the cosmetic field. A randomized triple-blind, placebo-controlled, parallel study was performed to evaluate the efficacy of collagen from Pangasius hypophthalmus on skin wrinkles and elasticity. Participants were females between the ages of 45–60 and displayed visible signs of natural and photoaging on their face and they consumed 10 g of hydrolysed collagen powder daily. At week 12, participants in the collagen consuming group reported improvements in overall skin score (9%) and wrinkle (15%), elasticity (23%), hydration (14%), radiance (22%), and firmness (25%) [ 161 ]. Moreover, a fish collagen drink was tested for improvement of skin aging. Collagen drink improved cell viability of UVA irradiated human skin fibroblast (CCD-966Sk), their mitochondrial activity was restored and reactive oxygen species (ROS) production triggered by UVA treatment was reduced. Moreover, collagen drink promoted the expression of SOD1, SOD2, CAT genes, and improved both collagen and elastin production [ 162 ]. In addition, skin-derived peptides from the sutchi catfish P. hypophthalmus showed matrix metalloproteinase (MMP) inhibitory activity [ 163 , 164 ]. In particular, Lu et al. [ 162 ] found that cod skin gelatin hydrolysates were especially active at 0.1 mg/mL inhibiting MMP-1 expression in fibroblasts irradiated with UVB of 20 mJ/cm 2 . Two peptides, GEIGPSGGRGKPGKDGDAGPK and GFSGLDGAKGD, were isolated from these hydrolysates and found to exhibit a significant inhibition of MMP-1, phosphorylated extracellular signal-regulated kinase (p-ERK) and p-p38. GEIGPSGGRGKPGKDGDAGPK also inhibited phospho–c-Jun N -terminal kinase (p-JNK) in mitogen-activated protein kinase (MAPK) signaling pathways. Pyun et al. [ 164 ] analysed the anti-photoaging effects of collagen tripeptide (167–333 mg/kg/day oral administration for 14 weeks) on a hairless mouse model. Mice showed significantly reduced wrinkle formation, skin thickening, transepidermal water loss, skin hydration and prevented UVB-induced MMP-3 and -13 expression, and MMP-2 and -9.
Hydrolysed fish collagen has been found to induce osteoclastin differentiation of bone marrow mesenchymal stromal cells (BMSCs), improving expression levels of anti-inflammatory mediators, such as inteleukin-6 (IL-6), TGF-β1, and prostaglandin E2 (PGE2). Moreover, collagen hydrolysed reduces the expression level of pro-inflammatory mediator, such as interleukin 1 beta (IL-1β) and tumor necrosis factor alpha (TNF-α) [ 165 ].
Subcritical water-hydrolysed fish collagen peptide (SWFCP) from tuna skin shown inhibitory activity of adipocyte differentiation, which was followed by decreased expression of CCAAT-enhancer-binding protein-α (C/EBPα), peroxisome proliferator-activated receptor-γ (PPAR-γ), and adipocyte protein 2 (aP2) genes, regulators of differentiation of adipocytes [ 166 ].
Peptides isolated from only two species, SPGSSGPQGFTG, GPVGPAGNPGANGLN, PPGPTGPRGQPGNIGF from the halibut Hippoglossus stenolepis and IPGDPGPPGPPGP, LPGERGRPGAPGP and GPKGDRGLPGPPGRDGM from the tilapia O. niloticus, have been reported to have anti-hyperglycemic activity [ 135 ]. Daily administration of tilapia skin gelatin hydrolysate (750 mg/kg/day) for 30 days was more potent to improve the glucose tolerance in streptozotocin-induced diabetic rats than hydrolysates from halibut skin (750 mg/kg/day), inducing the inhibition of plasma dipeptidyl peptidase IV (DPP-IV) activity, enhancement of glucagon-like peptide-1 (GLP-1) and insulin secretion. Hydrolysates from frame of the sole Limanda aspera were the only one that were reported to have anticoagulant activity [ 167 ]. The identified protein of 12.01 KDa, named yellowfin sole anticoagulant protein (YAP), inhibited the activated coagulation factor XII (FXIIa) and antagonized the platelet membrane glycoprotein integrin to arrest platelet aggregation by inhibiting thrombosis in vitro . In addition, hydrolysed collagen promoted osteogenic differentiation of bone marrow mesenchymal stromal cells (BMSCs) which preserved their immunomodulation ability. An anticancer peptide, FIMGLY, was also isolated from Raja porosa cartilage, and it was found to be active against HeLa cells [ 168 ]. Collagen hydrolysates obtained from skin of Aluterus monoceros have been found to possess anticancer, antidiabetic and wound healing properties, thus suggest that collagen may represent a flexible and multipurpose material [ 169 ].
Oral administration of collagen peptides from Oncorhynchus keta skin in growing rats promoted size, mineral density of long bone in treated rats [ 170 ]. In addition, collagen peptides from skin of Gadiformes fish species had chondroprotective effects in the osteoarthritis-induced model [ 171 ]. Finally, peptides from frame and bone of the hoki J. belengerii (VLSGGTTMYASLYAE and a phosphopeptide, named FBP, respectively) and frame of the pollack T. chalcogramma (VLSGGTTMAMYTLV) had calcium-binding properties, and peptides (GPAGPHGPPGKDGR, AGPHGPPGKDGR, AGPAGPAGAR) from Pacific cod skin had iron-chelating activity [ 172 , 173 , 174 , 175 ].
3.3. Chitin
An analysis of the business implications due to the Covid-19 crisis led to resizing the parameters of the global market for chitin and chitosan derivatives. It was estimated at 106.9 thousand metric tons in 2020 [ 176 ], and it is expected now to reach a revised size of 281.7 thousand metric tons by 2027 with an increase at a CAGR of 14.8% in the period 2020–2027 [ 177 ]. Before the Covid-19 pandemic occurrence, chitosan global market was expected to increase at a CAGR of 24.7% from 2020 to 2027, while it is estimated now to reach a 16.9% CAGR and 173.9 thousand metric tons by the end of the analysis period. Chitosan is used in large quantities in the production of several consumer products, i.e., antiseptics, food items, cosmetics, medicines, and textiles, and is very attractive for its numerous biological properties and as a therapeutic agent because of its antibacterial and antifungal characteristics. It was reported that the annual production of chitosan accounted for 2000 tons, with the main sources being shrimp and crab shell residues [ 178 ]. China should achieve a market size of 48.5 thousand metric tons by the year 2027 with a CAGR of 14.1% in the period 2020 to 2027, while in Europe, Germany is expected to reach a 10.6% CAGR.
Chitin is a long chain odourless/tasteless amino polysaccharide of white or off-white colour in its pure state, composed of N -acetyl- β -D-glucosamine units and monomers attached via β (1→4) linkages [ 179 ]. It is considered the second most abundant high MW natural biopolymer after cellulose, to which it is strongly similar in structure. The sole difference lies in the substitution of the secondary hydroxyl groups with acetamide groups on the alpha carbon atom of the cellulose molecule [ 180 , 181 ]. An important chitin derivative is the chitosan, obtained by deacetylation of chitin, and with a molecular structure in which the 2-amine-2-deoxy-D-glucopyranose units predominate [ 182 ]. Differently from most of the natural polysaccharides that are of neutral or acidic nature, chitin and chitosan are highly basic [ 183 , 184 ].
On the basis of the different arrangement of chains in the crystalline regions, chitin has been classified in three forms, namely the α-, β-, and γ-forms [ 10 ]. Each of them provides to the chitin specific properties, which could be affected also by the different degrees of deacetylation. All these structural combinations led to a wide range of possible activities of interest for several biotechnological industries and tissue engineering. According to [ 185 ], it is really hard to achieve the suitable purity degree due to some compounds that naturally occur within chitin, for which the complete elimination is difficult. Indeed, chitin is often included in a complex matrix including proteins and calcium carbonate, with the establishment of strict interactions. Thus, the common procedure employed for the extraction of chitin requires the removal of these associated components, and follows two sequential chemical steps: deproteinization, for the removal of proteins; demineralization, aimed at removing the inorganic component. In some cases an additional step of decolorization/deodorization is applied to remove pigments [ 186 , 187 ]. The experimental extraction procedure is a very delicate and determinant phase for the final application, because purity and crystallinity levels depend on it [ 188 ].
Until now, the main source of chitin and derivatives were members of the phylum Arthropoda, including crustaceans and insects, provided of an exoskeleton predominantly constituted of chitin [ 10 , 189 , 190 ] and therein references. Recently, fish wastes have acquired also great attention as potential sources of chitin and derivatives, with particular regard for fish scales, but the use of these kinds of sources is less investigated. Taking into account that the most commonly used resources for the extraction of chitin and chitosan are crustaceans, many approaches focused on the use of fish wastes have employed the same general procedure, with slight variations in some cases. The numerous efforts performed in the isolation of chitin and chitosan from crustaceans reported generally a final yield of chitin for an amount ranged from 14% to 25%, but the use of fish scales seems to be equally promising.
The available literature includes studies investigating the use of scales obtained from four fish families, including the carp, tilapia, red snapper, and parrotfish ( Table 3 ). The first attempt was carried out by Zaku and co-authors [ 191 ], which used scales of common carp fish Cyprinus carpio to isolate chitin. The fish scales were treated with maceration procedure after drying for three days, washed from residual minerals with acid (HCl 1M) at 30 °C and finally rinsed with deionized water and deproteinized at 95 °C. The yield of chitin expressed as percentage of the starting raw materials was 20.49%, thus comparable with results obtained from crustaceans.
Chitin/Chitosan from fish waste and their possible applications.
| Compound | Byproduct | Source | Applications | Reference |
|---|---|---|---|---|
| Chitin | Scales | Not specified | [ ] | |
| Chitin, Chitosan | Scales | Not specified | [ ] | |
| Chitin, Chitosan | Scales | Not specified | [ ] | |
| Chitosan | Scales | Not specified | [ ] | |
| Chitin, Chitosan | Scales | Not specified | [ ] | |
| Chitin | Scales | Not specified | [ ] | |
| Chitin | Scales | Not specified | [ ] | |
| Chitin, Chitosan | Scales | sp. | Not specified | [ ] |
| Chitosan | Scales | Coagulation-flocculation treatment for iron removal | [ ] |
Generally, the physicochemical properties of chitin are investigated through techniques including IR spectroscopy and scanning electron microscopy (SEM), but in some cases the approaches included more specific techniques. This is the cases reported by Kumari et al. [ 192 , 193 ], which obtained and characterized chitin and chitosan from scales of Labeo rohita by using X-ray diffraction, elemental analysis, Fourier-transform infrared spectroscopy (FTIR), SEM and differential scanning calorimetry (DSC). For these studies the scales were cleaned, washed, and dried in sunlight for 4 days and then subjected to demineralization and deproteinization. In addition to the studies of Kumari and co-authors, other researchers have also investigated the potential of the L. rohita species, even if they have focused more on the purification of chitosan, considered a compound with greater versatility of application. After a procedure including deproteinization and demineralization, the yield of prepared chitin was found to be 22.36%, while for chitosan a final yield of 7.72% was achieved [ 194 ]. The degree of deacetylation (DDA) is regarded as an important parameter for the effective application of chitosan, and the recognized good standard for a typical commercial chitosan ranges between 66% and 95% [ 195 ]. Muslim et al. [ 194 ] which their results, strongly encouraged the possible introduction of chitosan in several application fields, as they detected a %DDA of chitosan of 78.2%. Uawonggul et al. (2002) [ 196 ] and Boarin-Alcalde and Graciano-Fonseca [ 45 ] managed to obtain chitin and chitosan from scales of Nile tilapia ( Tilapia nilotica ). In particular, Boarin-Alalde and Graciano-Fonseca [ 45 ] tried to adjust the procedure generally used to isolate these compounds from crustaceans, including a pretreatment, demineralization, deproteinization, depigmentation and deodorizing and finally deacetylation to obtain chitosan. From 50 gr of raw materials (fish scales) the yield of chitin was the 20%, while the authors achieved a total amount of purified chitosan up to 39%. These percentages are lower than those obtained by crustaceans, but quite in line with other reports on the use of fish scales as source of isolation. More recently interesting results have been obtained from typical tropical saltwater fishes species, namely red snapper and parrotfish. Rumengan et al. [ 197 ] obtained a chitin yield of 45% and 33%, from parrotfish ( Chlorurus sordidus ) and red snapper ( Lutjanus argentimaculatus ), respectively, while Takarina and Fanani [ 198 ] achieved DDA values up to 75% for chitin and 90.83% for chitosan, by suggesting a huge potential for red snapper fish scales. An even higher value of deacetylation was retrieved in the chitosan obtained from Papuyu fish scales, with 97.40% against the 93.80% of commercial chitosan from shrimp shell [ 199 ]. In general, the studies conducted so far are limited to evaluating the possibility of fish scales as a possible alternative source, but are similar in scope and final considerations. The promising results strongly encouraged the exploitation and enhancement of this waste, whose production takes on increasing connotations every year.
Fish Chitin Applications
The general applications of chitin and chitosan have been extensively reviewed by several authors [ 10 , 42 , 200 , 201 ]. The properties possessed by chitin and chitosan, as for example ability to form polyoxysalts and films, to chelate metals and the optical structure characteristics [ 202 ] make them attractive compounds for utilization in a number of fields, such as the medical, pharmaceutical, food and cosmetic industries, nutraceuticals, bioremediation, gene therapy and cosmetics. The different approach led to the detection of a more crystalline structure for chitosan than for chitin and fish scale, by confirming its possible use as food supplement, drug preparation and water treatment. Although the results obtained are promising, the available studies that analysed the use of fish scales as a source of chitin and chitosan are limited to the chemical purification and characterization of these compounds, but do not investigate deeper their potential applications. Most of the researchers involved in this field only suggest the possible uses on the base of the chemical characteristics of chitin and chitosan. The only exception is represented by the study of Irawan et al., [ 201 ] which demonstrated the promising employment of chitosan in environmental recovery. In the bioremediation field, diverse chitin derivatives possess specific properties useful for treatment of contaminants, as the different functional groups they have could easily interact with heavy metal ions by helping their removal in aqueous solution [ 190 , 203 ]. The chitosan extracted from the fish scales of Papuyu fish has been proved to improve the removal of iron in the groundwater from 11.80 mg/L to 3.43 mg/L, by evidencing higher efficiency in coagulation/flocculation treatment than the commercial chitosan from shrimp shell [ 199 ].
Despite the inconsistency of available information, we can obtain some interesting insights from the existing studies referring to molecules extracted mainly from crustaceans, which, thanks to the intense work done in recent decades for their applicability, are considered more fruitful than cellulose [ 204 ]. In the attempt of summarize the huge amount of possible applications, notable uses have been reported in the medical and pharmaceutical fields due to the proved biological activities as such as antimicrobial, antioxidant, antitumor immunoadjuvant, antithrombogenic, anticholesteremic and bioadhesivity. Antibacterial activities have been reported for chitosan by several authors [ 205 , 206 ], and were also related to the MW of chitosan by suggesting interaction with cell permeability affecting the inhibition action [ 207 , 208 ]. A mechanism based on the chitosan ability to form permeable films has been proposed at the base of the observed antifungal specific activities [ 209 ]. Direct and indirect activity against tumoral cells have been extensively demonstrated for chitosan and its derivatives [ 210 , 211 , 212 ]. The many possible conformations of chitin and chitosan (fibers, powders, films, sponges, beads, gels, capsules) allow the exploitation of chitosan for drugs administration in several ways and for tissue engineering and wound care dressing [ 213 , 214 ]. Beneficial properties, such as biocompatibility, biodegradability, film-forming capacity and gas and aroma barrier are then at the base of the suggested and proved use for food preservation, packaging, and as colour stabilization agents [ 215 , 216 , 217 , 218 , 219 ]. This is particularly true for chitosan, which exhibited a higher solubility in various acidic solvents than the poor soluble and reactive chitin, and possess antimicrobial activity against many pathogenic microorganisms [ 220 ]. Films obtained from chitosan showed flexibility and resistance and an excellent ability to form oxygen barriers, thus ideal for storage of fruits, vegetables, eggs and dairy, cereal, meat, and seafood products.
In any case, theoretically, all these applications can also be envisaged for chitin and chitosan extracted from fish scales, considering the chemical data obtained in terms of net yield and degree of deacetylation. Certainly, further investigations should be carried out to verify the real effectiveness, and to evaluate the different yields and quality levels possible correlated to the species of fish considered, as the different environmental conditions or particular adaptations could affect the composition of the scales themselves.
The global fish oil market size was valued at $ 1,905.77 million in 2019, and is estimated to reach $ 2844.12 million by 2027 with a CAGR of 5.79% from 2021 to 2027 [ 221 ]. The EU produces approximately 120,000 tons of fish oil each year, for which Denmark is the largest producing nation. This production is mainly driven by the great demand of fish oil as ingredient in the aquaculture industry, now consuming as much as 90% of global fish oil supplies [ 222 ]. However, there is also good potential for the high end/high value markets, especially from fish byproducts. The increasing global trend of fish products processing will increase the volumes of byproducts. In 2016, the global production of fish oil from byproduct accounted for 26% of the total fish oil production [ 223 ]. In both the fishing and aquaculture industries, oil and fats represent a significant fraction of finfish processing waste and the amount of which depends upon the fat content of the specific fish species, the distribution of fat in fish parts, its age, sex, nutritional status, health, and time of year determine the amount of oil/fats [ 41 ]. For example, it is well noted that the visceral mass of fish discards has a significant amount of oil or fat apart from proteins [ 224 ]. Fish oil is found in the flesh, head, frames, fin, tail, skin and guts of fish in varying quantities. Generally, fish contains 2–30% fat, and about 50% of the body weight is generated as waste during the fish processing operation [ 225 ], meaning that there is a great potential for valorisation of this waste, mainly for human consumption or to produce biodiesel. Fish oil contains mainly triglycerides of fatty acids (glycerol combined with three similar or different acid molecules) with variable amounts of phospholipids, glycerol ethers and wax esters. It is considered the most nutritious and most digestible ingredient for farmed fish. The lipid composition in fish is quite different from land animal lipids and vegetable oils due to the large quantity long-chain PUFAs, including eicosapentaenoic acid (EPA, C20:5, n-3) and docosahexaenoic acid (DHA, C22:6, n-3) also known as omega-3. These fatty acids, which cannot be synthesized by human body, cover a wide range of critical functions for human health [ 226 , 227 , 228 ]. Among the best source of oily (EPA and DHA) fish, there are salmon, herring, mackerel, anchovies, sardines and tuna [ 229 ]. Fish are not able to synthetize omega-3 but they need to obtain them from the external, through algae and microalgae or plankton in their diets. A wide range of techniques have been used to extract oil from whole fish or fish waste and reported in several reviews [ 41 , 228 , 230 ]. The selection of the most suitable method depends on different factors, especially by the nature of the waste and by the final application of the oil, whose biodiesel production and food supplements are the most common [ 46 , 231 ]. The applied technologies range from chemical and enzymatic processes, cooking and pressing to more recent green technologies such as microwave and supercritical fluids. A recent work by Mendez and Concha [ 46 ] analysed in depth the different extraction methods applied for the production of omega-3. Summarising the main aspects, wet pressing and chemical extraction by solvents are the most common approaches, although the high pressures/temperature and the residual presence of solvents (which needs to be removed using a surplus of energy) limit their use. Less harsh methods use enzymatic hydrolysis by proteases. Different enzymes can be applied, but several studies demonstrated that the use of alcalases is a more efficient process [ 232 , 233 ]. In general enzymatic hydrolysis is a quick and easily reproducible method; it prevents extreme physical and chemical treatments; compared to chemical hydrolysis, it has the advantage of avoiding generating chemical waste, besides being more easily controlled showing great potential for fish waste valorisation. Recently, Araujo et. al., demonstrated that by increasing the initial concentration of alcalase (Eo from 0.94 AU/1 to 4.68 AU/1)—at a constant initial substrate concentration (So = 25 g of protein per liter)— the degree of hydrolysis values and the oil yield increased, obtaining 430 g of protein hydrolysate, 10 g of collagen and 350 g of oil from 1000 g of fish waste [ 234 ]. A disadvantage of enzymatic hydrolysis could be the high price of some enzymes which, most of the time, cannot be recycled. In a view of sustainability, more environmentally friendly approaches are needed. In this context supercritical fluid extraction (SFE) is an emerging extraction technology using solvents, mainly CO 2 , that has many advantages compared with traditional techniques because it uses moderate temperatures, reduces the lipid oxidation during the extraction process, allows a selective extraction of low polar lipid compounds, avoiding the co-extraction of polar impurities such as some inorganic derivatives with heavy metals [ 235 ]. Several studies demonstrated that this approach provides the same efficiency of oil extraction from fish waste but in a more sustainable way. The application of SFE from the viscera of African catfish Clarias gariepinus and from common carp C. carpio allowed to obtain a yield of oil which was comparable with the yield extracted using the Soxhlet method [ 236 , 237 ]. Furthermore, another cleaner and greener method for the oil extraction, which includes the use of microwaves, has been applied by [ 238 ], for biodiesel production. They showed that the microwave lipid extraction was approximately 50% more efficient compared to conventional solvent lipid extraction (Bligh and Dyer method). In the same study they demonstrated also that microwave-assisted transesterification reaction in the presence of KOH catalyst for 10 min at 65 °C lead to an efficient conversion into biodiesel. The Gas Chromatography–Mass Spectrometry (GC/MS) analysis confirmed the presence of good quantity of palmitoleic acid, palmitic acid, oleic acid and eicosapentaenoic acid, which are essential biodiesel components [ 238 ]. Depending on the extraction methodology, the extracted crude oil could contain impurities [ 239 ], and requires a purification process to reach quality features that make it acceptable for human consumption [ 240 ]. Moreover, although fish oil possesses several health benefits, the presence of highly unsaturated fatty acids results in auto-oxidation of fish oil, so the final application of the oil product should be taken into consideration since the first stage of the processing.
Fish Oil Applications
Industrial fish processing operations generate a significant amount of wastes, which contain long-chain fatty acids and that can be utilized in a variety of markets [ 230 ] including industrial uses, food, feed, and aquaculture and nutraceutical applications ( Table 4 ). The reason for the great interest in fish oil is that it contains two important PUFAs called EPA and DHA or otherwise called omega-3 fatty acids. The two main PUFA applications are as feed/food supplements and biofuel production.
Oil from fish waste and their possible applications. MIC, minimum inhibitory concentration.
| Compound | By-product | Source | Applications | Activity | Reference |
|---|---|---|---|---|---|
| Oil | Viscera | Supplement in animal feeding | Reduction in cholesterol (9.2 to 16.6%) and in triglyceride (1.5 to 3.1%) | [ ] | |
| Cod liver oil | Liver | fish | Supplement in bacterial growth media | 14.8 U/mL (lipase production) | [ ] |
| Omega -3, -6, -7, -9 fatty acids | Head, tissues | Antimicrobial | MIC: 25 and 12.5 (% ) | [ ] | |
| oil | Viscera, fins, heads, skin, scales and mixed waste | Biodiesel | Not specified | [ ] | |
| Oil | Viscera | Biodiesel | Not specified | [ ] | |
| Fish Oil Methyl Ester (FOME) | Fish waste | Not specified | Biofuel | Not specified | [ ] |
| Liver oil | Ray liver waste | | Antioxidant | IC 0.92 to 2.1 mg/mL (DPPH) | [ ] |
| Sulphated fatliquor | Fish waste | Not specified | Lubricant | Not specified | [ ] |
The aquaculture sector is the predominant market and requires oils with low levels of oxidation, low levels of contaminants, and consistent quality. The nutraceutical market requires oils low in oxidation and contaminants, but also with high levels of omega-3 fatty acids [ 241 ]. The omega-3 fatty acids are very well known to have beneficial bioactivities including prevention of atherosclerosis, arrhythmias, reduced blood pressure, benefit to diabetic patients, protection against manic-depressive illness, reduced symptoms in asthma patients, protection against chronic obstructive pulmonary diseases, alleviating the symptoms of cystic fibrosis, improving survival of cancer patients, reduction in cardiovascular disease and improved learning ability [ 173 , 226 , 227 , 228 , 242 , 243 , 244 , 245 ]. Medical research has provided strong evidence to indicate that a diet high in fish and marine omega-3 is linked to a lowered risk of cognitive decline and Alzheimer’s disease (AD) [ 246 ] and some clinical trials suggests that supplementation by n –3 PUFA extracted from fish improves cognitive functioning in elderly adults with mild to no cognitive impairment [ 247 ].
Considering the recognised benefits of EPA and DHA on human health, alternative ways of supplying dietary omega-3 to the consumers have been explored. One of the approaches is to enrich chicken meat with omega-3 fatty acid derived from sustainable marine sources. To investigate this aspect, Moula Ali added different concentrations of fermentative recovered fish oil (FFO) to broilers’ diet, which resulted in incorporation of EPA + DHA in the animal, observing a consequent reduction in cholesterol (ranging from 9.2 to 16.6% compared with control) and triglyceride (ranging from 1.5 to 3.1% compared with control) concentrations in serum, liver and meat of birds fed with FFO. This demonstrated that oil from fish waste can indirectly benefit human health through chicken meat [ 248 ]. Several studies demonstrated also the potential of fish oil as antioxidants. In their study, Sellami et al. [ 249 ] extracted oil from the waste liver of three ray species evaluating the extract composition and scavenging activity. The fatty acid profiles exhibited a dominance of unsaturated fatty acids (UFAs) exceeding for all samples 65% of the total fatty acids content. The major n-3 PUFAs were EPA (C20:5) and DHA (C22:6) with contents varying from 3.36 to 5.51% and from 9.07 to 30.50% respectively. Interestingly, they found that these oils contained also carotenoids and phenolic compounds, displaying antioxidant activity comparable to that of olive oil. Oil from fish waste was also successfully applied at microbiological level both as bacterial growth substrate and as antimicrobial. The antioxidant as well as the immunomodulatory properties of the combination of fish (salmon) oil with plant extracts was reported by [ 250 ], demonstrating that the synergistic effect lead to an increasing of bioactivity compared with fish oil extract alone.
Oil from waste fish has been used also to induce the production of bacterial enzymes. In particular, it is known that microbial lipase production is highly influenced by medium components like nitrogen sources, carbon sources such as fatty acids, triglycerides and carbohydrates which can stimulate or repress lipase production. Cod liver oil 1.5% added to the growth medium of Staphylococcus epidermidis CMST Pi 1 demonstrated to be a suitable triglyceride source to increase lipase production (14.8 U/mL) compared with castor oil, palm oil, and other vegetable oils [ 251 ]. On the other side, Inguglia et al. [ 252 ] qualitatively characterized and investigated the antimicrobial effects of the fish oil extracted from S. salar waste samples derived from Italian fish markets and evaluated the potential use of these compounds for treating pathogen infections. By using GC/MS they showed that the specific fatty acid composition of the salmon waste oils was enriched in MUFAs and PUFAs, with special reference to omega-3, -6, -7, -9 fatty acids. The oleaginous extract was tested against two Gram-positive and Gram-negative, respectively S. aureus ATCC 6538 and ATCC 25923 and P. aeruginosa ATCC 9027 and ATCC15442 demonstrating an inhibition effect with a MIC of 25 and 12.5%, presumably attributed to their action on cell membrane alteration and destabilisation [ 253 ].
Another relevant application of oil from fish waste is the production of eco-friendly fuels, especially biodiesel. Waste oils are potentially advantageous over petroleum and virgin vegetable oil based fuels due to waste utilization, and an overall lowering emissions over the life cycle of fuel production, disposal, lower price (25 cents per gallon for fish oil compared to $1.19 per gallon for diesel fuel), and similar calorific value to petroleum distillates [ 254 ]. For these reasons, several studies explored and confirmed the potential of fish waste oil for biodiesel production. In a study conducted by Martins et. al. [ 255 ] the physicochemical features of the fish-based biodiesel obtained from tilapia waste oil were checked in accordance with the standard requirements established by Brazilian National Petroleum Agency. They confirmed that the obtained biodiesel is in accordance with the specifications of specific mass, kinematic viscosity, water content, acidity level, flash point and oxidation stability proving the potentiality of using residual oil from tilapia waste as a quality raw material in biodiesel production. The same results were obtained by the pyrolysis at 525 °C of waste fish oil as an animal source of triglycerides, showing that it is possible to obtain biofuels with a good similarity to petroleum-based fuels [ 256 ]. Velasquez et. al., showed that oil from viscera of Mexican snook (Centropomus Poeyi), black seabream (Spondyliosoma cantharus), king mackerel (Scomberomorus cavalla) and striped mojarra (Eugerres plumieri) collected in a fish market can be successfully converted into fatty acid methyl ester by enzymatic catalysis [ 30 ] and Prakash et. al., compared the performance and emissions of Fish Oil Methyl Ester (FOME) with fossil diesel and demonstrated that FOME could be used as alternative fuel to diesel in stationary diesel engines [ 32 ].
A recent application of oil from fish waste was its use as fatliquoring agent in leather processing. As a matter of fact, the physical and mechanical properties of the leather lubricated with the sulphated fish oil fat (fish oil converted into fatliquor by a sulphation process using sulphuric acid) were better than those processed using the commercial fat-liquoring agent [ 48 ]. The authors calculated the techno-economic feasibility of this kind of application. Including raw materials, transportation and manpower, the price of the fish oil fatliquor is about 1.98 USD/L, which can save 0.07–0.35 USD/L compared to the commercial fatliquoring agent, suggesting that tanneries would be inclined to employ fish waste oil-based fatliquoring process due to its benefits in terms of environmental safety and cost competitiveness.
3.5. Enzymes
The enzyme market size was around $ 6.3 billion in 2017 and will see growth of around 6.8% in the CAGR through 2024. The expansion of the food and beverage industry due to the growing needs of the population, including the need to improve the flavor, quality and texture of food is leading to continued growth of enzymes market. Furthermore, the growth of this sector is also attributable to the enormous applications that enzymes can have in the detergent industry, increasing the effectiveness of detergents by aiding in stain removal [ 257 ].
The bioprospecting of aquatic organisms has led to the discovery of several enzymes with catalytic properties potentially useful for several biotechnological applications. These organisms living in diverse and often hostile environmental conditions, incomparable with the terrestrial habitats, have developed enzymes with unique characteristics [ 26 , 258 , 259 ], capturing the interest of many researchers.
The internal organs, including stomach, pancreas, and intestines, are the most important fish byproducts in terms of the number of enzymes found, many of which are cold active, present high catalytic activities also at relatively low concentrations, and stability in a wide range of pH. The complete list of isolated and characterized enzymes from fish have been reviewed by [ 10 , 43 ].
Proteases represent the largest group of enzymes naturally found in fish. These enzymes catalyse the hydrolysis of peptide bonds, through different mechanisms of action [ 260 ]. They are referred to as exo-peptidases (or peptidases), when they cleave the terminal amino acid of the polypeptide chain, and as endo-peptidases (or proteinases), when the peptide bonds they cleave are internal. Digestive proteolytic enzymes from fish byproducts belong to four different groups, according to the substrate specificity, classified as aspartic proteases (e.g., pepsin, cathepsin D), serine proteases (e.g., trypsin, chymotrypsin), thiol or cysteine proteases (e.g., calpain, cathepsins B, H, L), and metalloproteases (e.g., collagenases) [ 258 , 261 ]. The most extensively studied proteases that are available in fish include pepsin, trypsin, chymotrypsin, and collagenase. Fish pepsins are generally located within the fish stomach and have greater activity in acidic conditions [ 262 ]. However, many fish species secrete at least two different pepsins with different optimal pH, commonly referred to as pepsin I and pepsin II [ 263 , 264 ]. As reviewed by [ 44 ], these enzymes have been isolated from the gastric mucosa of several fish species, such as sardine, capelin, cod, salmon, shark, mackerel, orange roughy, tuna, trout, and carp. Trypsin is involved in the hydrolysis of peptide bonds in the carboxyl-terminal ends of lysine and arginine residues [ 265 ]; it plays an essential role in the digestion of ingested proteins and is also responsible for the activation of the precursor forms of several other digestive proteinases including chymotrypsin [ 262 ]. The use of fish trypsin is increasing enormously, thanks to the unique features of these enzymes including the stability and the high catalytic activity in a wide range of pH and temperature values, including hard conditions (38–70 °C; pH value 8–11) [ 266 ]. As reported by [ 43 ], fish trypsins have been isolated from sardines, capelin, salmon, cod, bluefish, anchovy, Atlantic croaker, carp, aquacultured tilapia, ray fish, mackerel, threadfin hakeling, red snapper, smooth hound. Compared to trypsin, less work has been reported on fish chymotrypsins, isolated so far from carp, capelin, herring, Atlantic cod, rainbow trout, spiny dogfish, sardine. Collagenases are enzymes capable of hydrolysing the polypeptide backbone of native collagen, without denaturing the protein [ 267 ], and are classified as metallocollagenases and serine-collagenases, with different physiological roles. Unlike metallocollagenases, zinc-containing enzymes, which show exclusive specificity for collagen, serine-collagenases have a wide proteolytic activity in addition to collagenolytic activity [ 258 ]. Collagenases have been isolated from epithelial, cartilaginous, bony tissues, and digestive tracts of several fish [ 43 ].
Besides proteases, another family of enzymes widely found in fish are lipases. These enzymes are glycerol-ester hydrolases, which catalyse the hydrolysis of ester bonds in substrates, such as triglycerides, phospholipids, cholesteryl esters, and vitamin esters [ 268 ]. Lipases show specificity in terms of fatty acids, nature of the alcohol, and stereospecificity. Digestive lipases have been isolated and characterized from several fish species, including Atlantic cod ( G. morhua ) pyloric ceca/pancreas, Atlantic salmon ( S. salar ) pancreas, red sea bream ( P. major ) hepatopancreas, rainbow trout ( O. mykiss ) inter-cecal pancreatic tissue and pyloric ceca, oil sardine ( Sardinella longiceps ) hepatopancreas, spiny dogfish ( Squalus acanthius ) pancreas, Nile tilapia ( O. niloticus ) stomach, intestine, grey mullet (Mugil cephalus) viscera [ 10 ] and reference within.
Considering the high perishability of the waste, it must be of good quality and in relatively high quantities for commercial extraction of enzymes. Some of the valid processes for the isolation of enzymes from waste are: precipitation by salts and polyacrylic acids, isoelectric solubilization/precipitation, ultrafiltration, pH shift, flocculation and membrane filtration, and overcooled acetone extraction [ 43 , 258 ] and reference within.
Fish Enzyme Applications
Today enzymatic methods play a key role in the processes used by modern industries to produce a wide range of products for human consumption. Proteases are one of the most interesting groups of industrial enzymes, widely used for several applications, especially in the food industry and as components of laundry detergents [ 269 , 270 ]; approximately 60% of the total enzyme market involves the use of proteases [ 271 ]. Marine organisms, including fish, are an excellent source of enzymes and could contribute to the total number of enzymes available on the world market.
The potential advantages of using enzymes include the development of industrial processes as alternatives to mechanical or chemical treatments which often cause damage to the product and reduce its recovery, ensuring better process control, and low energy requirements and costs. Furthermore, enzymes from fish, being extracted from edible animals, have the added advantage of being safe, so that toxicological tests on raw materials are not necessary [ 43 , 258 ].
Fish proteinases generally have high activity in a large range of pH and temperature values, making them suitable for several industrial applications, such as in the detergent, food, pharmaceutical, and agrochemical industries [ 272 ]. In particular, several of these enzymes have been isolated from cold-water fish, then inactivated at relatively low temperatures, which is potentially useful in food applications [ 273 ] where it is desirable to inactivate the enzyme with a mild heat treatment. For example, trypsin and alkaline phosphatase purified from cold-water fish shown the temperature optimums about 30 °C lower than the homologues from warm-water fish or mammals [ 274 , 275 ].
Cold-active fish pepsins from Atlantic cod ( G. morhua ) and orange roughly ( Hoplostetus atlanticus ) are used for caviar production from the roe of different species. For example, in salmon ( S. salar ), the use of these enzymes facilitates the riddling process, increasing the yield from 70% to 90% [ 27 ]. Moreover, it has been shown that cod pepsin could be used in the descaling of fish such as hake and haddock; after a treatment of fish with pepsin in weak acid conditions, the scales could be easily removed with a quick passage through a water jet system [ 276 ]. Crude pepsin from Atlantic cod is often used for industrial descaling [ 277 ]. A commercial product that includes cold tolerant protease from North Atlantic cod is designated as Penzim [ 44 ]. In addition, a cod Uracil-DNA Glycosylase (Cod UNG) from Atlantic cod is marketed by ArcticZymes ( http://arcticzymes.com , 5 December 2020); it is a heat-labile enzyme, completely and irreversibly inactivated by moderate heat treatment (55 °C). This enzyme catalyses the hydrolysis of the N-glycosylic bond between uracil and sugar, releasing an apyrimidinic site in uracil-containing single-stranded or double-stranded DNA, therefore it is a good candidate for several molecular biology applications.
Fish gastric proteases are salt-activated as these animals absorb salt-water during feeding. This represents an advantageous feature in several applications, such as fermentations, silage and fish sauce, where significant amounts of salt are used; on the contrary, homologues from mammals are generally inhibited by NaCl [ 278 ]. Among the enzymes, pepsin plays an important role in the preparation of silage [ 277 ]. For example, pepsins isolated from cod viscera were effectively used for the aqueous phase production of cod viscera silage under acidic conditions [ 279 ]. Fish proteases could be also used for skin removal from fish, as an alternative method to mechanical or chemical treatments, which often involve damage or reduction of the recovered product [ 280 ].
It has been shown that hydrolysis of proteins used in food industries could increase yields in recovery processes, improve functional properties or improve process methodology. In general, under the conditions that characterize enzyme-catalysed processes, the nutritional value of a protein is preserved better than in traditional acid or alkaline hydrolysis [ 281 , 282 ]. Moreover, autolysis via digestive enzymes of the fish itself is a simple and economical industrial method for preparing fish protein concentrate [ 283 , 284 ], with no enzyme costs involved for the process [ 258 ]. Fish sauce prepared by autolysis of fish is a popular condiment due to its distinctive flavor and taste. Enzymatic hydrolysis of dietary proteins offers a rapid and reproducible method for the production of large number of fish bioactive peptides. Compared to microbial proteases that are generally applied, endogenous fish proteases have relatively narrow and unique specificities [ 283 , 285 ], therefore have been also used for the preparation of fish hydrolyzates with several bioactivities (see Section 3.2 ). In particular, seafood proteases from Atlantic salmon and trypsin from fish pyloric ceca have been used [ 286 , 287 ]. Moreover, intestinal protease extracted from bluefin tuna ( Thunnus thynnus ) was used to hydrolyze hoki ( Johnius belengerii ) bones [ 174 ]. In addition, marine collagen was extracted from the fins, scales, skins, bones, head, and swim bladders of bighead carp by using collagenases, pepsin from tuna or trypsin from cod or tuna pyloric caeca [ 288 , 289 , 290 , 291 , 292 , 293 ]
Lipases isolated from Atlantic cod ( Gadus morhua ) guts have been reported to mainly hydrolyze PUFAs compared to shorter chain fatty acids [ 294 ]. Thanks to their high affinity for long-chain fatty acids, specificity for particular fatty acids, and regiospecificity [ 295 ], fish lipolytic enzymes can be used in the synthesis of structured lipids. Lipase isolated from sea bass liver was used to defat fish skin, leading to the removal of 84.57% fat from the skin [ 296 ].
Digestive lipases from fish could also be used for lipid interesterification reactions, such as the transesterification, with minimal or no byproducts, which generally represent a problem in chemical transformations [ 297 ]. Some fish digestive lipases are both stereospecific and enantiospecific, and for this reason they could be used in the synthesis of enantiomerically pure products that can be used as specialty pharmaceuticals or agrochemicals. Furthermore, the use of these lipases could be useful for specialty esters for personal care products and environmentally safe surfactants for applications in detergents or as food emulsifiers [ 297 ].
Table 5 lists the most interesting fish enzymes because marketed/used for several applications.
Enzymes from fish waste and their applications. PSC, pepsin-solubilised collagen.
| Compound | Byproduct | Source | Applications | Reference |
|---|---|---|---|---|
| Pepsin | Stomach | Atlantic cod ( ) | Caviar productionFish descaling | [ ] |
| Pepsin | Viscera | cod | Silage production | [ ] |
| Pepsin | Stomach | Orange roughy ( ) | Caviar production | [ ] |
| Proteases | Pyloric ceca | Atlantic Salmon ( ) | Fish hydrolyzates production | [ ] |
| Trypsin | Pyloric ceca | Unicorn leatherjacket ( ) | Fish hydrolyzates production | [ ] |
| Proteases | Intestine | Bluefin tuna ( ) | Fish hydrolyzates production | [ ] |
| Proteases | Stomach | Albacore tuna ( ) | PSC extraction | [ ] |
| Proteases | Stomach | Yellowfin tuna ( ) | PSC extraction | [ ] |
| Uracil-DNA Glycosylase | Liver | Atlantic cod ( ) | Molecular biology | [ ] |
| Lipases | Intestine/Pyloric ceca | Atlantic cod ( ) | Potential lipids synthesis | [ ] |
| Lipases | Liver | Sea bass ( ) | Defatting of fish skin | [ ] |
4. Conclusions
Fish waste is not only a major environmental problem, but also a huge economic loss. For this reason, a better fish-waste management is needed to overcome these important issues. Therefore, today, the development of a sustainable fish waste management plays a key role, as it is closely related to the Waste Framework Directive, which aims to prevent the generation of waste as much as possible, and to use the waste generated as a resource for reuse, recycling, and recovery. In this way, the use of fish byproducts could contribute to the development of products with high-commercial value, and consequently, to economic growth.
The review comprehensively illustrates the compounds that can be obtained from fish discards and byproducts, highlighting how this waste could become an enormous resource for the production of value-added products (e.g., peptides, proteins, collagen, chitin, oil, and enzymes), with several potential applications. The extraction and purification techniques are mainly based on procedures as acid extraction, enzymatic hydrolysis, and fermentation. The attention in choosing the best extraction technique is generally focused on the final yield and, especially in the case of compounds possibly intended for human consumption, on the preservation of the nutritional value as much as possible. Interestingly, new and more sustainable approaches are in progress, i.e., SFE, which we evidenced as emerging technology for fish oil recovery or the use of green and sustainable deep eutectic solvents. Main extraction methods and further processing technologies reported in the text are summarized in Figure 1 .

Summary of the main extraction methods and further processing technologies used to obtain high-commercial value compounds from fish byproducts.
The range of applications for all compounds deriving from fish discards treatment is very wide, by covering the medical, pharmaceutical, and packaging, food and biofuel production fields.
In consideration of the scale of demand for seafood worldwide, the use of fish discards as a source of high-value compounds is an excellent cost recovery strategy, especially in the historical period we are experiencing. Indeed, this review provided also the opportunity to highlight how the global market forecasts for some of these products (e.g., an increase of global fish oil market size from $ 1905.77 million in 2019 to $ 2844.12 million by 2027) and how these forecasts have been changed following the Covid-19 outbreak. For instance, the chitosan global market before the Covid-19 pandemic occurrence was expected to increase at a CAGR of 24.7% from 2020 to 2027, while this percentage is now reduced to 16.9%. Among several social and economic fields, the pandemic has affected also the current food systems, based on a linear and globalized production and consumption model. This highlights the need of a new economic model focused on the social wellbeing and environmental sustainability at the core of the EU’s economic recovery. The circular economy is now considered a pivotal component of the recovery plan [ 301 ], and in this context, herein, we present the possible concretization of this kind of economic model in the field of marine resources and fish products.
Author Contributions
D.C. and D.d.P. conceptualized the idea and provided the framework for the document. D.C., C.L., F.P.E., G.R., C.R. and D.d.P. gave an important contribution to search for the available bibliographic information and to prepare the final version of the manuscript. All authors have read and agreed to the published version of the manuscript.
The authors thank the H2020-MSCA-RISE Ocean Medicines, GA 690944 and the H2020-MSCA-ITN-ETN MarPipe, GA 721421 for the partial support. The authors also thank the ERA-Net Marine Biotechnology BlueShell and ERA-Net BlueBio Cofund BlueCC projects.
Institutional Review Board Statement
Informed consent statement, data availability statement, conflicts of interest.
The authors declare no conflict of interest.
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Academia.edu no longer supports Internet Explorer.
To browse Academia.edu and the wider internet faster and more securely, please take a few seconds to upgrade your browser .
- We're Hiring!
- Help Center
Fishing industry
- Most Cited Papers
- Most Downloaded Papers
- Newest Papers
- Analysis of Work Accidents Follow Following
- Small and Medium Sized Enterprises Follow Following
- Social Navigation Follow Following
- Cornwall Follow Following
- Sheffield Follow Following
- Inventories Follow Following
- PhD education Follow Following
- Journalistikk Follow Following
- Caribbean Ecosystems Follow Following
- Samfundsfag Follow Following
Enter the email address you signed up with and we'll email you a reset link.
- Academia.edu Journals
- We're Hiring!
- Help Center
- Find new research papers in:
- Health Sciences
- Earth Sciences
- Cognitive Science
- Mathematics
- Computer Science
- Academia ©2024
Advertisement
Intelligent Cutting in Fish Processing: Efficient, High-quality, and Safe Production of Fish Products
- Published: 08 July 2023
- Volume 17 , pages 828–849, ( 2024 )
Cite this article

- Jiaying Fu ORCID: orcid.org/0000-0002-8086-3820 1 ,
- Yingchao He ORCID: orcid.org/0000-0002-3429-3126 1 &
- Fang Cheng ORCID: orcid.org/0000-0001-7293-9082 1 , 2
801 Accesses
2 Citations
Explore all metrics
Fish processing is an indispensable part of fish food production. It mainly involves de-heading, gutting, filleting, skinning, trimming, and slicing, with the cutting operations holding a critical role. Unfortunately, inefficiency, low quality, and poor safety are the primary problems facing the fish processing industry today, dramatically hindering the automation and intelligence of fish processing. Consequently, it is vital to develop intelligent cutting in current fish processing in an efficient, high-quality, and safe manner. This review summarizes the main cutting techniques for fish processing. The critical techniques to achieve intelligent cutting in fish processing from imaging, image processing, and modeling dimensions are outlined, with their applications in practical fish processing. Fish characteristics, cutting mechanisms, and cutting process control are emphasized. In addition, Industry 4.0 technologies, especially the Internet of Things (IoT), big data analytics, and digital twins (DT), are emphasized. Finally, challenges and future work are highlighted, which will serve as references for subsequent researchers and enterprises engaged in this field to promote the automation and intelligence of fish processing production, ultimately realizing the high-efficiency, high-quality, and safe production of fish food products.
This is a preview of subscription content, log in via an institution to check access.
Access this article
Subscribe and save.
- Get 10 units per month
- Download Article/Chapter or Ebook
- 1 Unit = 1 Article or 1 Chapter
- Cancel anytime
Price includes VAT (Russian Federation)
Instant access to the full article PDF.
Rent this article via DeepDyve
Institutional subscriptions

Similar content being viewed by others

Intelligent fish farm—the future of aquaculture
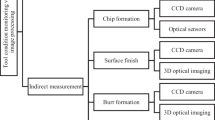
State-of-the-art review of applications of image processing techniques for tool condition monitoring on conventional machining processes
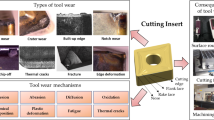
Artificial intelligence based tool condition monitoring for digital twins and industry 4.0 applications
Abbreviations.
Two-dimensional
Three-dimensional
Automated Dynamic Analysis of Mechanical Systems
- Artificial intelligence
Artificial neural networks
Blade sharpness index
Convolutional neural networks
Computer tomography
Deep learning
Digital twins
Food and agriculture organization of the united nations
Finite element
Fuzzy-rough set model based on the thermal-charge algorithm
Gaussian maximum likelihood
Gray value analysis
Hyperspectral imaging
Internet of Things
Linear discriminant analysis
Machine learning
Magnetic resonance imaging
Machine vision systems
Not applicable
Principal component analysis
Region based convolutional neural network
Sequential forward selection
Support vector data description
Support vector machine
Traditional image processing
Ultraviolet
Visual geometry group
Visible and near-infrared
Water jet cutting
Abdollahi, M., Wu, H., & Undeland, I. (2021). Impact of processing technology on macro- and micronutrient profile of protein-enriched products from fish backbones. Foods. https://doi.org/10.3390/foods10050950
Article PubMed PubMed Central Google Scholar
Ageev, O. V., Dowgiałło, A., Sterczyńska, M., Piepiórka-Stepuk, J., Giurgiulescu, L., Janowicz, M., & Jakubowski, M. (2021). Experimental characterization and theoretical modeling of fracture and friction resistance forces during tuna cutting. Journal of Food Engineering . https://doi.org/10.1016/j.jfoodeng.2021.110648
Article Google Scholar
Alom, M. Z., Taha, T. M., Yakopcic, C., Westberg, S., Sidike, P., Nasrin, M. S., Hasan, M., Van Essen, B. C., Awwal, A. A. S., & Asari, V. K. (2019). A state-of-the-art survey on deep learning theory and architectures. Electronics, 8 (3), 292. https://doi.org/10.3390/electronics8030292
Anderssen, K. E., Stormo, S. K., Skara, T., Skjelvareid, M. H., & Heia, K. (2020). Predicting liquid loss of frozen and thawed cod from hyperspectral imaging. LWT-Food Science and Technology . https://doi.org/10.1016/j.lwt.2020.110093
Arthington, A. H., Dulvy, N. K., Gladstone, W., & Winfield, I. J. (2016). Fish conservation in freshwater and marine realms: Status, threats and management. Aquatic Conservation-Marine and Freshwater Ecosystems, 26 (5), 838–857. https://doi.org/10.1002/aqc.2712
Azarmdel, H., Mohtasebi, S. S., Jafari, A., & Rosado Muñoz, A. (2019). Developing an orientation and cutting point determination algorithm for a trout fish processing system using machine vision. Computers and Electronics in Agriculture, 162 , 613–629. https://doi.org/10.1016/j.compag.2019.05.005
Azarmdel, H., Mohtasebi, S. S., Jafary, A., Behfar, H., & Rosado Muñoz, A. (2021). Design and simulation of a vision-based automatic trout fish-processing robot. Applied Sciences-Basel . https://doi.org/10.3390/app11125602
Balaban, M. O., Misimi, E., & Ayvaz, Z. (2016). Quality evaluation of seafoods. In D.-W. Sun (Ed.), Computer Vision Technology for Food Quality Evaluation (2nd ed., pp. 243–270). Academic Press, San Diego. https://doi.org/10.1016/B978-0-12-802232-0.00010-4
Balaban, M. O., Sengor, G. F. U., Gil Soriano, M., & Guillen Ruiz, E. (2010). Using image analysis to predict the weight of Alaskan salmon of different species. Journal of Food Science, 75 (3), E157–E162. https://doi.org/10.1111/j.1750-3841.2010.01522.x
Article CAS PubMed Google Scholar
Balaban, M. O., Sengor, G. F. U., Soriano, M. G., & Ruiz, E. G. (2011). Quantification of gaping, bruising, and blood spots in Salmon fillets using image analysis. Journal of Food Science, 76 (3), E291–E297. https://doi.org/10.1111/j.1750-3841.2011.02060.x
Banerjee, A., Das, A., Behra, S., Bhattacharjee, D., Srinivasan, N. T., Nasipuri, M., & Das, N. (2022). Carp-DCAE: Deep convolutional autoencoder for carp fish classification. Computers and Electronics in Agriculture . https://doi.org/10.1016/j.compag.2022.106810
Bar, E., Mathiassen, J. R., Eilertsen, A., Mugaas, T., Misimi, E., Linnerud, Å. S., Salomonsen, C., & Westavik, H. (2016). Towards robotic post-trimming of salmon fillets. Industrial Robot-the International Journal of Robotics Research and Application, 43 (4), 421–428. https://doi.org/10.1108/ir-11-2015-0205
Bland, J. M., Grimm, C. C., Bechtel, P. J., Deb, U., & Dey, M. M. (2021). Proximate composition and nutritional attributes of ready-to-cook catfish products. Foods. https://doi.org/10.3390/foods10112716
Bogue, R. (2008). Cutting robots: A review of technologies and applications. Industrial Robot-the International Journal of Robotics Research and Application, 35 (5), 390–396. https://doi.org/10.1108/01439910810893554
Bonder, M. S., Mathiassen, J. R., Vebenstad, P. A., Misimi, E., Bar, E. M. S., Toldnes, B., & Ostvik, S. O. (2011). An automated salmonid slaughter line using machine vision. Industrial Robot-the International Journal of Robotics Research and Application, 38 (4), 399–405. https://doi.org/10.1108/01439911111132085
Borderias, A. J., & Sanchez-Alonso, I. (2011). First processing steps and the quality of wild and farmed fish. Journal of Food Science, 76 (1), R1–R5. https://doi.org/10.1111/j.1750-3841.2010.01900.x
Article CAS PubMed PubMed Central Google Scholar
Bremer, F., & Matthiesen, S. (2020). A review of research relating to food slicing in industrial applications. Journal of Food Engineering . https://doi.org/10.1016/j.jfoodeng.2019.109735
Bremer, F., & Matthiesen, S. (2021). High-speed cutting with involute blades: Experimental research on cutting forces. Journal of Food Engineering . https://doi.org/10.1016/j.jfoodeng.2020.110380
Buljo, J. O., & Gjerstad, T. B. (2013). Robotics and automation in seafood processing. In D. G. Caldwell (Ed.), Robotics and Automation in the Food Industry (1st ed., pp. 354–384). Woodhead Publishing, Oxford. https://doi.org/10.1533/9780857095763.2.354
Cao, W., Liu, Q., & He, Z. (2020). Review of pavement defect detection methods. IEEE Access, 8 , 14531–14544. https://doi.org/10.1109/ACCESS.2020.2966881
Chaki, J., & Dey, N. (2018). A beginner’s guide to image preprocessing techniques . CRC Press.
Book Google Scholar
Chan, S. S., Roth, B., Skare, M., Hernar, M., Jessen, F., Løvdal, T., Jakobsen, A. N., & Lerfall, J. (2020). Effect of chilling technologies on water holding properties and other quality parameters throughout the whole value chain: From whole fish to cold-smoked fillets of Atlantic salmon (Salmo salar). Aquaculture . https://doi.org/10.1016/j.aquaculture.2020.735381
Chen, L., & Opara, U. L. (2013). Approaches to analysis and modeling texture in fresh and processed foods – A review. Journal of Food Engineering, 119 (3), 497–507. https://doi.org/10.1016/j.jfoodeng.2013.06.028
Cheng, J.-H., Nicolai, B., & Sun, D.-W. (2017). Hyperspectral imaging with multivariate analysis for technological parameters prediction and classification of muscle foods: A review. Meat Science, 123 , 182–191. https://doi.org/10.1016/j.meatsci.2016.09.017
Article PubMed Google Scholar
Cui, D., Li, H., He, J., Wang, Q., Lu, C., Hu, H., Cheng, X., & Wang, C. (2022). Applications of water jet cutting technology in agricultural engineering: A review. Applied Sciences-Basel . https://doi.org/10.3390/app12188988
Diamond, K. M., Avants, B. B., & Maga, A. M. (2021). Machine learning-based segmentation and landmarking of 2D fish images. Integrative and Comparative Biology, 61 , E1100–E1101.
Google Scholar
Dowgiallo, A. (2008). The effect of cutting and fish-orientation systems on the deheading yield of carp. International Journal of Food Science and Technology, 43 (9), 1688–1692. https://doi.org/10.1111/j.1365-2621.2008.01750.x
Article CAS Google Scholar
Dowgiallo, A., & Dutkiewicz, D. (2007). Possibilities of utilizing the differences of fish tissues stiffness in the mechanization of cyprinid deheading. Journal of Food Engineering, 83 (1), 111–115. https://doi.org/10.1016/j.jfoodeng.2007.01.028
Dowlati, M., Mohtasebi, S. S., & de la Guardia, M. (2012). Application of machine-vision techniques to fish-quality assessment. TrAC Trends in Analytical Chemistry, 40 , 168–179. https://doi.org/10.1016/j.trac.2012.07.011
Duong, L. N. K., Al-Fadhli, M., Jagtap, S., Bader, F., Martindale, W., Swainson, M., & Paoli, A. (2020). A review of robotics and autonomous systems in the food industry: From the supply chains perspective. Trends in Food Science & Technology, 106 , 355–364. https://doi.org/10.1016/j.tifs.2020.10.028
Einarsdottir, H., Guethmundsson, B., & Omarsson, V. (2022). Automation in the fish industry. Animal Frontiers, 12 (2), 32–39. https://doi.org/10.1093/af/vfac020
Erikson, U., Misimi, E., & Fismen, B. (2010). Bleeding of anaesthetized and exhausted Atlantic salmon: Body cavity inspection and residual blood in pre-rigor and smoked fillets as determined by various analytical methods. Aquaculture Research, 41 (4), 496–510. https://doi.org/10.1111/j.1365-2109.2009.02338.x
FAO. (2022). The state of world fisheries and aquaculture 2022. Towards Blue Transformation, Rome. https://doi.org/10.4060/cc0461en
Gupta, K. (2020). A review on green machining techniques. Procedia Manufacturing, 51 , 1730–1736. https://doi.org/10.1016/j.promfg.2020.10.241
Hao, Y., Yin, H., & Li, D. (2022). A novel method of fish tail fin removal for mass estimation using computer vision. Computers and Electronics in Agriculture . https://doi.org/10.1016/j.compag.2021.106601
Hassoun, A., Aït-Kaddour, A., Abu-Mahfouz, A. M., Rathod, N. B., Bader, F., Barba, F. J., Biancolillo, A., Cropotova, J., Galanakis, C. M., Jambrak, A. R., Lorenzo, J. M., Måge, I., Ozogul, F., & Regenstein, J. (2022a). The fourth industrial revolution in the food industry—Part I: Industry 4.0 technologies. Critical Reviews in Food Science and Nutrition . https://doi.org/10.1080/10408398.2022.2034735
Hassoun, A., Cropotova, J., Trollman, H., Jagtap, S., Garcia-Garcia, G., Parra-López, C., Nirmal, N., Özogul, F., Bhat, Z., Aït-Kaddour, A., & Bono, G. (2023). Use of industry 4.0 technologies to reduce and valorize seafood waste and by-products: A narrative review on current knowledge. Current Research in Food Science, 6 , 100505. https://doi.org/10.1016/j.crfs.2023.100505
Hassoun, A., Siddiqui, S. A., Smaoui, S., Ucak, İ, Arshad, R. N., Garcia-Oliveira, P., Prieto, M. A., Aït-Kaddour, A., Perestrelo, R., Câmara, J. S., & Bono, G. (2022b). Seafood processing, preservation, and analytical techniques in the age of Industry 4.0. Applied Sciences . https://doi.org/10.3390/app12031703
Hlavacova, I. M., & Geryk, V. (2017). Abrasives for water-jet cutting of high-strength and thick hard materials. International Journal of Advanced Manufacturing Technology, 90 (5–8), 1217–1224. https://doi.org/10.1007/s00170-016-9462-y
Jayraj, P., Machavaram, R., Sahu, G., & Paradkar, V. (2019). Measurement of morphometric dimensions and mechanical properties of Rohu fish for design of processing machines. Journal of Aquatic Food Product Technology, 28 (2), 150–164. https://doi.org/10.1080/10498850.2019.1569741
Jeebhay, M. F., Robins, T. G., Miller, M. E., Bateman, E., Smuts, M., Baatjies, R., & Lopata, A. L. (2008). Occupational allergy and asthma among salt water fish processing workers. American Journal of Industrial Medicine, 51 (12), 899–910. https://doi.org/10.1002/ajim.20635
Kapłonek, W., Nadolny, K., Zieliński, B., Plichta, J., Pimenov, D. Y., & Sharma, S. (2020). The role of observation–Measurement methods in the surface characterization of X39Cr13 stainless-steel cutting blades used in the fish processing industry. Materials . https://doi.org/10.3390/ma13245796
Kasperowicz, M. B., Chomka, G. P., & Bil, T. (2019). Determination of supply pressure during cutting fish using high-pressure water stream taking into account the cutting place and diameter of the water nozzle. International Journal of Food Engineering . https://doi.org/10.1515/ijfe-2018-0395
Kelkar, S., Boushey, C. J., & Okos, M. (2015). A method to determine the density of foods using X-ray imaging. Journal of Food Engineering, 159 , 36–41. https://doi.org/10.1016/j.jfoodeng.2015.03.012
Khodabandehloo, K. (2022). Achieving robotic meat cutting. Animal . Frontiers, 12 (2), 7–17. https://doi.org/10.1093/af/vfac012
Kirtil, E., & Oztop, M. H. (2016). 1H Nuclear magnetic resonance relaxometry and magnetic resonance imaging and applications in food science and processing. Food Engineering Reviews, 8 (1), 1–22. https://doi.org/10.1007/s12393-015-9118-y
Kogiannou, D., Kotsiri, M., & Grigorakis, K. (2022). A method to assess gaping in Sparidae species fillets. Aquaculture Research, 53 (2), 689–693. https://doi.org/10.1111/are.15590
Koulouris, A., Misailidis, N., & Petrides, D. (2021). Applications of process and digital twin models for production simulation and scheduling in the manufacturing of food ingredients and products. Food and Bioproducts Processing, 126 , 317–333. https://doi.org/10.1016/j.fbp.2021.01.016
Larouche, O., Cloutier, R., & Zelditch, M. L. (2015). Head, body and fins: Patterns of morphological integration and modularity in fishes. Evolutionary Biology, 42 (3), 296–311. https://doi.org/10.1007/s11692-015-9324-9
Li, D., Miao, Z., Peng, F., Wang, L., Hao, Y., Wang, Z., Chen, T., Li, H., & Zheng, Y. (2021). Automatic counting methods in aquaculture: A review. Journal of the World Aquaculture Society, 52 (2), 269–283. https://doi.org/10.1111/jwas.12745
Li, D., Wang, Q., Li, X., Niu, M., Wang, H., & Liu, C. (2022). Recent advances of machine vision technology in fish classification. ICES Journal of Marine Science, 79 (2), 263–284. https://doi.org/10.1093/icesjms/fsab264
Lima, D. A. S., Santos, M. M. F., Duvale, R. L. F., Bezerra, T. K. A., Araujo, I. B. D., Madruga, M. S., & da Silva, F. A. P. (2021). Technological properties of protein hydrolysate from the cutting byproduct of serra Spanish mackerel (Scomberomorus brasiliensis). Journal of Food Science and Technology-Mysore, 58 (8), 2952–2962. https://doi.org/10.1007/s13197-020-04797-5
Lima, D. A. S., Santos, M. M. F., Sousa, A. M. B. L., Bezerra, T. K. A., da Silva Araújo, Í. B., Madruga, M. S., & da Silva, F. A. P. (2022). The cutting by-product of fish filleting on the band saw machine: Nutritional quality and technological potential. Waste Biomass Valorization, 13 (11), 4575–4584. https://doi.org/10.1007/s12649-022-01818-6
Liu, D., Zeng, X.-A., & Sun, D.-W. (2013). NIR spectroscopy and imaging techniques for evaluation of fish quality—A review. Applied Spectroscopy Reviews, 48 (8), 609–628. https://doi.org/10.1080/05704928.2013.775579
Liu, S., Wang, H., & Cai, Y. (2021). Research on fish slicing method based on simulated annealing algorithm. Applied Sciences-Basel . https://doi.org/10.3390/app11146503
Liu, W., Lyu, J., Wu, D., Cao, Y., Ma, Q., Lu, Y., & Zhang, X. (2022). Cutting techniques in the fish industry: A critical review. Foods. https://doi.org/10.3390/foods11203206
Liu, X. C., Liang, Z. W., Wen, G. L., & Yuan, X. F. (2019). Waterjet machining and research developments: A review. International Journal of Advanced Manufacturing Technology, 102 (5–8), 1257–1335. https://doi.org/10.1007/s00170-018-3094-3
Lopez, M., Maia, P., Figueiredo, R., Alves, N., & Gonzalez, D. (2019). Automated vision system for cutting fixed-weight or fixed-length frozen fish portions. In Proceedings of the 8th International Conference on Pattern Recognition Applications and Methods , Prague, Czech Republic. https://doi.org/10.5220/0007482407070714
Mason, A., Romanov, D., Cordova-Lopez, L. E., Ross, S., & Korostynska, O. (2022). Smart knife: Technological advances towards smart cutting tools in meat industry automation. Sensor Review, 42 (1), 155–163. https://doi.org/10.1108/sr-09-2021-0315
McGeough, J. A. (2016). Cutting of food products by ice-particles in a water-jet. In 18th CIRP Conference on Electro Physical and Chemical Machining (ISEM XVIII) , Tokyo, Japan. https://doi.org/10.1016/j.procir.2016.03.009
Merkin, G. V., Stien, L. H., Pittman, K., & Nortvedt, R. (2013). Digital image analysis as a tool to quantify gaping and morphology in smoked salmon slices. Aquacultural Engineering, 54 , 64–71. https://doi.org/10.1016/j.aquaeng.2012.11.003
Mery, D., Lillo, I., Loebel, H., Riffo, V., Soto, A., Cipriano, A., & Aguilera, J. M. (2011). Automated fish bone detection using X-ray imaging. Journal of Food Engineering, 105 (3), 485–492. https://doi.org/10.1016/j.jfoodeng.2011.03.007
Misimi, E., Øye, E. R., Sture, Ø., & Mathiassen, J. R. (2017). Robust classification approach for segmentation of blood defects in cod fillets based on deep convolutional neural networks and support vector machines and calculation of gripper vectors for robotic processing. Computers and Electronics in Agriculture, 139 , 138–152. https://doi.org/10.1016/j.compag.2017.05.021
Murthy, L. N., Phadke, G. G., Siddaiah, V., & Boraiah, R. K. (2017). Rheological properties of washed and unwashed tilapia (Oreochromis mossambicus) fish meat: Effect of sucrose and sorbitol. Food Science and Biotechnology, 26 (5), 1177–1183. https://doi.org/10.1007/s10068-017-0162-7
Nakashima, Y. (2020). Development of a hand-held magnetic resonance sensor for the nondestructive quantification of fat and lean meat of fresh tuna. Journal of Food Measurement and Characterization, 14 (6), 2947–2955. https://doi.org/10.1007/s11694-020-00539-5
Olsen, S. H., Sorensen, N. K., Larsen, R., Elvevoll, E. O., & Nilsen, H. (2008). Impact of pre-slaughter stress on residual blood in fillet portions of farmed Atlantic cod (Gadus morhua) - Measured chemically and by Visible and Near-infrared spectroscopy. Aquaculture, 284 (1–4), 90–97. https://doi.org/10.1016/j.aquaculture.2008.07.042
Ooi, C. K., Lewis, T., Nowak, B., Lyle, J., & Haddy, J. (2022). The use of image analysis techniques for the study of muscle melanisation in sand flathead (Platycephalus bassensis)*. Environmental Pollution . https://doi.org/10.1016/j.envpol.2021.118360
Ørnholt-Johansson, G., Gudjónsdóttir, M., Nielsen, M. E., Skytte, J. L., & Frosch, S. (2017). Analysis of the production of salmon fillet – Prediction of production yield. Journal of Food Engineering, 204 , 80–87. https://doi.org/10.1016/j.jfoodeng.2017.02.022
Prasetyo, E., Suciati, N., & Fatichah, C. (2022). Yolov4-tiny with wing convolution layer for detecting fish body part. Computers and Electronics in Agriculture . https://doi.org/10.1016/j.compag.2022.107023
Ren, Q.-S., Fang, K., Yang, X.-T., & Han, J.-W. (2022). Ensuring the quality of meat in cold chain logistics: A comprehensive review. Trends in Food Science & Technology, 119 , 133–151. https://doi.org/10.1016/j.tifs.2021.12.006
Riorlykhaug, E., & Egeland, O. (2019). Vision system for quality assessment of robotic cleaning of fish processing plants using CNN. IEEE Access, 7 , 71675–71685. https://doi.org/10.1109/ACCESS.2019.2919656
Romanov, D., Korostynska, O., Lekang, O. I., & Mason, A. (2022). Towards human-robot collaboration in meat processing: Challenges and possibilities. Journal of Food Engineering . https://doi.org/10.1016/j.jfoodeng.2022.111117
Ross, K., & Edwards, J. (2015). Spatial variation in the mercury concentration of muscle myomeres in steaks of farmed Southern Bluefin Tuna. Foods, 4 (2), 254–262. https://doi.org/10.3390/foods4020254
Rustad, T., Storro, I., & Slizyte, R. (2011). Possibilities for the utilisation of marine by-products. International Journal of Food Science and Technology, 46 (10), 2001–2014. https://doi.org/10.1111/j.1365-2621.2011.02736.x
Saberioon, M., Gholizadeh, A., Cisar, P., Pautsina, A., & Urban, J. (2017). Application of machine vision systems in aquaculture with emphasis on fish: State-of-the-art and key issues. Reviews in Aquaculture, 9 (4), 369–387. https://doi.org/10.1111/raq.12143
Saha, D., & Manickavasagan, A. (2021). Machine learning techniques for analysis of hyperspectral images to determine quality of food products: A review. Current Research in Food Science, 4 , 28–44. https://doi.org/10.1016/j.crfs.2021.01.002
Sarah, S., Anne, S., John, G., Keith, S., Philip, C., Stefan, T., & Jonathan, D. W. (2019). Evaluation of Salmon (Salmo salar) and Rainbow Trout (Oncorhynchus mykiss) pin bones using textural analysis and micro X-ray computational tomography. Journal of Food Science and Technology-Mysore, 56 (7), 3313–3319. https://doi.org/10.1007/s13197-019-03803-9
Schneider, Y., Zahn, S., & Rohm, H. (2011). Ultrasonic cutting of foods. In H. Feng, G. Barbosa-Canovas, & J. Weiss (Eds.), Ultrasound Technologies for Food and Bioprocessing . pp. 211–237). Springer New York, New York, NY. https://doi.org/10.1007/978-1-4419-7472-3_9
Schuldt, S., Schneider, Y., & Rohm, H. (2018). High-speed cutting of foods: Cutting behavior and initial cutting forces. Journal of Food Engineering, 230 , 55–62. https://doi.org/10.1016/j.jfoodeng.2018.02.024
Secci, G., & Parisi, G. (2016). From farm to fork: Lipid oxidation in fish products. A review. Italian Journal of Animal Science, 15 (1), 124–136. https://doi.org/10.1080/1828051X.2015.1128687
Siche, R., Vejarano, R., Aredo, V., Velasquez, L., Saldaña, E., & Quevedo, R. (2016). Evaluation of food quality and safety with hyperspectral imaging (HSI). Food Engineering Reviews, 8 (3), 306–322. https://doi.org/10.1007/s12393-015-9137-8
Simat, V., Miletic, J., Bogdanovic, T., Poljak, V., & Mladineo, I. (2015). Role of biogenic amines in the post-mortem migration of Anisakis pegreffii (Nematoda: Anisakidae Dujardin, 1845) larvae into fish fillets. International Journal of Food Microbiology, 214 , 179–186. https://doi.org/10.1016/j.ijfoodmicro.2015.08.008
Sivertsen, A. H., Chu, C.-K., Wang, L.-C., Godtliebsen, F., Heia, K., & Nilsen, H. (2009). Ridge detection with application to automatic fish fillet inspection. Journal of Food Engineering, 90 (3), 317–324. https://doi.org/10.1016/j.jfoodeng.2008.06.035
Sivertsen, A. H., Heia, K., Hindberg, K., & Godtliebsen, F. (2012). Automatic nematode detection in cod fillets ( Gadus morhua L.) by hyperspectral imaging. Journal of Food Engineering, 111 (4), 675–681. https://doi.org/10.1016/j.jfoodeng.2012.02.036
Sivertsen, A. H., Heia, K., Stormo, S. K., Elvevoll, E., & Nilsen, H. (2011). Automatic nematode detection in cod fillets (Gadus Morhua) by transillumination hyperspectral imaging. Journal of Food Science, 76 (1), S77–S83. https://doi.org/10.1111/j.1750-3841.2010.01928.x
Skjelvareid, M. H., Heia, K., Olsen, S. H., & Stormo, S. K. (2017). Detection of blood in fish muscle by constrained spectral unmixing of hyperspectral images. Journal of Food Engineering, 212 , 252–261. https://doi.org/10.1016/j.jfoodeng.2017.05.029
Soltani Firouz, M., & Sardari, H. (2022). Defect detection in fruit and vegetables by using machine vision systems and image processing. Food Engineering Reviews, 14 (3), 353–379. https://doi.org/10.1007/s12393-022-09307-1
Song, S., Liu, Z., Huang, M., Zhu, Q., Qin, J., & Kim, M. S. (2020). Detection of fish bones in fillets by Raman hyperspectral imaging technology. Journal of Food Engineering . https://doi.org/10.1016/j.jfoodeng.2019.109808
Spagnoli, A., Brighenti, R., Terzano, M., & Artoni, F. (2019). Cutting resistance of soft materials: Effects of blade inclination and friction. Theoretical and Applied Fracture Mechanics, 101 , 200–206. https://doi.org/10.1016/j.tafmec.2019.02.017
Sung, H. J., Park, M. K., & Choi, J. W. (2020). Automatic grader for flatfishes using machine vision. International Journal of Control Automation and Systems, 18 (12), 3073–3082. https://doi.org/10.1007/s12555-020-0007-7
Urazoe, K., Kuroki, N., Maenaka, A., Tsutsumi, H., Iwabuchi, M., Fuchuya, K., Hirose, T., & Numa, M. (2021). Automated fish bone detection in X-ray images with convolutional neural network and synthetic image generation. IEEJ Transactions on Electrical and Electronic Engineering, 16 (11), 1510–1517. https://doi.org/10.1002/tee.23448
Uyar, R., & Erdogdu, F. (2009). Potential use of 3-dimensional scanners for food process modeling. Journal of Food Engineering, 93 (3), 337–343. https://doi.org/10.1016/j.jfoodeng.2009.01.034
Veliyulin, E., Misimi, E., Bondo, M., Vebenstad, P. A., & Ostvik, S. O. (2011). A simple method for weight estimation of whole herring (Clupea harengus) using planar X-ray imaging. Journal of Food Science, 76 (3), E328–E331. https://doi.org/10.1111/j.1750-3841.2011.02093.x
Verboven, P., Defraeye, T., Datta, A. K., & Nicolai, B. (2020). Digital twins of food process operations: The next step for food process models? Current Opinion in Food Science, 35 , 79–87. https://doi.org/10.1016/j.cofs.2020.03.002
Vidaček, S., & Bugge, E. (2016). Hygienic design of fish processing equipment. In H. Lelieveld, J. Holah, & D. Gabrić (Eds.), Handbook of hygiene control in the food industry (2nd ed., pp. 359–365). Woodhead Publishing, San Diego. https://doi.org/10.1016/B978-0-08-100155-4.00026-1
Wang, D., Zhang, M., Mujumdar, A. S., & Yu, D. (2022). Advanced detection techniques using artificial intelligence in processing of berries. Food Engineering Reviews, 14 (1), 176–199. https://doi.org/10.1007/s12393-021-09298-5
Wang, J., & Shanmugam, D. K. (2009). Cutting meat with bone using an ultrahigh pressure abrasive waterjet. Meat Science, 81 (4), 671–677. https://doi.org/10.1016/j.meatsci.2008.11.010
Wang, S., Nian, R., Cao, L., Sui, J., & Lin, H. (2015). Detection of fish bones in cod fillets by UV illumination. Journal of Food Protection, 78 (7), 1414–1419. https://doi.org/10.4315/0362-028X.JFP-14-358
Wu, D., & Sun, D.-W. (2013). Advanced applications of hyperspectral imaging technology for food quality and safety analysis and assessment: A review - Part I: Fundamentals. Innovative Food Science and Emerging Technologies, 19 , 1–14. https://doi.org/10.1016/j.ifset.2013.04.014
Wulfkuehler, S., Stark, S., Dietz, J., Schmidt, H., Weiss, A., & Carle, R. (2014). Effect of water jet cutting and moderate heat treatment on quality of fresh-cut red oak leaf lettuce ( Lactuca sativa L. var. crispa). Food and Bioprocess Technology, 7 (12), 3478–3492. https://doi.org/10.1007/s11947-014-1360-4
Xie, T., Li, X., Zhang, X., Hu, J., & Fang, Y. (2021). Detection of Atlantic salmon bone residues using machine vision technology. Food Control . https://doi.org/10.1016/j.foodcont.2020.107787
Xu, J.-L., & Sun, D.-W. (2018). Computer vision detection of salmon muscle gaping using convolutional neural network features. Food Analytical Methods, 11 (1), 34–47. https://doi.org/10.1007/s12161-017-0957-4
Article MathSciNet Google Scholar
Xu, W., He, Y., Li, J., Zhou, J., Xu, E., Wang, W., & Liu, D. (2023). Robotization and intelligent digital systems in the meat cutting industry: From the perspectives of robotic cutting, perception, and digital development. Trends in Food Science & Technology . https://doi.org/10.1016/j.tifs.2023.03.018
Xu, W., Wang, J., Deng, Y., Li, J., Yan, T., Zhao, S., Yang, X., Xu, E., Wang, W., & Liu, D. (2022). Advanced cutting techniques for solid food: Mechanisms, applications, modeling approaches, and future perspectives. Comprehensive Reviews in Food Science and Food Safety, 21 (2), 1568–1597. https://doi.org/10.1111/1541-4337.12896
Yang, X. L., Nian, R., Lin, H., Duan, C., Sui, J. X., & Cao, L. M. (2013). Detection of anisakid larvae in cod fillets by UV fluorescent imaging based on principal component analysis and gray value analysis. Journal of Food Protection, 76 (7), 1288–1292. https://doi.org/10.4315/0362-028X.JFP-12-471
Yildiz, G., Palma, S., & Feng, H. (2019). Ultrasonic cutting as a new method to produce fresh-cut red delicious and golden delicious apples. Journal of Food Science, 84 (12), 3391–3398. https://doi.org/10.1111/1750-3841.14798
Yildiz, G., Rababah, T. M., & Feng, H. (2016). Ultrasound-assisted cutting of cheddar, mozzarella and Swiss cheeses – Effects on quality attributes during storage. Innovative Food Science & Emerging Technologies, 37 , 1–9. https://doi.org/10.1016/j.ifset.2016.07.013
Yu, C., Fan, X., Hu, Z., Xia, X., Zhao, Y., Li, R., & Bai, Y. (2020). Segmentation and measurement scheme for fish morphological features based on Mask R-CNN. Information Processing in Agriculture, 7 (4), 523–534. https://doi.org/10.1016/j.inpa.2020.01.002
Zhao, S., Zhang, S., Liu, J., Wang, H., Zhu, J., Li, D., & Zhao, R. (2021). Application of machine learning in intelligent fish aquaculture: A review. Aquaculture . https://doi.org/10.1016/j.aquaculture.2021.736724
Zhong, J., Zhang, F., Lu, Z., Liu, Y., & Wang, X. (2019). High-speed display-delayed planar X-ray inspection system for the fast detection of small fishbones. Journal of Food Process Engineering . https://doi.org/10.1111/jfpe.13010
Zhou, L., Zhang, C., Liu, F., Qiu, Z., & He, Y. (2019). Application of deep learning in food: A review. Comprehensive Reviews in Food Science and Food Safety, 18 (6), 1793–1811. https://doi.org/10.1111/1541-4337.12492
Zhu, L., Spachos, P., Pensini, E., & Plataniotis, K. N. (2021). Deep learning and machine vision for food processing: A survey. Current Research in Food Science, 4 , 233–249. https://doi.org/10.1016/j.crfs.2021.03.009
Zieliński, B., Kapłonek, W., & Nadolny, K. (2018). Regeneration of industrial cutting blades made from X39Cr13 steel used in skinning process of pleuronectidae-family flatfishes. Journal of Mechanical and Energy Engineering , 2 (4), 277–284. https://doi.org/10.30464/jmee.2018.2.4.277
Download references
This work was supported by the Zhejiang Provincial Natural Science Foundation of China [Grant No. LZ23C200005].
Author information
Authors and affiliations.
College of Biosystems Engineering and Food Science, Zhejiang University, 866 Yuhangtang Rd., Hangzhou, Zhejiang, 310058, China
Jiaying Fu, Yingchao He & Fang Cheng
Key Laboratory of Intelligent Equipment and Robotics for Agriculture of Zhejiang Province, Zhejiang University, Hangzhou, Zhejiang, 310058, China
You can also search for this author in PubMed Google Scholar
Contributions
Jiaying Fu: Conceptualization; Methodology; Investigation; Writing—original draft; and Writing—review & editing. Yingchao He: Conceptualization; Methodology; Investigation; and Writing—review & editing. Fang Cheng: Conceptualization; Methodology; Resources; Funding acquisition; and Writing—review & editing; Supervision.
Corresponding author
Correspondence to Fang Cheng .
Ethics declarations
Competing interests.
The authors declare no competing interests.
Additional information
Publisher's note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Reprints and permissions
About this article
Fu, J., He, Y. & Cheng, F. Intelligent Cutting in Fish Processing: Efficient, High-quality, and Safe Production of Fish Products. Food Bioprocess Technol 17 , 828–849 (2024). https://doi.org/10.1007/s11947-023-03163-5
Download citation
Received : 20 May 2023
Accepted : 30 June 2023
Published : 08 July 2023
Issue Date : April 2024
DOI : https://doi.org/10.1007/s11947-023-03163-5
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Cutting techniques
- Image techniques
- Modeling analysis
- Industry 4.0 technologies
- Find a journal
- Publish with us
- Track your research
Information
- Author Services
Initiatives
You are accessing a machine-readable page. In order to be human-readable, please install an RSS reader.
All articles published by MDPI are made immediately available worldwide under an open access license. No special permission is required to reuse all or part of the article published by MDPI, including figures and tables. For articles published under an open access Creative Common CC BY license, any part of the article may be reused without permission provided that the original article is clearly cited. For more information, please refer to https://www.mdpi.com/openaccess .
Feature papers represent the most advanced research with significant potential for high impact in the field. A Feature Paper should be a substantial original Article that involves several techniques or approaches, provides an outlook for future research directions and describes possible research applications.
Feature papers are submitted upon individual invitation or recommendation by the scientific editors and must receive positive feedback from the reviewers.
Editor’s Choice articles are based on recommendations by the scientific editors of MDPI journals from around the world. Editors select a small number of articles recently published in the journal that they believe will be particularly interesting to readers, or important in the respective research area. The aim is to provide a snapshot of some of the most exciting work published in the various research areas of the journal.
Original Submission Date Received: .
- Active Journals
- Find a Journal
- Proceedings Series
- For Authors
- For Reviewers
- For Editors
- For Librarians
- For Publishers
- For Societies
- For Conference Organizers
- Open Access Policy
- Institutional Open Access Program
- Special Issues Guidelines
- Editorial Process
- Research and Publication Ethics
- Article Processing Charges
- Testimonials
- Preprints.org
- SciProfiles
- Encyclopedia


Article Menu

- Subscribe SciFeed
- Recommended Articles
- Author Biographies
- Google Scholar
- on Google Scholar
- Table of Contents
Find support for a specific problem in the support section of our website.
Please let us know what you think of our products and services.
Visit our dedicated information section to learn more about MDPI.
JSmol Viewer
Traditional fish leather dyeing methods with indigenous arctic plants.

1. Introduction
2. the project, 2.1. project aims, 2.2. materials and methods, 2.2.1. fish leather a food waste by-product, 2.2.2. traditional tanning methods, 2.2.3. traditional dyeing methods, 2.3. collection and processing of plant material: extraction of tannins and dyes, 2.4. dyeing process, 3. traditional swedish natural dyes by lotta rahme, 3.1. gallnut tanning, 3.2. sallow bark tanning, 3.3. dyeing with lichen, 3.4. dyeing with roots, 3.5. dyeing with mushrooms, 4. icelandic natural dyes by katrín maría káradóttir and sigmundur páll freysteinsson, 4.1. icelandic lichens, 4.2. icelandic seaweed, 4.3. lupine flowers, 4.4. wood crane’s bill, 5. traditional japanese dyes: matsuyama issey and mitsuhiro kokita, 5.1. koganebana/scutellaria, 5.2. rakkudai/lac dye, 5.3. suoh/sappanwood, 6. traditional indigo dyeing: takayuki ishii, elisa palomino and lotta rahme, 7. light fastness tests, 8. conclusions, author contributions, data availability statement, acknowledgments, conflicts of interest.
- Hurcombe, L.M.; Williams, L. Fish skin as a prehistoric material. Bull. Primit. Technol. 2002 , 23 , 39–41. [ Google Scholar ]
- Hurcombe, L. Perishable Material Culture in Prehistory: Investigating the Missing Majority ; Routledge: London, UK, 2014. [ Google Scholar ]
- Dubreuil, C.O. Ainu Art on the Backs of Gods—Two Exquisite Examples in the DIA Collection. Bull. Detroit Inst. Arts 2002 , 76 , 4–17. [ Google Scholar ] [ CrossRef ]
- Palomino, E. Indigenous Arctic Fish Skin Heritage: Sustainability, Craft and Material Innovation. Ph.D. Thesis, University of the Arts, London, UK, 2022. Available online: https://ualresearchonline.arts.ac.uk/id/eprint/20124/ (accessed on 24 April 2024).
- Cooper, J. The Arctic experience of climate change. In Arctic: Culture and Climate ; Lincoln, A., Cooper, J., Loovers, J.P.L., Eds.; Thames and Hudson; The British Museum: London, UK, 2020. [ Google Scholar ]
- Hald, M. Primitive Shoes: An Archaeological-Ethnological Study Based upon Shoe Finds from the Jutland Peninsula ; Publications of the National Museum, Archaeological-Historical Series; The National Museum of Denmark: Copenhagen, Denmark, 1972; Volume 13. [ Google Scholar ]
- Palomino, E.; Rahme, L. Indigenous Arctic Fish Skin-A study of different traditional skin processing technology. Soc. Leather Technol. Chem. J. 2021 , 105 , 59–71. [ Google Scholar ]
- Pitka Blumenstein, R. Earth Dyes: Nuunam Qaralirkat ; Steinbright, J., Ed.; The Institute of Alaska Native Arts: Fairbanks, AK, USA, 1983. [ Google Scholar ]
- Florian, M.L.E.; Kronkright, D.P.; Norton, R.E. The Conservation of Artifacts Made from Plant Materials ; Getty Conservation Institute: Marina del Rey, CA, USA, 1990. [ Google Scholar ]
- Tamburini, D. (2021) Colour Analysis: An Introduction to the Power of Studying Pigments and Dyes in Archaeological and Historical Objects. Heritage 2021 , 4 , 4366–4371. [ Google Scholar ] [ CrossRef ]
- Budeanu, R.; Curteza, A.; Cezar-Doru, R. Experimental Research Regarding the Ecological Dyeing with Natural Extracts. Autex Res. J. 2014 , 14 , 290–298. [ Google Scholar ] [ CrossRef ]
- Walton, P.; Taylor, G. The characterisation of dyes in textiles from archaeological excavations. Chromatogr. Anal. 1991 , 17 , 5–7. [ Google Scholar ]
- Abid, U.; Mughal, T.A.; Saddiqe, Z.; Anwar, M. Vegetable Tanning of Sole Fish Skin by Using Tannins Extracted from Plants. Asian J. Res. Biosci. 2020 , 2 , 59–67. Available online: https://www.globalpresshub.com/index.php/AJORIB/article/view/882 (accessed on 24 April 2024).
- Forbes, R.J. Studies in Ancient Technology ; E.J. Brill: Leiden, The Netherlands, 1955. [ Google Scholar ]
- Emmerich Kamper, T.; Hurcombe, L. Fur and Skin Processing in Prehistory: An Experimental Approach to Prehistoric Tanning Technologies. In Leather 2019: Proceedings of the 11th Interim Meeting of the ICOM-CC Leather and Related Materials Working Group, Paris, France, 6–7 June 2019 ; Robinet, L., Dignard, C., Sturge, T., Eds.; International Council of Museums—Committee for Conservation (ICOM-CC): Paris, France, 2021; pp. 44–50. [ Google Scholar ]
- Sigfusson, T.; Arnason, R. The Importance of the Ocean Cluster for the Icelandic Economy ; Islandsbanki & Iceland Ocean Cluster: Reykjavik, Iceland, 2012; Available online: http://www.sjavarklasinn.is/wp-content/uploads/2012/03/Sjavarklasinn_Skyrsla-enska-low.pdf (accessed on 24 April 2024).
- Rahme, L.; Hartman, D. Fish Leather: Tanning and Sewing with Traditional Methods ; Lottas Tannery: Sigtuna, Sweden, 2012. [ Google Scholar ]
- Reed, D. Anciente leathers, parchments and skin products. In Konservering og Restaurering of Leader, Skin Dog Pergament. Compendium fra Nordisk Videreuddannelseskursus ; Konservatorskolen: Kobenhavn, Denmark, 1980. [ Google Scholar ]
- Waterer, J.W. Leather in Life, Art and Industry, Being an Outline of Its Preparation and Uses in Britain Yesterday and Today, Together with Some Reflections on Its Place in the World of Synthetics Tomorrow ; Faber and Faber: London, UK, 1946. [ Google Scholar ]
- Leather International (2013) ‘A History of New Ideas in Tanning’ Leather International. Available online: https://www.leathermag.com/features/featurea-history-of-new-ideas-intanning/ (accessed on 24 April 2024).
- Martin, T. The Circle of the Mechanical Arts ; The Toolemera Press: London, UK, 1813; pp. 542–546. [ Google Scholar ]
- Pluenneke, M. Shift in Icelandic Plant Populations Due to Climate Change: Through the Lens of Natural Dyes. Independent Study Project (ISP) Collection 2751. 2017. Available online: https://digitalcollections.sit.edu/isp_collection/2751 (accessed on 24 April 2024).
- Magnus, O. A Description of the Northern Peoples ; Thirteenth Book; Routledge: Abingdon, UK, 1555; Volume 48, p. 632. [ Google Scholar ]
- Svanberg, I. Plant Knowledge as Indicator of Historical Cultural Contacts: Tanning in the Atlantic Fringe. In Traveling Cultures and Plants: The Ethnobiology and Ethnopharmacy of Human Migrations ; Pieroni, A., Vandebroek, I., Eds.; Berghahn Books: New York, NY, USA, 2009. [ Google Scholar ]
- Falcão, L.; Araújo, M.E.M. Vegetable Tannins Used in the Manufacture of Historic Leathers. Molecules 2018 , 23 , 1081. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ PubMed Central ]
- Potts, D.T. Mesopotamian Civilization: The Material Foundations ; Cornell University Press: Ithaca, NY, USA, 1997. [ Google Scholar ]
- Fatma, N.; Sarikaya, H. Investigation of nutgall and some natural dyes with mordants cotton dyeing and fastness level in the context of the ecological textile production. IJASOS Int. E-J. Adv. Soc. Sci. 2015 , 1 , 452–459. [ Google Scholar ]
- Flemestad, P. Theophrastos of Eresos on Plants for Dyeing and Tanning. In Purpureae Vestes IV: Textiles and Dyes in Antiquity. Production and Trade of Textiles and Dyes in the Roman Empire and Neighbouring Regions: Actas del IV Symposium Internacional Sobre Textiles y Tintes del Mediterráneo en el Mundo Antiguo ; Alfaro, C., Tellenbach, M., Ortiz, J., Eds.; Universitat de València: Valencia, Spain, 2014; pp. 203–209. [ Google Scholar ]
- Rodenstam, S. Hemfärgning med växtämnen Hemmets stora rådgivare ; AB Nordiska Bokförlaget, Erdheim & Co.: Jersey, Island, 1928; pp. 353–379. [ Google Scholar ]
- Hatt, G. Arctic Skin Clothing in Eurasia and America an Ethnographic Study. Arct. Anthropol. 1969 , 5 , 3–132. [ Google Scholar ]
- Holmberg, A.E. Bohusläns historia och beskrifning ; Del 1. s 300; N.M Lindh: Örebro, Sweden, 1867. [ Google Scholar ]
- Linnaeus, C.V. Öländska och Gotländska resa ; Wahlström & Widstrand: Stockholm, Sweden, 1741; p. 33. [ Google Scholar ]
- Westring, P. Svenska Lafvarnas Färghistoria ; J.P Första Bandet: Stockholm, Sweden, 1805. [ Google Scholar ]
- Bjarnadóttir, G.; Hilmarsson, J.Ó. Grasnytjar á Íslandi—Þjóðtrú og Saga ; Hespuhúsið: Ölfus, Iceland, 2018. [ Google Scholar ]
- Friend, H. Flower Lore ; Para Research: Rockport, MA, USA, 1981. [ Google Scholar ]
- Takahashi, T.; Tsurunaga, Y.; Aso, Y.; Yoshino, K. Color Fastness of Sappanwood-Dyed Silk and Insights into the Clothing Life of the Heian Period. J. Fiber Sci. Technol. 2016 , 72 , 206–219. [ Google Scholar ] [ CrossRef ]
- Toho University Fauculty of Pharmaceutical Sciences Medicinal Botanical Garden. Scutellaria Baicalensis. 2022. Available online: https://www.lab2.toho-u.ac.jp/ (accessed on 24 April 2024).
- Yamazaki, A. Japanese Plant Dyeing ; Textile and Lifestyle Ltd.: Tokyo, Japan, 1986; pp. 38–39. [ Google Scholar ]
- Yamazaki, A. Monograph of Plant-Dyeing Peculiar to Japan (Nippon Kuzaki-Zome Fu) ; Getsumeikai-Ltd.: Yamaguchi, Japan, 1961; pp. 16–17. [ Google Scholar ]
- DuFault, A. The Many Faces of Sappanwood Extract. 2021. Available online: https://botanicalcolors.com/the-many-faces-of-sappanwood-extract-just-add-more-or-less-dye-iron/ (accessed on 24 April 2024).
- Fitzhugh, E.W. Artists’ Pigments—A Handbook of Their History and Characteristics ; National Gallery of Art: Washington, DC, USA, 1997; Volume 3, pp. 191–217. [ Google Scholar ]
- Schweppe, H. Indigo and Woad in E.W. Fitzhugh. Artists’ Pigments: A Handbook of Their History and Characteristics ; National Gallery of Art: Washington, DC, USA, 1997; Volume 3, pp. 81–108. [ Google Scholar ]
- Shimoyama, S.; Noda, Y. Non-destructive analysis of dyes in a Chinese brocade. In Dyes in History and Archaeology ; Textile Research Associates: Clarksville, TN, USA, 1996; Volume 15, pp. 70–84. [ Google Scholar ]
- Tokushima Prefecture (2022) “Awa Indigo” Supporting Japan Blue. Available online: https://www.pref.tokushima.lg.jp/en/japanese/natural_culture/traditional_culture/awa-ai (accessed on 24 April 2024).
- Ishii, T. The Way of Indigo ; Graham Keegan: Los Angeles, CA, USA, 2021. [ Google Scholar ]
- ISO 15701:2022|IULTCS/IUF 412 ; Leather—Tests for Colour Fastness—Colour Fastness to Migration into Polymeric Material. International Organization for Standardization: Geneva, Switzerland, 2022. Available online: https://www.iso.org/standard/81922.html (accessed on 24 April 2024).
- ISO 11641:2012|IULTCS/IUF 426 ; Leather—Tests for Colour Fastness—Colour Fastness to Perspiration. International Organization for Standardization: Geneva, Switzerland, 2012. Available online: https://www.iso.org/standard/54445.html (accessed on 24 April 2024).
- Textiles—Tests for Colour Fastness Part B02: Colour Fastness to Artificial Light: Xenon arc Fading lamp Test ; International Organization for Standardization: Geneva, Switzerland, 2014. Available online: https://www.iso.org/standard/65209.html (accessed on 24 April 2024).
Click here to enlarge figure
| The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
Share and Cite
Palomino, E.; Rahme, L.; Káradóttir, K.M.; Kokita, M.; Freysteinsson, S.P. Traditional Fish Leather Dyeing Methods with Indigenous Arctic Plants. Heritage 2024 , 7 , 3643-3663. https://doi.org/10.3390/heritage7070173
Palomino E, Rahme L, Káradóttir KM, Kokita M, Freysteinsson SP. Traditional Fish Leather Dyeing Methods with Indigenous Arctic Plants. Heritage . 2024; 7(7):3643-3663. https://doi.org/10.3390/heritage7070173
Palomino, Elisa, Lotta Rahme, Katrín María Káradóttir, Mitsuhiro Kokita, and Sigmundur Páll Freysteinsson. 2024. "Traditional Fish Leather Dyeing Methods with Indigenous Arctic Plants" Heritage 7, no. 7: 3643-3663. https://doi.org/10.3390/heritage7070173
Article Metrics
Article access statistics, further information, mdpi initiatives, follow mdpi.

Subscribe to receive issue release notifications and newsletters from MDPI journals
Your browser is not supported
Sorry but it looks as if your browser is out of date. To get the best experience using our site we recommend that you upgrade or switch browsers.
Find a solution
- Skip to main content
- Skip to navigation
- hot-topics Extras
- Newsletters
- Reading room
Tell us what you think. Take part in our reader survey
Celebrating twenty years
- Back to parent navigation item
- Collections
- Chemistry of the brain
- Water and the environment
- Chemical bonding
- Antimicrobial resistance
- Energy storage and batteries
- AI and automation
- Sustainability
- Research culture
- Nobel prize
- Food science and cookery
- Plastics and polymers
- Periodic table
- Coronavirus
- More navigation items
Zinc–air batteries created from paper industry waste

- No comments
A team of researchers has shown that paper industry waste can be transformed into a zinc–air battery. 1 The battery, created from cellulose and lignin, exhibited a high power density and a wide operating temperature, while avoiding the longstanding issue of dendrite growth.
Zinc–air batteries are popular due to their low cost and high energy density. They are environmentally friendly and have a good safety profile, carrying none of the thermal runaway risks associated with lithium-ion batteries. These benefits facilitate their use in wearable electronics, such as hearing aids. 2
The wood-based battery created by Lei Zhang of Qilu University of Technology in China and colleagues uses an iron cluster single atom catalyst as the air cathode and lignin from pulping black liquor as the carbon source. The study states that the single atom catalyst displays exceptional oxygen reduction reaction performance and the specific surface area exceeds that of most reported iron-based single atom catalysts. In contrast, traditional transition metal-based single atom catalysts often suffer from slow electron transfer during the oxygen reduction and oxygen evolution reactions.
The battery also contains a polyvinyl alcohol organohydrogel solid state electrolyte reinforced with cationic nanocellulose from pulped wood. Compared to conventional aqueous or alkaline hydrogel electrolytes, this cellulose-reinforced electrolyte showed improved flexibility and mechanical strength in addition to increased water retention capacity and ionic conductivity, which ensures continued stability and function. Moreover, conventional aqueous or alkaline hydrogel electrolytes exhibit poor electrolyte interface stability and dendrite growth, hindering long-term battery function. Functionalised solid-state electrolytes, such as this, can prevent zinc dendrite growth and improve the operational temperature range of the battery.
The study reports that the battery demonstrates ‘high power density and remarkable cycling stability’ over a wide temperature range (−60°C to 50°C). And that it surpasses most reported Zn–air batteries, highlighting their potential for clean energy storage and conversion applications.

Source: © Lei Zhang/Qilu University of Technology
The batteries still performed well at very low temperatures
‘What [they did] here is solve problems […] related to lithium–air rechargeable batteries,’ says Thomas Gregory , a battery expert and consultant at Borealis Technology Solutions, US. ‘One being the need for a low cost, high functioning electric catalyst for the air electrode. The other one being an electrolyte which resists dendrite penetration and suppresses dendrite growth at the zinc electrode. Those have been longstanding issues… and what [they have done] is address that using relatively low cost, abundant components.’
Beyond wearable electronics, there is a growing interest in using zinc–air batteries for larger scale energy storage, especially in more challenging environments. However, this is a small-scale study using a single source of wood, specifically eucalyptus. ‘How does this technology behave in a larger format?’ asks Gregory. ‘If they used a different wood species from a different source, would they behave the same electrochemically?’ These questions must be answered to enable larger scale applications and increase the pool of sustainable materials in the future.
1 J Chen et al , Energy Environ. Sci. , 2024, DOI: 10.1039/d4ee01226g
2 P Tan et al , Energy Environ. Sci. , 2017, 10 , 2056 (DOI: 10.1039/c7ee01913k )

More Lauren Yarrow-Wright

Chemists and linguists unite to encode Confucian proverb using sequence-defined polymers
- Electronic materials
- Energy storage
- Renewable feedstocks
Related articles

World’s largest sodium–ion battery goes live
2024-07-10T13:30:00Z
By Jamie Durrani

Intelligent ionotronic wood device can keep an eye on people’s health
2024-04-03T13:30:00Z
By Zahra Khan

Clever cathode design opens doors to first rechargeable calcium battery
2024-02-07T16:00:00Z
By Victoria Atkinson

The long-term energy storage challenge
2023-04-24T10:57:00Z
By Rachel Brazil

The wonderful wizards of wood
2023-02-20T11:18:00Z
By Kit Chapman

Sub-zero water splitting marks a new dawn for solar hydrogen production
2021-07-05T13:35:00Z
By Jamie Purcell
No comments yet
Only registered users can comment on this article..

Spanish government unveils new cross-collaborative strategy for scientific policy
2024-07-12T08:30:00Z
By Fernando Gomollón-Bel

How can your lab cut water use in reflux reactions?
2024-07-11T13:30:00Z
By Andy Extance
Explainer: How is the US pharmaceutical patent system being misused?
2024-07-11T09:26:00Z
By Julia Robinson

Glycolysis method breaks down mixed textiles for recycling
2024-07-10T08:30:00Z
By Tosin Thompson

US–China tensions appear to be damaging countries’ science, analysis finds
2024-07-09T13:57:00Z
By Rebecca Trager
Plastic recycling studies need reliable polymer data. This database is ready to inform them
2024-07-09T07:24:00Z
By Hugh Ryan
- Contributors
- Terms of use
- Accessibility
- Permissions
- This website collects cookies to deliver a better user experience. See how this site uses cookies .
- This website collects cookies to deliver a better user experience. Do not sell my personal data .
- Este site coleta cookies para oferecer uma melhor experiência ao usuário. Veja como este site usa cookies .
Site powered by Webvision Cloud
The trends defining the $1.8 trillion global wellness market in 2024
From cold plunges to collagen to celery juice, the $1.8 trillion global consumer wellness market is no stranger to fads, which can sometimes surface with limited clinical research or credibility. Today, consumers are no longer simply trying out these wellness trends and hoping for the best, but rather asking, “What does the science say?”
About the authors
This article is a collaborative effort by Shaun Callaghan , Hayley Doner, Jonathan Medalsy, Anna Pione , and Warren Teichner , representing views from McKinsey’s Consumer Packaged Goods and Private Equity & Principal Investors Practices.
McKinsey’s latest Future of Wellness research—which surveyed more than 5,000 consumers across China, the United Kingdom, and the United States—examines the trends shaping the consumer wellness landscape. In this article, we pair these findings with a look at seven wellness subsets—including women’s health, weight management, and in-person fitness—that our research suggests are especially ripe areas for innovation and investment activity.
The science- and data-backed future of wellness
In the United States alone, we estimate that the wellness market has reached $480 billion, growing at 5 to 10 percent per year. Eighty-two percent of US consumers now consider wellness a top or important priority in their everyday lives, which is similar to what consumers in the United Kingdom and China report (73 percent and 87 percent, respectively).
This is especially true among Gen Z and millennial consumers, who are now purchasing more wellness products and services than older generations, across the same dimensions we outlined in our previous research : health, sleep, nutrition, fitness, appearance, and mindfulness (Exhibit 1). 1 “ Still feeling good: The US wellness market continues to boom ,” McKinsey, September 19, 2022.
Across the globe, responses to our survey questions revealed a common theme about consumer expectations: consumers want effective, data-driven, science-backed health and wellness solutions (Exhibit 2).
Five trends shaping the consumer health and wellness space in 2024
Fifty-eight percent of US respondents to our survey said they are prioritizing wellness more now than they did a year ago. The following five trends encompass their newly emerging priorities, as well as those that are consistent with our earlier research.

Trend one: Health at home
The COVID-19 pandemic made at-home testing kits a household item. As the pandemic has moved into its endemic phase, consumers are expressing greater interest in other kinds of at-home kits: 26 percent of US consumers are interested in testing for vitamin and mineral deficiencies at home, 24 percent for cold and flu symptoms, and 23 percent for cholesterol levels.
At-home diagnostic tests are appealing to consumers because they offer greater convenience than going to a doctor’s office, quick results, and the ability to test frequently. In China, 35 percent of consumers reported that they had even replaced some in-person healthcare appointments with at-home diagnostic tests—a higher share than in the United States or the United Kingdom.
Although there is growing interest in the space, some consumers express hesitancy. In the United States and the United Kingdom, top barriers to adoption include the preference to see a doctor in person, a perceived lack of need, and price; in China, test accuracy is a concern for approximately 30 percent of consumers.
Implications for companies: Companies can address three critical considerations to help ensure success in this category. First, companies will want to determine the right price value equation for at-home diagnostic kits since cost still presents a major barrier for many consumers today. Second, companies should consider creating consumer feedback loops, encouraging users to take action based on their test results and then test again to assess the impact of those interventions. Third, companies that help consumers understand their test results—either through the use of generative AI to help analyze and deliver personalized results, or through integration with telehealth services—could develop a competitive advantage.
Trend two: A new era for biomonitoring and wearables
Roughly half of all consumers we surveyed have purchased a fitness wearable at some point in time. While wearable devices such as watches have been popular for years, new modalities powered by breakthrough technologies have ushered in a new era for biomonitoring and wearable devices.
Wearable biometric rings, for example, are now equipped with sensors that provide consumers with insights about their sleep quality through paired mobile apps. Continuous glucose monitors, which can be applied to the back of the user’s arm, provide insights about the user’s blood sugar levels, which may then be interpreted by a nutritionist who can offer personalized health guidance.
Roughly one-third of surveyed wearable users said they use their devices more often than they did last year, and more than 75 percent of all surveyed consumers indicated an openness to using a wearable in the future. We expect the use of wearable devices to continue to grow, particularly as companies track a wider range of health indicators.
Implications for companies: While there is a range of effective wearable solutions on the market today for fitness and sleep, there are fewer for nutrition, weight management, and mindfulness, presenting an opportunity for companies to fill these gaps.
Wearables makers and health product and services providers in areas such as nutrition, fitness, and sleep can explore partnerships that try to make the data collected through wearable devices actionable, which could drive greater behavioral change among consumers. One example: a consumer interested in managing stress levels might wear a device that tracks spikes in cortisol. Companies could then use this data to make personalized recommendations for products related to wellness, fitness, and mindfulness exercises.
Businesses must keep data privacy and clarity of insights top of mind. Roughly 30 percent of China, UK, and US consumers are open to using a wearable device only if the data is shared exclusively with them. Additionally, requiring too much manual data input or sharing overly complicated insights could diminish the user experience. Ensuring that data collection is transparent and that insights are simple to understand and targeted to consumers’ specific health goals or risk factors will be crucial to attracting potential consumers.
Trend three: Personalization’s gen AI boost
Nearly one in five US consumers and one in three US millennials prefer personalized products and services. While the preference for personalized wellness products was lower than in years prior, we believe this is likely due to consumers becoming more selective about which personalized products and services they use.
Technological advancements and the rise of first-party data are giving personalization a new edge. Approximately 20 percent of consumers in the United Kingdom and the United States and 30 percent in China look for personalized products and services that use biometric data to provide recommendations. There is an opportunity to pair these tools with gen AI to unlock greater precision and customization. In fact, gen AI has already made its way to the wearables and app space: some wearables use gen AI to design customized workouts for users based on their fitness data.
Implications for companies: Companies that offer software-based health and wellness services to consumers are uniquely positioned to incorporate gen AI into their personalization offerings. Other businesses could explore partnerships with companies that use gen AI to create personalized wellness recommendations.
Trend four: Clinical over clean
Last year, we saw consumers begin to shift away from wellness products with clean or natural ingredients to those with clinically proven ingredients. Today, that shift is even more evident. Roughly half of UK and US consumers reported clinical effectiveness as a top purchasing factor, while only about 20 percent reported the same for natural or clean ingredients. This trend is most pronounced in categories such as over-the-counter medications and vitamins and supplements (Exhibit 3).
In China, consumers expressed roughly equal overall preference for clinical and clean products, although there were some variations between categories. They prioritized clinical efficacy for digestive medication, topical treatments, and eye care products, while they preferred natural and clean ingredients for supplements, superfoods, and personal-care products.
Implications for companies: To meet consumer demand for clinically proven products, some brands will be able to emphasize existing products in their portfolios, while other businesses may have to rethink product formulations and strategy. While wellness companies that have built a brand around clean or natural products—particularly those with a dedicated customer base—may not want to pivot away from their existing value proposition, they can seek out third-party certifications to help substantiate their claims and reach more consumers.
Companies can boost the clinical credibility of their products by using clinically tested ingredients, running third-party research studies on their products, securing recommendations from healthcare providers and scientists, and building a medical board that weighs in on product development.
Trend five: The rise of the doctor recommendation
The proliferation of influencer marketing in the consumer space has created new sources of wellness information—with varying degrees of credibility. As consumers look to avoid “healthwashing” (that is, deceptive marketing that positions a product as healthier than it really is), healthcare provider recommendations are important once again.
Doctor recommendations are the third-highest-ranked source of influence on consumer health and wellness purchase decisions in the United States (Exhibit 4). Consumers said they are most influenced by doctors’ recommendations when seeking care related to mindfulness, sleep, and overall health (which includes the use of vitamins, over-the-counter medications, and personal- and home-care products).
Implications for companies: Brands need to consider which messages and which messengers are most likely to resonate with their consumers. We have found that a company selling products related to mindfulness may want to use predominately doctor recommendations and social media advertising, whereas a company selling fitness products may want to leverage recommendations from friends and family, as well as endorsements from personal trainers.
Seven areas of growth in the wellness space
Building upon last year’s research, several pockets of growth in the wellness space are emerging. Increasing consumer interest, technological breakthroughs, product innovation, and an increase in chronic illnesses have catalyzed growth in these areas.
Women’s health
Historically, women’s health has been underserved and underfunded . Today, purchases of women’s health products are on the rise across a range of care needs (Exhibit 5). While the highest percentage of respondents said they purchased menstrual-care and sexual-health products, consumers said they spent the most on menopause and pregnancy-related products in the past year.
Digital tools are also becoming more prevalent in the women’s health landscape. For example, wearable devices can track a user’s physiological signals to identify peak fertility windows.
Despite recent growth in the women’s health space, there is still unmet demand for products and services. Menopause has been a particularly overlooked segment of the market: only 5 percent of FemTech start-ups address menopause needs. 2 Christine Hall, “Why more startups and VCs are finally pursuing the menopause market: ‘$600B is not “niche,”’” Crunchbase, January 21, 2021. Consumers also continue to engage with offerings across the women’s health space, including menstrual and intimate care, fertility support, pregnancy and motherhood products, and women-focused healthcare centers, presenting opportunities for companies to expand products and services in these areas.
Healthy aging
Demand for products and services that support healthy aging and longevity is on the rise, propelled by a shift toward preventive medicine, the growth of health technology (such as telemedicine and digital-health monitoring), and advances in research on antiaging products.
Roughly 70 percent of consumers in the United Kingdom and the United States and 85 percent in China indicated that they have purchased more in this category in the past year than in prior years.
More than 60 percent of consumers surveyed considered it “very” or “extremely” important to purchase products or services that help with healthy aging and longevity. Roughly 70 percent of consumers in the United Kingdom and the United States and 85 percent in China indicated that they have purchased more in this category in the past year than in prior years. These results were similar across age groups, suggesting that the push toward healthy aging is spurred both by younger generations seeking preventive solutions and older generations seeking to improve their longevity. As populations across developed economies continue to age (one in six people in the world will be aged 60 or older by 2030 3 “Ageing and health,” World Health Organization, October 1, 2022. ), we expect there to be an even greater focus globally on healthy aging.
To succeed in this market, companies can take a holistic approach to healthy-aging solutions , which includes considerations about mental health and social factors. Bringing products and services to market that anticipate the needs of aging consumers—instead of emphasizing the aging process to sell these products—will be particularly important. For example, a service that addresses aging in older adults might focus on one aspect of longevity, such as fitness or nutrition, rather than the process of aging itself.
Weight management
Weight management is top of mind for consumers in the United States, where nearly one in three adults struggles with obesity 4 Obesity fact sheet 508 , US Centers for Disease Control and Prevention, July 2022. ; 60 percent of US consumers in our survey said they are currently trying to lose weight.
While exercise is by far the most reported weight management intervention in our survey, more than 50 percent of US consumers considered prescription medication, including glucagon-like peptide-1 (GLP-1) drugs, to be a “very effective” intervention. Prescription medication is perceived differently elsewhere: less than 30 percent of UK and China consumers considered weight loss drugs to be very effective.
Given the recency of the GLP-1 weight loss trend, it is too early to understand how it will affect the broader consumer health and wellness market. Companies should continue to monitor the space as further data emerges on adoption rates and impact across categories.
In-person fitness
Fitness has shifted from a casual interest to a priority for many consumers: around 50 percent of US gym-goers said that fitness is a core part of their identity (Exhibit 6). This trend is even stronger among younger consumers—56 percent of US Gen Z consumers surveyed considered fitness a “very high priority” (compared with 40 percent of overall US consumers).
In-person fitness classes and personal training are the top two areas where consumers expect to spend more on fitness. Consumers expect to maintain their spending on fitness club memberships and fitness apps.
The challenge for fitness businesses will be to retain consumers among an ever-increasing suite of choices. Offering best-in-class facilities, convenient locations and hours, and loyalty and referral programs are table stakes. Building strong communities and offering experiences such as retreats, as well as services such as nutritional coaching and personalized workout plans (potentially enabled by gen AI), can help top players evolve their value proposition and manage customer acquisition costs.
More than 80 percent of consumers in China, the United Kingdom, and the United States consider gut health to be important, and over 50 percent anticipate making it a higher priority in the next two to three years.
One-third of US consumers, one-third of UK consumers, and half of Chinese consumers said they wish there were more products in the market to support their gut health.
While probiotic supplements are the most frequently used gut health products in China and the United States, UK consumers opt for probiotic-rich foods such as kimchi, kombucha, or yogurt, as well as over-the-counter medications. About one-third of US consumers, one-third of UK consumers, and half of Chinese consumers said they wish there were more products in the market to support their gut health. At-home microbiome testing and personalized nutrition are two areas where companies can build on the growing interest in this segment.
Sexual health
The expanded cultural conversation about sexuality, improvements in sexual education, and growing support for female sexual-health challenges (such as low libido, vaginal dryness, and pain during intercourse) have all contributed to the growth in demand for sexual-health products.
Eighty-seven percent of US consumers reported having spent the same or more on sexual-health products in the past year than in the year prior, and they said they purchased personal lubricants, contraceptives, and adult toys most frequently.
While more businesses began to sell sexual-health products online during the height of the COVID-19 pandemic, a range of retailers—from traditional pharmacies to beauty retailers to department stores—are now adding more sexual-health brands and items to their store shelves. 5 Keerthi Vedantam, “Why more sexual wellness startups are turned on by retail,” Crunchbase, November 15, 2022. This creates marketing and distribution opportunities for disruptor brands to reach new audiences and increase scale.
Despite consistently ranking as the second-highest health and wellness priority for consumers, sleep is also the area where consumers said they have the most unmet needs. In our previous report, 37 percent of US consumers expressed a desire for additional sleep and mindfulness products and services, such as those that address cognitive functioning, stress, and anxiety management. In the year since, little has changed. One of the major challenges in improving sleep is the sheer number of factors that can affect a good night’s sleep, including diet, exercise, caffeination, screen time, stress, and other lifestyle factors. As a result, few, if any, tech players and emerging brands in the sleep space have been able to create a compelling ecosystem to improve consumer sleep holistically. Leveraging consumer data to address specific pain points more effectively—including inducing sleep, minimizing sleep interruptions, easing wakefulness, and improving sleep quality—presents an opportunity for companies.
As consumers take more control over their health outcomes, they are looking for data-backed, accessible products and services that empower them to do so. Companies that can help consumers make sense of this data and deliver solutions that are personalized, relevant, and rooted in science will be best positioned to succeed.
Shaun Callaghan is a partner in McKinsey’s New Jersey office; Hayley Doner is a consultant in the Paris office; and Jonathan Medalsy is an associate partner in the New York office, where Anna Pione is a partner and Warren Teichner is a senior partner.
The authors wish to thank Celina Bade, Cherry Chen, Eric Falardeau, Lily Fu, Eric He, Sara Hudson, Charlotte Lucas, Maria Neely, Olga Ostromecka, Akshay Rao, Michael Rix, and Alex Sanford for their contributions to this article.
This article was edited by Alexandra Mondalek, an editor in the New York office.
Explore a career with us
Related articles.

Still feeling good: The US wellness market continues to boom

How to thrive in the global wellness market

Feeling good: The future of the $1.5 trillion wellness market

COMMENTS
An International Journal on Fisheries Science, Fishing Technology and Fisheries Management This journal provides an international forum for the publication of papers in the areas of fisheries science, fishing technology, fisheries management and relevant socio-economics. The scope covers fisheries in salt, brackish and freshwater systems, and all aspects of associated ecology, environmental ...
Market system comprises wholesale market, retail market, and fish retail markets (Gupta & Gupta, Citation 2008). The increase in quantity of fish production as well as advancement of fishery sector in terms of economy and infrastructure is profoundly dependent on an efficient fish marketing system (Chourey et al., Citation 2014). Local fish ...
The FAO notes that over 34% of fish stocks are overfished, emphasizing the need for better fisheries management. ... The aim was to retrieve and select those papers that investigate the utilization of new seafood emerging technologies, in order to evaluate the research trends based on literature in the Web of Science and Elsevier Scopus ...
1. Introduction. There is increasing depletion of wild fish stocks globally, where fish is an important source of high protein products and omega 3 (FAO, 2020).Aquaculture has been one of the fastest growing food sectors in agriculture globally (Tahar, Kennedy, et al., 2018; 2018b; FAO, 2020).Xia et al. (2022) noted that aquaculture has produced more fish for human consumption than wild-caught ...
1. Introduction. Fish production is a growing industry, with more than 196 million tons of fish predicted to be processed in 2025 (Sónia & Maria, 2019, pp. 185-211).This industry generates large amounts of by-products, such as heads, skin, trimmings, fins, viscera, frames, and sometimes muscle, which are currently wasted, underutilized, or used to produce low value-added products such as ...
Fishing, as the world's last major hunting and gathering industry, supports livelihoods, food security, and human health (1-4).However, it is unclear whether, and if so, how fishing can achieve the aspirations of the United Nations that specify three pillars of sustainability: economic development, social development, and environmental protection ().
Research into fish chemoreception was largely unexploited by the recreational fishing tackle industry until the mid-1980s. Nearly all of the so called "fish scents" on the market in the 1980s were based on various fish oil formulas, which were insoluble in water and thus incompatible with the olfactory and gustatory organs of fish (Carr and ...
Conclusion: sustainability in the fish-processing industry References Ayer, N , Cote, R P , Tyedmers, P H and Martin Willson, J H , 2009 , Sustainability of seafood production and consumption: An introduction to this special issue , Journal of Cleaner Production , 17 , 3 , 321 - 324 .
The rising consumption trends along with the establishment of Exclusive Economic Zones and related international agreements have boosted the international trade in fish and fish products (Ababouch, 2006; Anderson & Fong, 1997; Shinoj et al., 2009).The foreign exchange earnings from fisheries also demonstrate an increasing trend, and thus this sector possesses a great potential for developing ...
Affiliations 1 Key Laboratory of Aquatic Product Processing, Ministry of Agriculture and Rural Affairs of The People's Republic of China, National R&D Center for Aquatic Product Processing, South China Fisheries Research Institute, Chinese Academy of Fishery Sciences, Guangzhou, China.; 2 Co-Innovation Center of Jiangsu Marine Bio-industry Technology, Jiangsu Ocean University, Lianyungang, China.
In the 1980s alone, the global ornamental fish industry was estimated to be worth USD 7.2 billion and in 1997 it had reached between USD 20 billion and 30 billion [16,29,30,31,32]. The discrepancies grew extensively in 2004 when the total value was estimated to be between USD 800 million and 30 billion annually [7,33,34,35] (Figure 1).
The aquaculture industry has developed rapidly since aquaculture production is the only substitute for reduced production from capture fisheries. In the aquaculture industry, the use of breeding in purpose-built hatcheries and culturing in ponds, cages, or tanks helped to boost technological advances (Burnell and Allan 2009). Aquaculture has ...
United Nation's Sustainable Development Goal (SDG) 14 'life below water', is directed to the sustainable use and conservation of the oceans and marine resources. However, there is very limited information available on how the large-scale commercial fishing industry might contribute to the achievement of SDG 14. This paper shows engagement opportunities for the fishing industry, with a ...
2 University of Coimbra, MARE-Marine and Environmental Sciences Centre/ARNET-Aquatic Research. Network, Department of Life Sciences, Calçada Martim de Freitas, 3000-456, Coimbra, Portugal ...
Fish and fishery products are the most traded food commodities across the world. However, fermented fish are nominal in the global food map due to its strong odoriferous flavour that repulse western taste (Sally, 2020).Fermented fish products are popular cuisine in Africa, Asia, and Europe (Xu et al., 2021).Each continent has unique fermented fish products prepared in an artisanal way based on ...
Fish waste (mainly heads, bones, skin, viscera and sometimes whole fish and parsley) was heated at 65, 80, 105 and 150 °C for 12 h in order to reduce the moisture content to 10-12%, which is the recommended moisture content in animal feed (NRC (National Research Council), 1998). Fish waste proved to be a great source of minerals, protein [58 ...
3. Fish Byproducts as Source of High Added-Value Compounds. Due to this massive increase in fish consumption, in 2015 fish accounted for approximately 17% of the global population's intake of animal protein and 6.7% of all proteins consumed in the world [].A fish-based diet has a huge positive nutritional impact, also playing an important role in correcting unbalanced diets and countering ...
This food fi sh supply is increasing at a rate. 3.2% each year since 2005, far out pacing the world population growth 1.6% (FAOUN 2018). Figure 1. Total tonnes of fish caught each year 1950 ...
In 2018, the total production from three sectors, e.g., aquaculture, municipal, and commercial fisheries, was about 4.36 million MT. With this, the Philippines ranked 13th as the top fish ...
This extensive research paper examines the history and cultural development of the fishing industry in Japan. In addition, it looks at sustainable and unsustainable practices and their solutions and culture's influence on sustainability.
Fish processing is an indispensable part of fish food production. It mainly involves de-heading, gutting, filleting, skinning, trimming, and slicing, with the cutting operations holding a critical role. Unfortunately, inefficiency, low quality, and poor safety are the primary problems facing the fish processing industry today, dramatically hindering the automation and intelligence of fish ...
Research on sustainability entrepreneurship and industry creation often highlights how value-driven agents individually and collectively create significant and lasting change. We attempt to complement these agent-focused approaches by addressing the underlying business-environmental conditions that make participation in industry creation attractive and potentially successful. Accordingly, we ...
Moreover, coupled with the current estimated use of 5 to 6 million tonnes of trash fish as a direct food source for farmed fish, it is estimated that the aquaculture sector consumed the equivalent ...
Along the Arctic and sub-Arctic coasts of Alaska, Siberia, north-eastern China, Hokkaido, Scandinavia and Iceland, people have dressed in clothes or worn shoes made of fish skin for millennia. (Within this article, the terms fish skin and fish leather are used to indicate different processes of the same material. Fish skin: Skin indicates the superficial dermis of an animal. Fish skin is ...
A team of researchers has shown that paper industry waste can be transformed into a zinc-air battery. 1 The battery, created from cellulose and lignin, exhibited a high power density and a ...
Highlights of the White Paper. The White Paper on International Economy and Trade 2024 primarily presents the importance of the reconstruction of a rule-based international trade order, amid a global economy facing a crisis of division, and the significance of strengthening collaboration with the Global South countries, which are expected to achieve high growth and may become important ...
A review on the cold chain management of fish and fish products in developing countries. Journal of Cleaner Production, 234, 1201-1212. Does aquaculture add resilience to the global food system
This is especially true among Gen Z and millennial consumers, who are now purchasing more wellness products and services than older generations, across the same dimensions we outlined in our previous research: health, sleep, nutrition, fitness, appearance, and mindfulness (Exhibit 1). 1 "Still feeling good: The US wellness market continues to ...
Abstract: Marketing of fish and fishery products in the three markets of Gazipur district were studied during the. period from November 2014 to April 2015. Data were collected from thre e ...
Greater availability of financial access related data in recent years is increasingly enabling policymakers to better track and monitor financial access trends and developments. However, data on barriers to financial access, including costs associated with using financial services—a key factor of financial exclusion—remain scarce. To gain insight into the costs of financial access faced by ...