

Top 17 Clinical Research Organizations (CRO) in 2023
In clinical research and treatment development, clinical research organizations (CROs) are frequently a sponsor’s most important partner and ally.
Depending on the nature of the clinical trial, and your existing capabilities as a sponsor to run the trial, the CRO company of your choice will typically be responsible for facilitating most of the micro and macro processes that go into designing and running a successful clinical trial.
When contracting a CRO to help you with your trial, you are transferring over a large portion of responsibility into the hands of your clinical research partner. The CRO of your choice will have the responsibility to control a variety of factors and processes of a clinical trial, and depending on their expertise, team structures, service offerings, internal resources and many other capabilities.
Your ability to find and contract a top CRO company that is the right fit for your unique trial will be a determinant of whether or not you will be able to operate a high-quality clinical trial that meets your expected timelines, budget and delivers a top-notch patient experience.
At ClaraHealth (a patient-centric recruitment acceleration platform) , we have put together an extensive list of the top CRO companies in the US and around the world.
This is not a cro rankings list, but rather a compiled list of some of the top clinical research organizations around the world. We have highlighted their strengths and core service offerings to make it easier for you to find the right fit clinical research partner.
In addition, we’ve put together a list of 9 fundamental questions to ask the prospective clinical research organization , which will help you to save time and ensure a right fit in picking the CRO.
Formerly known as Quintiles and IMS Health, IQVIA is one of the largest CROs in the world, with a large range of service offerings to help advance clinical research.
The company was founded in North Carolina in 1982, and has since grown to over 88,000 employees in more than 100 countries.
Some clinical trial solutions offered by IQVIA include:
- Assistance with protocol design
- Design of phase 1 clinical trials
- Assessment and improvement of phase 2 and 3 clinical trials
- Site identification & selection
- Patient recruitment
- Access to global laboratories via their wholly owned subsidiary Q2 Solutions
Parexel is a global clinical research organization that was founded in 1982, and specializes in conducting clinical studies on behalf of its pharmaceutical partners in order to accelerate and ensure the drug approval process of up-and-coming potential treatments. It currently operates in more than 50 countries, and is run by more than 18,000 employees around the world.
The company has a wide range of service offerings, covering nearly every type of clinical trial service to assist sponsors in running successful clinical studies.
Some clinical trial solutions offered by Parexel include:
- Clinical trial design and development for early phase, phase 2 & 3, and late phase clinical trials
- Clinical data management
- Decentralized clinical trials
- Clinical supply chain management
- Medical writing
- Regulatory affairs consulting
- Pharmacovigilance
3. PRA Health Sciences
PRA Health Sciences is one of the largest contract research organizations in the world. Founded in 1976 under the name “Anti-Inflammatory Drug Study Group”, the company was renamed to PRA in 1982. PRA Health Sciences employees more than 17,000 people, and provides coverage to more than 90 countries.
In 2021, PRA Health Sciences was acquired by the Ireland-headquartered global CRO leader ICON, which is also reviewed in this list.
Some clinical trial solutions offered by PRA Health Sciences include:
- Decentralized Clinical Trials Platform
- Protocol Consultation & Study Design
- Onsite Support services
- Customized Solutions for Biotech (such as asset valuation, regulatory strategy, engagement and support, drug development strategy and funding solutions)
- Clinical Diagnostics
- Site Commercial Solutions
- PRA’s Laboratories for Drug Development
Headquartered in Ireland, ICON was founded in 1990 in Dublin by co-founders John Climax and Ronan Lambre. The company has since grown to be one of the largest CROs in the world. As of September 2020, the company employs more than 15,000 people in 94 locations and across 40 countries.
ICON offers clinical research services which include consulting, clinical development and commercialization across a wide range of therapeutic areas.
In 2021, ICON acquired PRA Health Sciences, which is another CRO and global leader in clinical research services.
Some clinical trial solutions offered by ICON:
- Commercial Positioning
- Early Phase
- Functional Services Provision
- Laboratories
- Language Services
- Medical Imaging
- Real World Intelligence
- Site & Patient Solutions
- COVID-19 Clinical Operations
5. Syneos Health
Formerly known as InVentiv Health Incorporated and INC Research, Syneos Health is a publicly listed and global contract research organization. The company is based in Morrisville, North Carolina, and specializes in assisting companies with late-stage clinical trials. Syneos Health currently employs more than 25,000 people, and has offices across 91 locations.
In early 2018, INC Research was acquired inVentiv Health, and the merged company was named Syneos Health.
Some clinical trial solutions offered by Syneos Health include:
- Decentralized Clinical Trials Solutions
- Bioanalytical Solutions
- Phase II-III/Phase IIIb-IIIV
- Medical Device Diagnostics
- Clinical Data Management
- Clinical Project Management
- Clinical Monitoring
- Drug Safety & Pharmacovigilance
- Site and Patient Access
6. Labcorp Drug Development (Formerly Covance)
Formerly known as Covance and renamed to Labcorp Drug Development in early 2021, this CRO is one of the largest contract research organizations in the world. The company claims to provide the world’s largest central laboratory network, and has been rated as one of the best places to work for LGBTQ+ equality by the Human Rights Campaign organization in 2018 to 2021. Currently, Labcorp employs over 70,000 people and is able to support clinical research efforts in almost 100 countries around the world.
Some clinical trial solutions offered by Labcorp Drug Development include:
- Preclinical Services
- Clinical Trials
- Clinical Trial Laboratory Services
- Post-Marketing Solutions
- Medical Devices
- Data & Technology
Also known as Pharmaceutical Product Development, PPD is a large global contract research organization headquartered in Wilmington, North Carolina. Started as a one-person consulting firm in 1985, PPD has grown to over 27,000 employees worldwide, and provides a wide range of clinical research services to pharmaceutical and biotech companies.
Some clinical trial solutions offered by PPD include:
- Clinical Development
- Early Development
- Peri- and Post-Approval
- PPD Biotech
- PPD Laboratories
- Product Development and Consulting
- Site and Patient Centric Solutions
8. Fisher Clinical Services
Part of Thermo Fisher Scientific, Fisher Clinical Services is a global clinical research organization with headquarters in Center Valley, Philadelphia.
The company has been in the business of clinical supply chain management for over 20 years, and is focused exclusively on working with the packaging and distribution requirements of clinical trials across the globe.
Some clinical trial solutions offered by Fisher Clinical Services include:
- Biologistics Management
- Cell & Gene Therapy
- Clinical Ancillary Management
- Clinical Label Services
- Clinical Trial Packaging & Storage
- Clinical Supply Optimization Services
- Cold Chain Management & Expertise
- Direct-to-Patient
- Distribution & Logistics
- Strategic Comparator Sourcing
- Public Health Research
Established in 1997 under the name Kiecana Clinical Research, KCR is a full-service contract research organization that provides a variety of services for clinical monitoring, safety & pharmacovigilance, clinical project management, quality assurance and regulatory affairs.
KCR operates globally, and has offices in North America, Western Europe, Central Europe and Eartern Europe. The company currently employs more than 700 staff.
Some clinical trial solutions offered by KCR include:
- Trial Execution
10. Medpace
Founded in 1992 and based in Cincinnati, Ohio, Medpace is a midsize clinical contract research organization. The company has operations in over 45 countries, and employs over 2,800 people. Medpace provides support services for Phase I-IV clinical trials for pharmaceutical and biotechnology companies, which include central laboratory services and regulatory services.
Some clinical trial solutions offered by Medpace include:
- Biostatistics and Data Sciences
- Clinical Trial Management
- Drug Safety and Pharmacovigilance
- Medical Writing
- Quality Assurance
- Regulatory Affairs
- Risk-Based Monitoring
- Medpace Laboratories
11. Clintec
Now in business for over 22 years, Clintec is a medium-sized global contract research organization for pharmaceutical, biotech and medical device industries, with large expertise in oncology and rare diseases.
The company provides the flexibility and agility of a smaller-sized CRO, while also having a wide global coverage that large CRO companies are known for. Clintec is based in more than 50 countries, and was acquired by the leading global CRO IQVIA in late 2018.
Some clinical trial solutions offered by Clintec include:
- Project Management
- Data Management
- Biostatistics
- Global Feasibilities
- Patient Recruitment & Retention
12. Worldwide Clinical Trials
Bringing over 30 years of experience to the clinical research market, Worldwide Clinical Trials is a leading medium-sized global contract research organization. Founded by physicians with a dedication and commitment to advancing medical research, Worldwide Clinical Trials was the first customer-centric CRO.
Currently the company has coverage in more than 60 countries, and has extensive experience in a wide range of therapeutic areas, including central nervous system, metabolic, cardiovascular, oncology, rare diseases and general medicine.
Some clinical trial solutions offered by Worldwide Clinical Trials include:
- Bioanalytical Lab
- Early Phase Development
- Clinical Phase IIB-II Clinical Trials
- Phase IIIB-IV Clinical Trials
- Trial Management Technologies
Named #1 CRO in the world for operational excellence at the 2021 CRO Leadership Awards, CTI Clinical Trial And Consulting Services is a medium-sized global contract research organization that has been serving pharmaceutical companies since 1999.
Based in Covington, Kentucky, CTI has offices around the world in more than 60 countries, with coverage in North America, Europe, Latin America, Middle-East, Africa, and Asia-Pacific regions.
Some clinical trial solutions offered by CTI include:
- Feasibility
- Regulatory Affairs Study Start-Up
- Medical Monitoring
- Safety & Pharmacovigilance
- Clinical Services
14. Wuxi AppTec
Founded in 2000 as WuXi PharmaTech in the city of Wuxi, China, Wuxi AppTec has grown from a single laboratory into a leading global contract research organization with more than 28,000 employees, including 23,000 scientists and more than 30 research & development and manufacturing sites around the world.
With offices in Asia, U.S, Europe and the Middle East, the company is able to provide coverage to more than 30 countries around the world.
Some clinical trial solutions offered by Wuxi AppTec include:
- Small Molecule Drug R&D and Manufacturing
- Cell Therapy and Gene Therapy
- Drug R&D and Medical Device Testing
- Clinical Services (Phase I-IV)
15. Advanced Clinical
Founded in 1994 and based out of Deerfield, Illinois, Advanced Clinical is a midsize and full-service CRO that helps sponsors with running clinical trials. The company employs more than 700 staff, and offers a wide variety of services across many therapeutic areas. Advanced Clinical has global representation in over 50 countries around the world.
Some clinical trial solutions offered by Advanced Clinical include:
- eTMF & Document Management
- Global Medical Services
- Quality & Validation
16. Pharm-Olam
Pharm-Olam is a leading midsize CRO with global headquarters located in Houston, Texas and its European headquarters in Bracknell, United Kingdom. The company employs more than 800 staff, and has 25 offices around the world, with a global coverage in more than 60 countries.
The company has therapeutic expertise in 5 areas, including Rare & Orphan Disease, Infectious Disease & Vaccine, Oncology-Hematology, Allergy and Autoimmune.
Some clinical trial solutions offered by Pharm-Olam include:
- Study Feasibility
- Site Activation
- Patient Recruitment
- Medical Affairs
- Compliance & Training
- Clinical Monitoring & Operations
17. Clinipace
Founded in 2003 and based out of Morrisville, North Carolina, Clinipace is a global midsize full-service CRO with a focus on solution customization for clinical trials. The company has a large global coverage in more than 50 countries, and has offices in North America, South America, Europe and Asia-Pacific regions.
Clinipace’s therapeutic focus areas include Oncology, Nephrology and Urology, Rare Disease, Gastroenterology and Women’s Health. The company also has complete therapeutic expertise in Infectious Disease & Vaccines, Cardiology, CNS, Immunology, and Respiratory.
Some clinical trial solutions offered by Clinipace include:
- Clinical Analytics
- Clinical Technology and Ecosystem
- Functional Service Partnership (FSP)
- Regulatory & Strategic Product Development
9 Fundamental Questions To Ask A Top CRO Company Before Signing The Contract
1. which services does the cro provide.
CROs offload a lot of operational tasks from trial sponsors, which can touch any component of clinical trial operations. From formulating an overall study strategy and implementing technologies to support the operational processes of the trial, to picking and identifying sites, and supporting patients during the trial, the range of clinical services offered by a CRO tends to be vast and inclusive of all the typical services and support you will require for running a successful clinical trial.
However, not all CROs are the same in their service offerings, or are able to offer the same depth of capability within a seemingly same clinical trial support process. For this reason it is important to understand exactly which kind of clinical services and support you are looking to receive from the prospective CRO when running your clinical trial.
While services such as clinical monitoring and clinical trial management are offered by the majority of CROs, the specific needs of each trial are unique, and for this reason it is important to first identify what will be the unique services your trial requires. Completing this internal analysis first will help you to understand the extent to which a potential CRO partner will be able to provide all of these services.
Some CROs specialize in specific clinical trial functions which the company may label as a “core services”, in which case this is a sign the company will have more expertise, experience, and will be set up in a way to maximize their capabilities in providing support for these services compared to other services that the CRO offers.
For example, a CRO may include patient recruitment as part of its “core services”, which implies that they are highly skilled in and have the necessary infrastructure to design and implement a high-quality patient recruitment strategy.
Clara Health CRO Support Services: At Clara Health our specialty services include technology-augmented digital and patient advocacy recruitment, as well as patient support via our signature patient recruitment platform, which we use to upgrade clinical trials and deliver results sponsors look for in their recruitment and retention campaigns.
At Clara, we work alongside CROs to supplement and support clinical trials with modern and personalized capabilities that CROs do not typically have the bandwidth, corporate structure or infrastructure to support.
If you would like to learn more about exactly how our platform can upgrade your unique trial, feel free to book a Free 30 Minute Consultation Session Here with one of our in-house experts.
2. What Related Experience Does The CRO Have?
It is helpful to ask the prospective CRO company if they have any relevant experience in running clinical trials that would be an asset in designing and running your study. Previous experience in a related therapeutic area or in running a trial with a similar design allows CROs to have a deeper understanding into potential opportunities and challenges, increasing the likelihood of your clinical study being successful.
For example, if a sponsor is planning to run a trial in oncology, for the purpose of site identification and selection it would be valuable to partner with a CRO vendor that has expertise in this area, as they likely already have a good understanding of which sites will lead to optimal results.
However, it is also important to consider all factors when selecting a CRO vendor and not to rely on therapeutic experience as the sole qualifier for whether or not a potential CRO is a fit for your trial. While previous experience is beneficial, some sponsors close themselves off from working with vendors that have not worked in their therapeutic area, which significantly limits options when choosing a CRO partner that is truly a good fit for their clinical study.
This can impact the end result of your clinical study, as sponsors that are not successful in choosing a CRO vendor that is the right overall fit may face difficulties if the needs of their clinical study aren’t being properly met.
Clara Health: We have worked to provide support for clinical trials across a wide range of therapeutic areas and trial designs. Our specialty is filling in the gaps that CROs traditionally did not have to think about, which include digital patient recruitment, patient advocacy recruitment, and technology-augmented patient support.
Additionally, we are constantly building our proprietary data and running tests in a variety of therapeutic areas. These research efforts allow us to have a detailed understanding of the expected level of difficulty when recruiting particular patient populations, as well as allow us to predict with accuracy which segments of the targeted population will be likely to qualify in a particular study.
3. What Are The Communication Workflows & Expectations For Performing And Delivering Contracted Services?
It is important that you clarify what the expectations for communication will be between your prospective CRO vendor and your internal teams, as you will most likely be working with the CRO of your choice for the entire duration of your clinical trial.
There are a vast variety of factors and success determinants for a clinical trial, which are continuously undergoing change as the study unfolds. For this reason, it is recommended that you work with a CRO that is proactive in their communication, so that you are kept up to date with information about important changes as your clinical trial progresses.
A vendor that is proactive rather than reactive in their communication and approach to dealing with arising issues is one of the most important qualities in CRO. Challenging situations will naturally arise, and the promptness with which they are taken care of will significantly impact your clinical trial’s degree of success. Therefore, seeking a vendor that is able to match the standard of communication that you as a sponsor would like to experience throughout the duration of your partnership is one of the most critical steps in determining which CRO is the right fit for your clinical trial.
We’ve included a few additional questions pertaining to the communication structure and reporting expectations that you can ask a prospective CRO vendor to determine the degree of fit in this particular category:
Communication Expectations:
- If we were to move forward with you, which of your team members will be our main point of contact?
- How available will you be outside of the scheduled meetings to address any of our concerns or additional requests?
- What will be the frequency at which update meetings will be conducted, and who will be present at those meetings?
- Which clinical study processes will be reported on, and what will be the workflow for how we will receive this information?
- What will be the cadence at which we will receive progress reports?
- Would we be able to access metrics electronically via an interactive dashboard, or will you send us formal reports?
Clara Health: At Clara Health, we directly interact and actively work with several key stakeholders involved in running a clinical trial, which includes sponsors, CROs, sites, and patients. This unique position allows us to have a centralized perspective which helps us to see all the moving parts of a clinical trial at the same time, which helps to identify issues and relay this vital information and insight back to the sponsor (or other appropriate stakeholders) in the shortest time possible.
The ability to access this perspective allows us to gather the most accurate, complete, and up-to-date information about how the clinical trial is unfolding, and quickly becomes very valuable to sponsors for their clinical trial.
As an example, we may receive feedback from patients about having an unsatisfactory experience with a particular study site. We are able to aggregate and analyze this information, and relay our findings back to the sponsor and the study site to improve the experience for other patients.
4. What Is The CRO’s Client Satisfaction Record?
It is a good practice to request information or metrics from the prospective CRO vendor that can point to the degree of satisfaction of their past clients. Prior to signing the contract, vendors will naturally do their best to uplift their image and future value to you during their sales conversations with you and your team. It can be tricky to get an objective understanding of what the partnership experience will actually entail, especially when there are multiple vendors fighting for your commitment.
We recommend that you ask the prospective vendor to provide success metrics regarding areas of clinical trial operations that are going to be important for your trial.
For example, you may be interested in learning about the vendor’s relationship to finances, in which case it will be useful to ask them about situations in which they went over the planned budget, and investigate into the reasons behind that. Alternatively you may be concerned about potential delays in timelines, in which case it would be helpful to learn about metrics regarding the CRO’s ability to meet timeline expectations.
You may also request to talk to the prospective CRO’s past clients, which will help you to gain insight into what the relationship was like and give you the opportunity to examine if the way in which the particular CRO manages its relationships and performs its services meets the expectations that you would have for your potential relationship and for your clinical trial.
Clara Health: At Clara Health, our relationships with our partners and with our patients are most important to us. In the unique position where we fit in the clinical trial process, we have the opportunity to directly co-create the clinical trial patient experience with a variety of stakeholders, including sponsors, sites, CROs, and patients.
Our company’s values and culture have been directed and developed to be such that the client and patient experience is at the top of priority for all of our internal teams, and we work to provide the best quality of care to all stakeholders.
We have many testimonials from every type of partner we’ve worked with which we can happily share with you.
5. How Do You Adapt When Encountering Challenges With Running A Clinical Trial?
It is inevitable that challenges and unforeseen changes will arise throughout the operational clinical trial process, and for this reason it is important to work with a CRO vendor that can provide you with evidence of their flexibility and ability to adapt to sudden changes.
The ideal CRO partner is one that is highly consultative throughout the entire process, and has an ability and the initiative to deal with challenges at their seed stage, prior to them turning into major obstacles for the success of your trial.
CROs naturally have a large reach, and there are a lot of different clinical trial mechanisms and processes that are under their control. They are able to monitor and respond to what is going on in every key link in the chain of the clinical trial operation.
It is reasonable to expect this level of oversight from a CRO, and additional questions that can help you gain insight into this include:
- What are some examples where the CRO was effective at monitoring the health of clinical trials they’ve helped operate in the past?
- How quickly does the CRO respond to challenges or opportunities for improving the clinical trial experience?
- How well does the CRO gather & process information from study sites, study teams, patients & the sponsor, and what are their typical data analysis workflows?
It is also recommended to speak to the prospective CROs past clients to help you gain insight into how well they respond and adapt to the naturally arising challenges in clinical trials.
Clara Health: While CROs do have a large reach within the clinical trial, no CRO has complete visibility into every clinical process. They are not typically set up to support full visibility, which can manifest as a potential threat to your clinical trial as it unfolds. This is especially true for parts of the clinical trial processes that CROs naturally do not specialize and often subcontract, such as clinical trial recruitment.
At Clara, we are in a unique position in relation to other key partners involved in operating the clinical trial. We are in direct and frequent contact with patients, CROs, study sites, study teams, and the sponsor, and have a very deep understanding of the patient pipeline. This allows us the unique ability to go very deep into specific parts of the recruitment chain and investigate what is working and what is not working.
In addition, Clara functions as a resource for all partners in the clinical trial. For example, we work directly with site teams to ensure that they have access to a 3rd party that they can relay their needs to and receive fast support in case there is anything they require that can improve the patient recruitment process.
6. Which Parts Of Operating The Clinical Trial Will You Be Outsourcing?
Since there are so many processes and mechanisms that go into operating a clinical trial, CROs will always outsource some parts of running and managing the study. While you can expect that the prospective CRO will subcontract some of the work, it is important to find out which exact parts the clinical study will be outsourced.
There are certain basic and key clinical processes (such as site selection) that CROs almost always help with, and if you find that these parts of your trial are going to be subcontracted to another company, it is recommended to find out why the CROs operations are set up this way and how this would impact the service you will receive.
Ultimately what matters to you as a partner and client is that the quality of service and care that you will receive will be up to standard, and meet what was promised and what you are expecting. While this trust is important after you have signed the contract, it is recommended that prior to entering into such a significant commitment that you have evidence and the conviction that the CRO of your choice is truly the right fit and will deliver the quality of service that was being discussed.
Since it is impossible to predict exactly what the quality of this relationship and services performed will actually be like in practice, it is recommended that you understand the details of what will be done for your trial and how. Investigating how the CRO outsources and subcontracts services for a clinical trial will help you to gain necessary insight that you would need to make the correct vendor selection decision.
Clara Health: At Clara, we maximize the effectiveness of the digital component across the entire digital & recruitment spectrum, which is added on top of the existing capabilities of the CROs and other vendors involved in operating your clinical trial. In addition, we offer services that augment the CROs efforts, which has the potential to significantly improve the patient experience, operations flows, recruitment and retention performance, which is so important in ensuring the success of a clinical trial.
For example, if a CRO wants to have a great site relationship, we are able to come in as a third party on behalf of the sponsor and CRO and act as a resource and additional support for sites.
In another example, If a sponsor wants to have great relationships with the patient community, Clara is able to come in on behalf of the sponsor and develop these relationships while being perceived more neutrally by the patient community.
7. Do You Have Experience Running International Trials?
If you are planning on operating an international clinical trial, it is recommended to work with a CRO that has extensive experience in this area. While many CROs will offer near-global coverage, the level of experience with specific geographic locations can significantly vary from one vendor to another.
It is important to work with a CRO that has experience running clinical trials in the specific countries and regions you are planning to conduct your research in. Being compliant with the local rules and regulations for clinical testing is a very complex process that requires existing understanding and familiarity in order to ensure logistical smoothness and to mitigate legal risks. In operating a clinical trial, there are a multitude of clinical services and processes, which can greatly vary across the many regions in which you can conduct clinical testing.
A CRO that is lacking experience in operating international trials or operating in particular regions where you plan on conducting research may not be able to meet your desired quality and agility expectations, and therefore may not be the right fit for your international clinical trial.
Clara Health: In the past, we have provided international patient recruitment and digitally-augmented trial support services for clinical trials in the EU, Canada, UK, Australia and South America.
Clara Health is fully compliant to operate international studies everywhere in the world, with the exception of Russia and China.
8. What Is Your Relationship With Patients?
Patient-centric approach to designing and operating a clinical trial is becoming more and more crucial in the clinical research space. The ability of a sponsor and their CRO partner to understand the needs and characteristics of their target patient community is a significant determinant of whether or not the study will be a success.
A sponsor that has close and authentic relationships with the patient community tends to have a deeper understanding of how to create the best clinical trial experience that will attract patients and keep their interest throughout the clinical trial.
In addition, strong relationships with patients allow sponsors and CROs to forecast recruitment and patient retention pipeline with much higher accuracy. This ability is critical for ensuring the success of the trial and mitigating the risk of low enrollment. After an understanding of the patient population is acquired, sponsors gain the necessary insight to design a clinical trial that is not only favorable to their research results, but is also practical and will result in the enrollment numbers they are looking for.
While many CROs have already recognized the importance of patient-centricity and evolved the ways in which they design and operate clinical trials, other CROs have not yet made such a pivot in their values. It is important to understand the degree of importance the prospective CRO places on creating a favorable patient experience, and what kind of infrastructure the company has to support it.
At Clara, we recommend choosing a CRO partner that is adapting to the patient-centric model which is becoming more and more important for running a successful clinical trial.
Clara Health: Since early stages of our development, we’ve had a dedicated patient advocacy team that has been integral in shaping our company’s vision and operations. We have built our entire platform and recruitment infrastructure around creating the best experience for patients. Our teams, corporate values, service offerings and company infrastructure all work in the service of the patient.
In addition, over the many years of being in business we have heavily invested in building authentic patient community relationships that span across a variety of therapeutic areas. This has given us a unique ability to receive feedback directly from patients that is genuine and authentic around marketing materials, strategy for patient recruitment, and other services that we build for specific trials.
This ability to build partnerships with the patient community in an authentic way gives us a very unique ability to engage with the patient community on behalf of a pharmaceutical company, allowing our sponsor & CRO partners the opportunity to start conversations with patients through our in-house patient advocacy team.
If you would like to learn how Clara can help you to build a strong & authentic relationship with your target patient community, get in touch with us and we’d be happy to share our capabilities and previous results with you as they relate to your current or upcoming clinical trial.
9. How Is The CRO Going To Utilize Patient Input For Developing The Trial?
In the initial stages of clinical trial design, sponsors often determine the ideal patient profiles that would help them to drive the most favorable research outcomes for their study. While it is important for the success of your trial to determine who your ideal patients are, very often these projections do not match up with what is viable in practice.
At Clara, we often encounter study protocols that are not set up realistically for successful recruitment to be possible.
Common mistakes that are made when determining trial eligibility criteria and trial design include:
- Overestimating the interest in the clinical trial from the target patient population
- A lack of patient focus in the trial design
- A lack of convenience for patients in their participation
- Complicated and/or inefficient study experience flows
- Crafting the eligibility criteria around the patient population that is most likely to lead to favorable study outcomes, without conducting sufficient research to more accurately estimate the recruitment and retention difficulty of the group for a particular study
It is natural for there to be a “push & pull” between the research ideal and the real world practicality. It is important to determine the correct balance between these two sides for your trial, as going too far in either direction will decrease the chance of your clinical study’s success.
The nature of the industry as it is right now is such that there is excess research idealization and not enough emphasis on patient centricity. This distorted orientation has resulted in many clinical trials being unsuccessful, negatively impacting sponsors, patients and the entire clinical trials industry.
The ideal CRO partner should help you make sure that your protocol design sets your study up for success. The CRO should be able to help you determine the proper balance between the research ideal and the real world practicality, and back up their findings with sufficient research and patient data that can project your trial being a success.
Clara Health: When formulating a recruitment and retention plan for our clients, we begin with conducting thorough research into the target trial patient population. This allows us to get a clear understanding of which recruitment channels will yield the best results and what kind of marketing materials will resonate with the prospective study participants.
To ensure accuracy and real-world applicability of our research, we consult and collaborate with our internal patient advocacy and patient support teams, as well as with our clients and patients representing the target trial patient profiles. We then tie our findings back with any existing proprietary data that we have in connection with the therapeutic area or the prospective target patient group.
Our unique position within the clinical recruitment chain gives us the presence and deep-rooted access needed to effectively tap into any of the three patient traffic sources: digital recruitment, offline recruitment, or patient advocacy recruitment.
Once a recruitment campaign has gone live, we constantly monitor, analyze and optimize our performance to make sure that the processes we have in place are as efficient as possible and drive the greatest results. In addition, we have the capability to layer in any traditional advertising (such as billboard ads) if requested by the study sponsor.
1-888-664-9690
[email protected]
- Corporate Brochure
- Case Studies
- Testimonials
- Press Release
- Vascular Surgeons Email List
- Pathologists Email List
- Gastroenterologist Email List
- Chiropractors Email List
- Anesthesiologists Email List
- Plastic Surgeons Email List
- Pediatricians Email List
- Geriatricians Email List
- General Surgeons Email List
- Diabetes Specialist Email List
- Allergist / Immunologist Email List
- Arthritis Specialists Email List
- Cardiologist Email Lists
- Dermatologist Email List
- Neurologists Email List
- Orthopedic Surgeons Email List
- Urologists Email List
- Podiatrist Email List
- Dermatopathologist Email List
- Hematologists Email List
- Pulmonologists Email List
- Nephrologists Email List
- Oncologist Email List
- Rheumatologist Email List
- Radiologist Email List
- Optometrist Email List
- Ophthalmologist Email List
- Acupuncturist Email List
- Optician Email List
- Neonatologist Email List
- Neuropathologist Email List
- Hepatologist Email List
- Traumatologist Email List
- Gynecologists Email List
- Psychiatrists Email List
- Endocrinologist Email List
- Hygienists Email List
- Primary Care Physicians Email List
- General Practitioners Email List
- Osteopathic Physicians Email List
- Emergency Physicians Email List
- Bariatric Physician Email List
- Family Medicine Specialist Email List
- Epidemiologist Email List
- Clinical Lipidologist Email List
- Registered Dieticians Email List
- Psychologists Email List
- Neurosurgeon Email List
- Nutritionists Email List
- Audiologist Email List
- Hypnotists Email List
- ENT Specialists Email List
- Sleep Medicine Specialists Email List
- Sports Medicine Physician Email List
- Physiatrists Email List
- Physician Assistants (PAs) Email List
- Athletic Trainers (ATs) Email List
- Cosmetologists Email List
- Pediatric Allergists Email List
- Naturopathic Doctors Email List
- Veterinarian Email Lists
- Phlebotomists Email List
- Pediatric Radiologists Email List
- AMA Physicians Email List
- Paramedics and EMTs Email List
- Pediatric Neurologist Email List
- Preventive Medicine Specialist Email List
- Reproductive Endocrinologist Email List
- Interventional Cardiologists Email List
- Nuclear Medicine Specialist Email List
- Infectious Disease Specialist Email List
- Radiation Oncologists Email List
- Internal Medicine Specialist Email List
- Mental Health Specialists/Professionals Email List
- Addiction Medicine Specialist Email List
- Aerospace Medicine Physicians Email List
- Hematology & Oncology Specialist Email List
- Medical Geneticist Email List
- Addiction Psychiatrist Email List
- Physical Medicine Rehabilitation Email List
- Cardiac Care Nurses Email List
- Critical Care Nurses Email List
- Emergency Nurses Email List
- Geriatric Nurses Email List
- Home Healthcare Nurses Email List
- Military Nurses Email List
- Nurse Anesthetists Email List
- Nurse Practitioners Email List
- Registered Nurses Email List and Mailing List
- Neurology Nurses Email List
- Orthopedic Nurses Email List
- Occupational Health Nurses Email List
- Pediatric Nurse Practitioners Email List
- Dermatology Nurses Email List
- Perioperative Nurses Email List
- Midwife Nurses Email List
- Family Nurse Practitioners (FNP) Email List
- Public Health Nurses (PHNs) Email List
- Endocrinology Nurses Email List
- Ambulatory Care Nurses Email List
- Women’s Health Nurse Practitioners Email List
- Adolescent Medicine Nurses Email List
- Reproductive Health Nurses Email List
- Transplant Nurses Email List
- Dialysis Nurses Email List
- Allergy / Immunology Nurses Email List
- Family Planning Nurses Email List
- ENT Nurse Practitioners Email List
- HIV/AIDS Nurses Email List
- Community Health Nurses Email List
- School Nurses Email List
- Student Nurses Email List
- Nurse Manager Email List
- Oncology Nurses Email List
- Child Psychiatric Nurses Email List
- IV Certification Nurses Email List
- Licensed Practical Nurses Email List
- Licensed Vocational Nurses Email List
- Clinical Nurse Specialist Email List
- Acute Care Nurse Practitioners Email List
- Office Based Nurses Email List
- Certified Registered Nurse Anesthetist Email List
- Certified Nursing Assistants Email List
- Psychiatric-Mental Health Nurse Practitioners Email List
- Adult Care Nurse Practitioner Email List
- Registered Pharmacist Email List
- Hospital Druggist Email List
- Druggist Email List
- Community Pharmacists Email List
- Pharmacist Clinicians (PhC) Email List
- Ambulatory Care Pharmacists Email List
- Pharmaceutical Email & Mailing List
- Prosthodontists Email List
- Periodontist Email List
- Endodontists Email List
- Pediatric Dentists Email List
- Orthodontist Email List
- Oral Pathologists Email List
- Denturist Email List
- ADA Dentists Email List
- Dental Hygienist Specialists Email List
- Oral and Maxillofacial Radiologists Email List
- Oral and Maxillofacial Surgeons Email List
- Subacute Care Centers Email List
- Cancer Centers and Services Email List
- Ambulatory Surgery Centers Email List
- Diagnostic Imaging Centers Email List
- Urgent Care Centers Email List
- Trauma Centers Email List
- Nursing Homes Email List
- Nursing Home Administrators Email List
- Hospital Email List
- Assisted Living Facilities Email List
- Medical Institutions Email List
- X-Ray Laboratories Email List
- Primary Care Centers Email List
- Dental Clinics Email List
- Blood Bank Centers Email List
- Dental Laboratories Email List
- Clinics Email List
- Physical Fitness Facilities Email List
- Med Spa Email List
- Dental Equipment / Supplies Manufacturers Email List
- Biomedical Equipment Manufacturers Email List
- Electro-Medical Equipment Manufacturers Email List
- X-Ray Apparatus and Tubes Manufacturers Email List
- Ophthalmic Goods Manufacturers Email List
- Surgical Appliances Manufacturers Email List
- Surgical / Medical Instruments Manufacturers Email List
- Neuromodulation Devices Manufacturers Email List
- Cardiovascular Devices Manufacturers Email List
- Therapeutic Devices Manufacturers Email List
- Spine Devices Manufacturers Email List
- Laboratory Equipment and Supplies Email List
- Hospital Equipment and Supplies Email List
The Top 10 Clinical Research Companies in the USA
- Dec 02, 2022
- Posted By: admin

To bring effective medicines and Innovations in healthcare industry , a Clinical Research Organization or Contract Research Organization (CRO) are a must for the Biotech, Medtech, and pharma industries. A CRO offers comprehensive support to their efforts to test, examine, refine and market the latest medicines and devices that help in improving Healthcare Industry.
In 2022, the global clinical research market stands at $63.6 billion , and by 2027, it is expected to reach around an exciting amount of 115.1 billion with a CAGR of 11.0% from 2022 to 2027.
This growth is powered by an increase in R&D activities and expenditures and a significant rise in clinical trials owing to the need for innovations in healthcare. For that reason, it is also essential to be aware of the most prominent global research organizations that offer vital support with their specialized clinical trial solutions. This post will walk you through the ten big Clinical Research Companies In USA . So, let’s begin:

- Founded: 1982
- Headquarters: Durham, North Carolina
- Annual Revenue : $13.87B
- Employee Size: 88,000
IQVIA is one of the largest Clinical Research companies in the world that works profusely to enhance healthcare by integrating the study of human science with discoveries in technology and data science to improve understanding of human health and provide better and quicker care.
It helps pharma companies, along with other medical organizations, to innovate and maximize positive outcomes. In addition to clinical research and development, IQVIA has also developed analytics and technology solutions to aid the medical sector in commercializing products and enhancing customer engagement.
Some clinical trial solutions given by IQVIA are as follows:
- Site identification & Selection
- Help with protocol design
- Design of clinical trial phase -1
- Investigation and improvements of the clinical trial in phases 2 and 3
- Accessibility to global laboratories
2. Pharmaceutical Product Development (PPD)

- Founded: 1985
- Headquarters: Wilmington, North Carolina
- Annual Revenue: $65.0M
- Employee Size: 24,000
PPD, also known as Pharmaceutical Product Development, is an extensive research organization headquartered in the United States but has a widespread global presence.
The company directs its attention to three prime areas: pharma development, lifecycle management services, and laboratory. Besides, it has clients and partners covering various sectors, including pharmaceutical companies, biotech firms, medical device manufacturers, and government or academic organizations.
Some clinical trials offered by PPD include:
- Early Development
- Consulting and Product Development
- PPD Biotech
- The site and patient-centered Solutions
- Clinical Development

- Headquarters: Waltham, Massachusetts
- Annual Revenue: $3B
- Employee Size: 18,000
Parexel is another important name in the list of Clinical Research Companies In USA that specializes on behalf of its pharmaceutical partners in carrying out clinical studies to boost and make certain that the medicine approval process takes place smoothly. It has a wide range of offerings that spreads across every type of clinical trial service to help sponsors work on creating successful trials.
Some of the clinical trial solutions which Paraxel offers:
- Management of clinical data
- Medical writing
- Regulatory affairs consulting
- Design and development of clinical trials at all phases
- Clinical supply chain management
With pride, the company operates in over 50 countries and owns over 95% of the top 200 selling biopharmaceuticals in today’s market.
4. PRA Health Services

- Founded: 1976
- Headquarters: Raleigh, North Carolina
- Annual Revenue: $64.3M
- Employee Size: 16,400
PRA Health Sciences is a contract research organization that provides coverage in more than 90 countries worldwide. It principally centers on contributing therapeutic and operational expertise through integrated applications and supplying local expertise in particular areas. Besides, it works in the early and end stages of clinical trial procedures and the domains of strategy, consultancy, technology, and bio-analytics.
Moreover, it works hard to speed up medicine development operations to introduce better and more effective medicine sooner in the market.
Let’s take a peek at the PRA Health Sciences clinical trial solutions:
- Onsite support services
- Personalized solutions for Biotech
- Clinical Diagnostics
- Site Commercial Solutions
- Protocol Consultation and Study Design
- PRA’s Laboratories for Medicine Development
5. Syneos Heath

- Founded: 1999
- Headquarters: Morrisville, North Carolina
- Annual Revenue: $5,213M
- Employee Size: 28,000
With the merger of inVentiv Health and INC Research, Syneos Health was created that combines every discipline involved in bringing new therapies or products to market, from clinical research to consulting and commercialization.
Though it provides clinical development services spanning all stages, it primarily owns a specialization in helping healthcare organizations with the late stages of clinical trials. Moreover, its commercial solutions cover communication, consulting, and medication adherence.
The clinical trial solutions it offers include:
- Early phase
- Decentralized clinical trial solutions
- Clinical data management
- Pharmaceutical
- Medical Device Diagnostics

- Founded: 1978
- Headquarters: Burlington, North Carolina
- Annual Revenue: $16.1B
- Employee Size: 70,000
Laboratory Corporation of America, or LabCorp, focuses on offering extensive clinical laboratory solutions and end-to-end medication and diagnostic development and commercialization.
The company develops special testing operations such as HIV genotyping, oncology testing, phenotyping, clinical trials, and diagnostic genetics. Moreover, its services are spread through care organizations, hospitals, government agencies, pharmaceutical companies, and physicians.
Some clinical trial solutions provided by LabCorp are:
- Preclinical Services
- Post-Marketing Solutions
- Medical Devices
- Clinical Trial Laboratory Services
- Clinical Trials
- Data & Technology
7. Charles River Lab

- Founded: 1947
- Headquarters: Wilmington, Massachusetts
- Annual Revenue: $690.4M
- Employee Size: 20,000
Charles River laboratories proudly span its capabilities throughout the medical R&D process from basic research to pre-clinical stage testing, manufacturing and commercialization within two significant services: Good Laboratory Practice (GLP) and non-GLP. It offers precise support to help its partners advance their research and efforts to introduce effective medicines in the market.
The company further serves biotechnology and pharmaceutical companies, hospitals, government agencies, and academic institutions.
Some of the clinical development solutions it offers include:
- Bioanalysis
- Clinical Kitting services
- Data management
- Quality control
- Stability testing
8. Fisher Clinical Services

- Founded: 1989
- Headquarters: Center Valley, Philadelphia
- Annual Revenue: $17B
- Employee Size: 50,000
Fisher Clinical Services is a part of Thermo Fisher Scientific that has been for over 20 years in the business of supply chain management. It focuses exclusively on working with distribution and packaging requirements of clinical trials happening around the globe.
The company is committed to providing high-value products which adhere to the standard of pharmaceutical companies and is reliable, sustainable and perform well.
Some of the clinical trial solutions offered by Fisher Clinical Services are:
- Cell & Gene Therapy
- Clinical Label Services
- Clinical Supply Optimization services
- Direct to patient
- Biologistics management
9. Medpace Holdings

- Founded: 1992
- Headquarters: Cincinnati, Ohio
- Revenue: $1B
- Employee Size: 1,001-5,000
Medpace offers support services for phases one to five of clinical trials for biotechnology, pharmaceutical companies, and medical device industries. It is a scientifically-driven organization that offers full service and helps industries make key differences with their contributions to the healthcare sector.
Some of the clinical research solutions that you receive from Medpace are:
- Medical Writing
- Quality Assurance
- Clinical Monitoring
- Clinical Trial Management
- Risk-based Monitoring
- Regulatory Affairs
- Biostatistics and Data sciences
10. Advanced Clinical

- Founded: 1994
- Headquartered: Deerfield, Illinois
- Annual Revenue: $106M
- Employee Size: 501-1,000
A mid-size and full-service Clinical Research organization that helps sponsors run clinical trials and offers various solutions across therapeutic fields. It allows clients to secure better outcomes through candid discussions, conversations, foresight, and innovative solutions.
The company works hard to improve every life touched by clinical research. As such, some clinical solutions provided by Advanced Clinical are:
- Project management
- Clinical monitoring
- eTMF and document management
- Quality & Validation
The Bottom Line
There you have it – the best Clinical Research Companies In USA to get support from while working on new medicine or medical devices.
CROs play a critical role in the pharmaceutical, biotech, and medical device industries to manage and lead their clinical trials now more than ever due to rapidly rising prices, regulations, and tighter deadlines. Also pharmaceutical companies approaching Clinical Research Organizations for their R&D activities to stay competitive, flexible, and profitable against all odds.

- Try for free

Top 15 Clinical Research Companies: Leaders in Medical Innovation
What’s on this page:

In the healthcare industry, the number of contract research organizations in the US has reached 2,823 in 2023. This marks a subtle but significant increase of 0.9% compared to the previous year.
This increase signals a vital trend: the growing complexity of finding the best clinical research companies in a crowded field. These organizations aren’t just businesses; they’re important in advancing medicine and developing drugs and therapies.
With such an important task, choosing the right company becomes essential. In this guide, we’ve looked closely at many companies along with their strengths and weaknesses and made a list of the top clinical research organizations.
By the end, you’ll know which company is the best fit for your needs.
Quick List of Top 15 Clinical Research Companies
Here is a quick overview of the best companies of clinical research:
- IQVIA: Best for data-driven insights and advanced analytics in healthcare research.
- ICON: Best for comprehensive clinical development services and therapeutic expertise.
- Parexel: Best for global biopharmaceutical services, emphasizing regulatory and clinical trial excellence.
- Syneos Health: Best for integrated biopharmaceutical solutions and clinical-commercial capabilities.
- PPD: Best for drug development services with innovative, technology-enhanced trial strategies.
- Labcorp: Best for comprehensive clinical testing and diagnostics services with global reach.
- Medpace: Best for expertise in clinical research and regulatory affairs for pharmaceutical companies.
- Charles River Laboratories: Best for preclinical research and development services, including animal testing and research models.
- PRA Health Sciences: Best for clinical trial expertise and integrated solutions for biopharmaceutical development.
- AdvanCell: Best for innovative cell and tissue-based research solutions for life science industries.
- Dynata: Best for data-driven insights and market research services for informed decision-making.
- Covance: Best for end-to-end drug development solutions, from preclinical to post-marketing.
- MedNet: Best for technology solutions and eClinical platforms for streamlined clinical trials.
- Fisher Clinical Services Inc: Best for global logistics and supply chain services for clinical trial materials.
- Worldwide Clinical Trials: Best for specialized CRO offering personalized clinical research solutions.
3 Best Clinical Research Organizations: Comparison Chart
Here’s a comparison table to highlight the key features and differences among the best companies of clinical research. This table aims to provide a quick overview of each company’s unique strengths and areas of expertise in the pharmaceutical and healthcare research sector.
| Data-driven insights, advanced analytics | Healthcare research, Data analytics | Technology-driven healthcare research services | |
| Comprehensive clinical development, expertise | Clinical development, Therapeutic research | Clinical trial and development services | |
| Regulatory expertise, clinical trial excellence | Biopharmaceutical services | Global clinical trial management |
3 Top Clinical Research Organization List For Advanced Medical Discoveries

Now, we’ll explore the top clinical research organizations (CROs) dedicated to advancing medical discoveries. Let’s jump into the details of these exceptional organizations.
IQVIA is a global leader in clinical research and healthcare data analytics. They play a crucial role in the medical field by providing comprehensive data, advanced analytics, and expert insights. This helps pharmaceutical and healthcare companies make smarter, more effective decisions.
Why is IQVIA among the best? Their strength lies in their vast database and advanced technology, which enable them to analyze complex healthcare data efficiently. This leads to a better understanding of diseases, more effective treatments, and faster drug development.
IQVIA’s work is essential because it speeds up the process of bringing new medicines to the market, ultimately benefiting patients worldwide. In short, IQVIA is a key catalyst in advancing global healthcare.

About IQVIA
- Founding Team: Dennis Gillings
- Founding Year: 1982
- Company Size: 86,000
Features of IQVIA
IQVIA, a prominent player in the life sciences sector, is dedicated to advancing healthcare through connected intelligence. Here are some key features of IQVIA in the world of clinical research:

Innovative Clinical Development
IQVIA is reimagining clinical development by intelligently connecting data, technology, and analytics. This approach leads to faster decision-making and reduced risk, enabling the delivery of life-changing therapies more quickly.
Efficient Payment Systems for Clinical Trials
They have simplified the process of paying sites involved in clinical trials. IQVIA offers the capability to make payments within 30 days, even in challenging locations. This significantly reduces the administrative burden of managing clinical trial payments by up to 90%.
Decentralized Trials Expertise
The company has conducted over 500 studies in more than 75 countries, covering over 30 indications using decentralized trial methodologies. This demonstrates their capability in managing complex, multinational clinical trials.
Global Reach and Impact
With a presence in various regions, including Australia, New Zealand, the Middle East, and Africa, IQVIA’s global footprint allows it to drive healthcare innovations worldwide.
AI and Technology Integration
The company is at the forefront of integrating AI and other technologies in healthcare. Their Healthcare-grade AI promises precision, speed, scale, trust, and reliability, essential for advancing health and improving patient outcomes.
- Extensive, reliable healthcare data enhances market research quality.
- Utilizes AI and machine learning for advanced healthcare insights.
- Specialized focus yields a deep understanding of healthcare dynamics.
- Broad international presence enables diverse and large-scale studies.
- Offers advanced tools for insightful healthcare data analysis.
- Advanced tools can be challenging to use without training.
- Handling sensitive health data raises privacy and security issues.
Our Review of IQVIA
IQVIA, a prominent player in the healthcare and life sciences industry, presents a mixed bag of strengths and weaknesses. On the positive side, we appreciate IQVIA’s extensive expertise in data analytics and healthcare consulting.
Their comprehensive research and analysis have undoubtedly driven valuable insights and innovations in the sector. Moreover, their global presence allows for diverse perspectives and access to critical healthcare data.
However, we must also acknowledge some shortcomings. IQVIA’s services can be prohibitively expensive for smaller organizations, limiting accessibility. Additionally, the sheer volume of data can sometimes lead to information overload, making it challenging to extract actionable insights.
ICON is a prominent company in the field of clinical research, playing a significant role in advancing medical science. They specialize in designing and conducting clinical trials for new medicines and treatments.
The work of ICON is crucial because they help determine the safety and effectiveness of these potential medical breakthroughs. They are considered one of the best in clinical research due to their high standards of accuracy, reliability, and ethical practices.
ICON’s expertise ensures that the clinical trials they manage are conducted efficiently and effectively, leading to faster approval of new treatments. This directly impacts patient care, as it allows quicker access to new, potentially life-saving medicines.
In essence, ICON’s contribution is vital in driving forward medical innovations.
- Founding Team: John Climax and Ronan Lambe
- Founding Year: 1990
- Company Size: 41,160
Features of ICON
Here are some of the key features of ICON in clinical research:
Diverse Clinical and Scientific Operations
ICON offers a wide range of clinical and scientific operations services, ensuring comprehensive support for various aspects of clinical trials. This includes everything from study design to execution and data analysis.
Decentralized Clinical Trial Solutions
They provide end-to-end services, operational models, and technology to deliver customized solutions for decentralized clinical trials. This approach is increasingly important in today’s clinical research landscape, offering flexibility and efficiency.
Specialized Therapeutic Areas
ICON has expertise across multiple therapeutic areas including cardiovascular, central nervous system, endocrine & metabolic disorders, infectious diseases, internal medicine & immunology, oncology, and more. This broad expertise allows them to handle a wide range of clinical research projects.
Innovative Solutions for Biotech
ICON provides full-service outsourcing and flexible support customized to the specific needs of biotech companies. This includes due diligence and asset valuation, which are critical for biotech firms navigating the complex landscape of drug development.
Advanced Medical Imaging Solutions
Their expert medical imaging solutions support all stages of clinical research, improving decision-making, increasing efficiency, and reducing trial costs.
- Decades of expertise ensure high-quality clinical research.
- Offers wide-reaching capabilities for multi-regional clinical studies.
- Deep understanding of global regulations enhances compliance and efficiency.
- Invests in new technologies for more efficient trial processes.
- Broad range of specialties contributes to comprehensive service offerings.
- Managing multi-regional trials can lead to logistical challenges.
- Rapid growth may strain resources and affect service quality.
Our Review of ICON
When we researched ICON, we found both commendable aspects and areas for improvement. On the positive side, we appreciate their commitment to clinical research and their global presence, which allows for diverse study options. Their experienced team and advanced technology contribute to reliable data collection and analysis.
However, there are some drawbacks to consider. We have noticed occasional delays in project timelines, which can be frustrating. Additionally, the cost of their services tends to be on the higher side, making it a potential barrier for smaller research endeavors.
Parexel is a globally recognized company in clinical research, known for its important role in developing new medical treatments. They are one of the biggest clinical research organizations. Parexel conducts clinical trials, crucial steps in testing the safety and effectiveness of new drugs.
The work of Parexel is essential because it bridges the gap between medical research and the availability of new treatments to patients. One of the reasons they stand out as one of the best in this field is their rigorous approach to research.
Their commitment to quality and their global network also enables diverse and large-scale studies, setting them apart from others in the field. These strengths allow Parexel to deliver reliable and valuable data, accelerating the process of bringing new, effective medicines to the market.
Simply put, Parexel is a key player in transforming medical research into real-world health solutions.
About Parexel
- Founding Team: Josef von Rickenbach and Anne B. Sayigh
- Company Size: 18,900
Features of Parexel
Parexel, a global biopharmaceutical services organization, offers a range of features in clinical research. Here are some key aspects of their approach:

Patient-Centric Approach
Parexel emphasizes a patient-first strategy in their clinical trials. This approach results in deeper and more relevant insights for trial design and execution. This ensures that the trials are more aligned with patient needs and experiences.
Innovative Trial Designs
Parexel employs innovative trial designs to optimize trials for maximum impact. This includes advanced modeling and simulation to predict drug effects ahead of time, which can save time, money, and resources.
Regulatory Compliance and Market Access
Parexel designs studies and endpoints with market access in mind, ensuring that they satisfy global regulations. This approach helps in getting treatments to patients safely and quickly.
Patient Advocacy and Engagement
The company includes patient advocates in their council, using their experiences to improve trial designs. This inclusion demonstrates their commitment to understanding and incorporating patient perspectives in clinical research.
Focus on Speed and Precision
Parexel aims to design neuroscience trials with speed and precision, utilizing the right experts and specializations. This focus is crucial in delivering effective treatments on time.
- Provides advanced technology and analytics for efficient data management.
- Extensive network provides global insights with regional knowledge.
- Expertise in navigating complex regulatory environments worldwide.
- Broad experience across various therapeutic areas ensures versatile solutions.
- Focuses on patient engagement for more effective trial outcomes.
- Rapid expansion can lead to challenges in resource management.
- Concentration in specific areas could pose risks in market shifts.
Our Review of Parexel
Parexel is a notable player in the field of clinical research and pharmaceutical services. We’ve thoroughly analyzed their offerings and found both strengths and areas that need improvement.
On the positive side, Parexel excels in its commitment to innovation and technology. We appreciate their continuous efforts to simplify clinical trials and drug development processes, making them more efficient.
However, we also noticed some downsides. Communication with clients could be more transparent, with clearer updates on project progress. Additionally, there’s room for improvement in terms of ensuring consistency in service quality across different projects.
Other 12 Companies of Clinical Research

In the world of clinical research, beyond the well-known names, there are 12 other companies making significant contributions. Let’s explore their vital role in advancing healthcare.
1. Syneos Health
Syneos Health helps develop medicines by managing clinical trials for new drugs. They’re essential because they ensure medicines are safe and effective. Syneos Health stands out in clinical research for its comprehensive services and global reach, making drug development smoother and faster.
About Syneos Health
- Founding Team: Colin Shannon
- Founding Year: 1980
- Company Size: 28,000
PPD is a group that tests new drugs to see if they’re good and safe. This is crucial for getting new treatments to people. They stand out for their thorough research and global reach.
- Founding Team: Fred Eshelman
- Founding Year: 1985
- Company Size: 40,000+3
Labcorp does important tests and research for health. They’re needed because they help find out if new treatments are good. They’re among the best for their big labs and fast results.
About Labcorp
- Founding Team: Matthew Benger
- Founding Year: 1978
- Company Size: 75,5000
Medpace focuses on making sure new health treatments are safe. This is key for better medicine. They’re a top choice because of their focus on quality and detail in research.
About Medpace
- Founding Team: August Troendle
- Founding Year: 1992
- Company Size: 5,400
5. Charles River Laboratories
Charles River Laboratories tests drugs and does research to help pets and people stay healthy. They’re essential for safe, new treatments. Their expertise makes them a leader in the field.
About Charles River Laboratories
- Founding Team: Henry Foster
- Founding Year: 1947
- Company Size: 21,400
6. PRA Health Science
PRA Health Science works on finding out if new medicines are safe. This helps everyone get better treatments. They’re known for their excellent research and care in studies.
About PRA Health Science
- Founding Year: 1976
- Company Size: 17,000+
7. AdvanCell
AdvanCell specializes in new treatments, checking if they’re safe and working. Their work is vital for progress in medicine. They’re recognized for their innovation in research.
About AdvanCell
- Founding Team: Andrew Adamovich
Dynata gathers data for health studies. They’re needed for understanding what works in healthcare. They’re a top name for their accurate and wide-reaching data collection.
About Dynata
- Founding Team: Mike Petrullo
- Founding Year: 1940
- Company Size: 5000-10000
Covance helps with drug tests and research to fight diseases. Their role is key for new treatments. They’re celebrated for their comprehensive services and global impact.
About Covance
- Founding Team: Fred Cummings
- Founding Year: 1981
- Company Size: 50,000
MedNet provides software for managing clinical trials. This helps in making research easier and faster. They’re among the best for their tech solutions in research.
About MedNet
- Founding Team: John “Rob” Robertson
- Founding Year: 1996
- Company Size: 51-200
11. Fisher Clinical Services Inc.
Fisher Clinical Services Inc. manages the logistics of clinical trials, ensuring that treatments are tested efficiently. Their work is crucial for the progress of medicine, and they are renowned for their reliability and global network.
About Fisher Clinical Services Inc.
- Founding Team: John Pickering
- Founding Year: 1989
12. Worldwide Clinical Trials
Worldwide Clinical Trials conducts essential research to evaluate new medical treatments. Their work is critical for advancing healthcare. They are distinguished by their global expertise and commitment to innovation in clinical research.
About Worldwide Clinical Trials
- Founding Team: Neal Cutler
- Founding Year: 1986
- Company Size: 3,147
What To Consider When Choosing the Best Clinical Research Companies?
Choosing the right clinical research company (CRC) is crucial for the success of any clinical trial. Here’s a detailed guide on what to consider:

Expertise and Specialization
Always ensure the CRC has expertise in your specific therapeutic area. Companies with experience in similar drug trials or medical devices can better navigate the complexities of your project.
Regulatory Compliance
The CRC must adhere to regulatory guidelines like FDA (US) , EMA (Europe), and others. Check their track record in meeting these standards to avoid compliance issues.
Reputation and Track Record
You should research the company’s history. Look for testimonials, case studies, and reviews from past clients. A company with a strong reputation is likely to deliver quality results.
Project Management Capabilities
Effective project management is key. Assess their ability to manage timelines, budgets, and communication. A CRC that provides transparent, regular updates is preferable.
Patient Recruitment Strategies
Patient recruitment can be challenging. Evaluate their strategies for participant recruitment and retention. Consider their demographic reach and methods for ensuring a diverse participant pool.
Data Management and Analysis
The CRC should have strong systems for data collection, management, and analysis. Ask about their use of Electronic Data Capture (EDC) systems and how they handle data security and confidentiality.
Cost and Financial Terms
Get a clear understanding of the cost structure. Consider the value for money rather than just the lowest cost. Ensure there are no hidden fees and clarify what is included in the quoted price.
How Heartbeat AI Can Help You Get the Best List of Clinical Research Organizations?
Heartbeat AI, with its advanced features, can significantly assist in identifying the best list of clinical research organizations (CROs). Here’s how its various features contribute to this process:

Data Analysis and Processing
Heartbeat AI excels in analyzing vast amounts of data. When it comes to selecting CROs, it can process and analyze information from numerous sources, including past performance records, clinical trial reports, and regulatory compliance data. This thorough analysis helps in identifying CROs with a proven track record of success and reliability.
Machine Learning Algorithms
These algorithms enable Heartbeat AI to learn from historical data and improve its recommendations over time. By understanding trends and patterns in the successful execution of clinical trials, it can better predict which CROs are likely to meet your specific needs.
Predictive Analytics
Heartbeat AI uses predictive models to forecast future trends and outcomes based on historical data. This can be invaluable in predicting the success rate of CROs in upcoming projects, thus aiding in making more informed choices.
Customization and Personalization
The AI can be customized to your specific requirements. If you’re focusing on a specific therapeutic area or clinical trial phase, Heartbeat AI can prioritize specialized CROs in these fields.
Real-time Data Updates
The healthcare and pharmaceutical landscapes are constantly changing. Heartbeat AI’s ability to integrate and analyze real-time data ensures that the recommendations are based on the most current information available.
Integration with External Databases
Heartbeat AI can integrate with various external databases and platforms. This enables it to pull in comprehensive information about CROs from diverse sources, enhancing the accuracy of its recommendations.
Claim $500 of Free Data
Summing up, we’ve explored the best clinical research companies, diving into their features, strengths, weaknesses, and more. Clinical research is vital in healthcare; it’s key for advancing medical knowledge and developing new treatments.
With this guide, you’re equipped to find the right clinical research company that meets your specific needs. Whether it’s for innovative therapies, drug development, or medical advancements, choosing the right partner is crucial. This guide serves as a valuable resource to help you make an informed decision in the complex world of clinical research.
Frequently Asked Question
What services do companies of clinical research offer.
Clinical research organizations offer a wide range of services, including protocol development, patient recruitment, data collection and analysis, regulatory compliance, and more.
What is the role of a clinical research coordinator?
A clinical research coordinator is responsible for managing various aspects of a clinical trial, including patient recruitment, data collection, and ensuring compliance with protocols.
What is informed consent in clinical research?
Informed consent is the process by which participants in a clinical trial are fully informed about the study’s purpose, risks, and benefits. They voluntarily agree to participate based on this information.
The leading clinical research organizations
We've ranked the top 10 clinical research organizations.
UNBIASED RESEARCH RANKINGS
HIGHEST STANDARDS REQUIRED
PROPRIETARY CRITERIA SYSTEM

What people are saying: #Passionate #Innovative #Dedicated
Parexel is a clinical research organization that provides biopharmaceutical services and solutions. They have expertise in clinical development, translational medicine, and early phase research. Parexel has over 10,000 employees in 50 countries. Parexel has been recognized by Frost & Sullivan with the 2022 Global Customer Value Leadership Award. Parexel's services are designed to help pharmaceutical and medical device companies bring new treatments and therapies to market. Parexel's clinical development services include phase I-IV clinical trials, biostatistics, clinical data management, clinical supply chain management, decentralized clinical trials, medical monitoring and consulting, medical writing, patient sensors, pharmacovigilance, and risk-based quality management. The company's expanded access services provide access to investigational treatments for patients with serious or life-threatening conditions who are not eligible for clinical trials. Their outsourcing services include functional services provider and strategic partnerships.Parexel's team of experts has the knowledge and experience to help bring new treatments and therapies to market.

IQVIA clinical research
What people are saying: #Involved #ResearchOriented #PatientFriendly
IQVIA clinical research is a company that provides information on clinical trials and patient communities. They also have a section on their website where you can register for updates on new clinical trials and community events. The company has a strong focus on helping to expand meningitis research and they are currently recruiting participants for a new clinical study. They are also seeking volunteers to help advance respiratory syncytial virus (RSV) research. In addition, they are working on a clinical study to help find a way to speed up recovery from COVID-19 and reduce the need for hospital care. Overall, IQVIA clinical research is a company that is heavily involved in a variety of research studies in order to help improve medical treatments and find new cures for diseases.

What people are saying: #Comprehensive #Quality #Dedicated
Medpace is a full-service CRO that provides support for drug, biologic, and medical device programs. They have a global reach and offer services including clinical monitoring, biostatistics and data sciences, regulatory affairs, and more. They are dedicated to making the complex seamless and strive to provide the highest level of quality and service.

Syneos Health
What people are saying: #Quality #Experienced #Committed
Syneos Health is a biopharmaceutical company that provides solutions for clinical development, commercialization, and consulting. The company has a wide range of services and products that cater to the needs of their clients. They have a team of experienced professionals that work together to provide the best possible service. The company is committed to providing the highest quality of care to their clients and patients. They have a wide range of experience in different therapeutic areas, which allows them to provide the best possible care to their patients.

Charles River Laboratories
What people are saying: #Experienced #Dedicated #Successful
Charles River Laboratories is one of the top clinical research organizations in the United States. They offer a variety of services to help pharmaceutical and biotechnology companies develop and commercialize their products. These services include preclinical research, clinical research, and manufacturing support. Charles River Laboratories has a long history of helping companies develop new drugs and therapies. They have a team of experienced scientists and clinicians who are dedicated to helping their clients succeed. They offer a wide range of services, from preclinical research to clinical research to manufacturing support. They have a proven track record of success, and they are a trusted partner for many companies. If you're looking for a top clinical research organization to help you develop and commercialize your products, Charles River Laboratories is a great choice.

What people are saying: #Transparent #Innovative #Unique
Caidya is a leading clinical research organization that provides full-service support and vast therapeutic expertise to its clients. The company has a personalized approach to clinical research and offers a unique combination of global experience and knowledge sharing. Caidya's clinical research services are designed to connect you with unrivaled access to specialist knowledge and resources. The company's cutting-edge clinical technology integrates effortlessly to create an ecosystem that meets the specific needs of your study. Caidya's Clarity platform synthesizes data from different systems to create a near real-time view of your trial. This holistic view allows integrated teams within Caidya to transparently share information and data, generating insights to inform your decisions.

What people are saying: #Healthy #Helpful #Compensated
ICON is a clinical research company that is looking for healthy volunteers to participate in studies. The company has locations in Lenexa, KS and Salt Lake City, UT. Participation in a study includes 1 screening visit, 1 stay of 11 nights (12 days) and 1 follow-up visit. Compensation for participation is up to $6400. ICON is a great opportunity for people who want to make some extra money and help out with medical research.

What people are saying: #Trusted #Innovative #Leading
PPD is one of the top clinical research organizations in the United States. They offer a wide range of services, from drug development to clinical trials. They have a team of experienced professionals who are dedicated to helping their clients succeed.

CTI Clinical Trial and Consulting
What people are saying: #Experienced #FullService #Comprehensive
CTI Clinical Trial and Consulting is a full-service CRO that specializes in clinical research and consulting services. They offer a wide range of services including regulatory development, clinical project management, clinical monitoring, biometrics, quality assurance, and real world evidence. They also have a research center where you can participate in clinical trials. The range of services offered by CTI Clinical Trial and Consulting is excellent. They seem to be a one-stop shop for all your clinical research needs. We also like that they have a research center where you can participate in clinical trials. This is a great way to get involved in the latest medical research and to potentially help develop new treatments for various diseases.

Advanced Clinical
What people are saying: #Comprehensive #CustomerFocused #AwardWinning
Advanced Clinical is a top-tier contract research organization that has provided clinical research services to the pharmaceutical and biotech industry for many years. Their global reach and comprehensive solutions make them a perfect partner for companies launching clinical trials. Their focus on satisfaction and customer service is evident in their high employee retention rate and repeat business rate. Their award-winning culture and commitment to excellence make them a great choice for anyone looking for a clinical research partner.
Clinical Research Organizations: What should you be looking for?
The website's mission is to provide accurate, timely, and reliable rankings of clinical research organizations. We aim to provide users with a comprehensive and up-to-date list of the top clinical research organizations so they can make informed decisions about which ones to use. We also provide detailed descriptions of each organization so users can learn more about their work.
Are Clinical Research Organizations (CRO) worth it?
Yes, it is worth hiring clinical research organizations. Here are three reasons why: 1. Clinical research organizations have the experience and expertise to conduct clinical trials effectively. They know how to design trials that meet regulatory requirements, recruit patients, and collect data. 2. Clinical research organizations can help you save time and money. They can manage all aspects of the clinical trial process, so you can focus on other aspects of your business. 3. Clinical research organizations can provide valuable insights into the clinical trial process. They can help you understand the data, identify potential problems, and make decisions about the best course of action.
What to look for when hiring Clinical Research Organizations (CROs)?
Using a Clinical Research Organizations can be a great way to get the medical care that you need. However, there are a lot of questions that you may have about using one. That is why we have provided FAQs for customers interested in using a Clinical Research Organizations. We want to make sure that you have all the information that you need so that you can make the best decision for your medical care.
What are the specific research needs of your organization?
The first step in finding the right Clinical Research Organization (CRO) for your needs is to identify the specific research needs of your organization. These needs will vary depending on the type of research you are conducting, the size and scope of your project, and your budget. Once you have a clear understanding of your research needs, you can begin to research different CROs and compare their services. When considering a CRO, it is important to look at their experience in conducting the type of research you need. Make sure to ask about their success rates and customer satisfaction levels. You should also inquire about their pricing structures and what type of services are included in their fees. It is also important to consider the size of the CRO and their ability to handle your project. Make sure they have the staff and resources necessary to complete your project on time and within budget. Finally, you should also consider the location of the CRO. If your project requires frequent travel, you may want to choose a CRO that is located near your site. If you are conducting research that requires special expertise, you may want to choose a CRO that is located near a research university or hospital.
What is the size and scope of your organization?
Next, you need to understand the size and scope of your organization, and thereby understand the size and scope of the CRO you need to hire. Is the CRO a large, full-service clinical research organization that offers a wide range of services to its clients? Does the CRO have a global reach and is it able to provide its services to clients in a number of different countries? Does the CRO have a large team of experienced and qualified staff who are able to provide a high level of service to its clients? Is the CRO accredited by a number of different bodies and is it able to offer its services to a wide range of clients?
What are the your clinical research priorities?
Finally, you need to understand your organization's clinical research priorities. Are they to develop new treatments for diseases and conditions, to improve the safety and efficacy of existing treatments, or to increase our understanding of the underlying causes of disease? This will help you select a CRO that aligns with your needs.
Key Takeaways about Clinical Research Organizations
When choosing a clinical research organization, it is important to consider the size of the organization, its focus, its location, and its resources. The best clinical research organizations are those that are able to provide the most comprehensive services and the most experienced staff. They should also be able to offer the latest technology and facilities. Top Clinical Research Organizations is dedicated to providing objective rankings of clinical research organizations. We believe that our rankings will help you choose the best organization for your needs. We are committed to providing the most accurate and up-to-date information possible.
Frequently Asked Questions
What is a clinical research organization (cro).
A clinical research organization (CRO) is a company that provides services to pharmaceutical and biotechnology companies to help them outsource and conduct clinical trials. A CRO manages all aspects of a clinical trial, from start to finish, and works with a variety of stakeholders, including investigators, research sites, and patients.
CROs are an important part of the drug development process, as they provide expertise and resources that allow companies to efficiently and effectively conduct clinical trials. Without CROs, many new drugs and treatments would not be possible.
What services do CROs provide?
CROs provide a variety of services to support clinical research, including study design, data management and analysis, regulatory affairs support, and clinical trial management. CROs also provide a variety of other services, such as market research, project management, and training.
What are the benefits of working with a CRO?
There are many benefits of working with a clinical research organization (CRO). CROs can provide expertise and resources that Sponsors may not have internally, and can therefore help to increase the efficiency and quality of clinical trials. In addition, working with a CRO can help to reduce costs associated with clinical trials. CROs also have experience working with a variety of different Sponsors, and can therefore provide valuable insights and perspectives.
What are the risks of working with a CRO?
The risks of working with a CRO include the potential for conflicts of interest, the potential for data manipulation, and the potential for data fraud.
How do CROs ensure the safety of trial participants?
There are a number of ways that clinical research organizations (CROs) ensure the safety of trial participants.
First, CROs typically have staff members who are responsible for monitoring the safety of participants throughout the course of a study. These staff members may be nurses, doctors, or other health care professionals. They typically have experience in managing clinical trials and are familiar with the potential risks and side effects of the drugs or treatments being studied.
Second, CROs typically have established procedures for reporting and managing adverse events. These procedures ensure that any potential safety concerns are promptly identified and addressed.
Third, CROs typically work closely with the sponsor of a clinical trial (usually a pharmaceutical company) to ensure that the sponsor is aware of any safety concerns that arise during the course of the trial. The sponsor is ultimately responsible for the safety of the participants, and the CRO ensures that the sponsor is kept informed of any safety issues that arise.
Fourth, CROs typically conduct regular audits of their clinical trials to ensure that they are being conducted in accordance with good clinical practices and that the rights and safety of participants are being protected.
Finally, CROs typically have insurance policies in place that protect participants in case of any unforeseen events that may occur during the course of a trial.
Overall, the procedures and policies that CROs have in place help to ensure the safety of participants in clinical trials.
How do CROs ensure the quality of data collected during a clinical trial?
CROs ensure the quality of data collected during a clinical trial by following Good Clinical Practices (GCPs).
GCPs are a set of international standards that ensure the quality, safety, and integrity of data collected during a clinical trial. All CROs must adhere to these standards in order to conduct clinical trials.
What happens if a CRO is unable to meet its contractual obligations?
If a CRO is unable to meet its contractual obligations, it may be required to pay damages to the sponsor. The sponsor may also be able to terminate the contract and seek a new CRO.
What is the role of the sponsor in a clinical trial?
What are the responsibilities of the principal investigator in a clinical trial.
The principal investigator (PI) is responsible for the overall conduct of the clinical trial. This includes ensuring that the trial is conducted in accordance with the protocol, applicable regulations, and standard operating procedures. The PI is also responsible for the safety of the trial participants and for ensuring that the data collected are of high quality.
The PI works with the sponsor, other investigators, and the research team to ensure that the clinical trial is carried out correctly and that the data collected are of high quality. The PI is also responsible for the safety of the trial participants.
How are clinical trial sites selected?
When selecting a clinical trial site, sponsors and contract research organizations (CROs) consider a number of factors.
The most important factor is usually the site’s experience and success in conducting similar trials.
Other important factors can include the site’s patient population, geographic location, and facilities.
How are clinical trial subjects recruited?
There are a number of ways that clinical trial subjects can be recruited. One common method is through advertisements in newspapers or other media outlets. Another common method is through word-of-mouth, where people who know about the clinical trial spread the word to others who might be interested. Additionally, clinical trial subjects can be recruited through doctors or other medical professionals, as well as through patient advocacy groups.
How is informed consent obtained from clinical trial subjects?
Informed consent is obtained from clinical trial subjects by providing them with information about the trial in a way that is easy for them to understand. This includes information about the purpose of the trial, the procedures that will be used, the risks and benefits of participating, and their right to withdraw from the trial at any time. Informed consent is not simply a form that subjects must sign; it is a process of communication between the researcher and the subject that should ensure that the subject understands the trial and is willing to participate.
- Reach Our Healthcare List Expert +1 (786) 408 5757
- Browse Data Cards
- GET A QUOTE

The Top Players in Clinical Research: A Review of the Leading Companies
Top 10 leading clinical research companies in the usa.
The landscape of drug discovery and development is intricate, demanding significant resources at each stage of the process. As pharmaceutical companies strive to bring groundbreaking therapies to market, they increasingly rely on the expertise of Contract Research Organizations (CROs). These organizations play a vital role in supporting drug manufacturers throughout the entire drug development journey, from initial discovery to final approval. These CROs offer a comprehensive range of services, including clinical trial management, data research, and project management. Their critical role has become even more evident during the pandemic-induced challenges, where the need for rapid vaccine and drug development resulted in a surge of clinical trials.

The Impact of Covid-19 on Clinical Trials
At the outset of the pandemic, the clinical research landscape faced initial setbacks. However, the urgency to combat Covid-19 created an unprecedented rise in the number of clinical trials. The pressing demand for effective vaccines and treatments drove pharmaceutical companies and CROs to collaborate more than ever before. Looking ahead, the future of CROs appears promising, fueled by the rise of technologies that enable decentralized clinical trials. These modern approaches leverage digital tools and remote patient monitoring, reducing the need for physical site visits and expanding the reach of clinical research.
The Growth of the Global CRO Services Market
The significance of CROs is evident as the global CRO services market continues to expand exponentially. Projections indicate that this market, which was valued at US$76.6 billion , is expected to reach an impressive US$127.8 billion by 2028. This growth reflects the pivotal role CROs play in revolutionizing drug development and medical research.
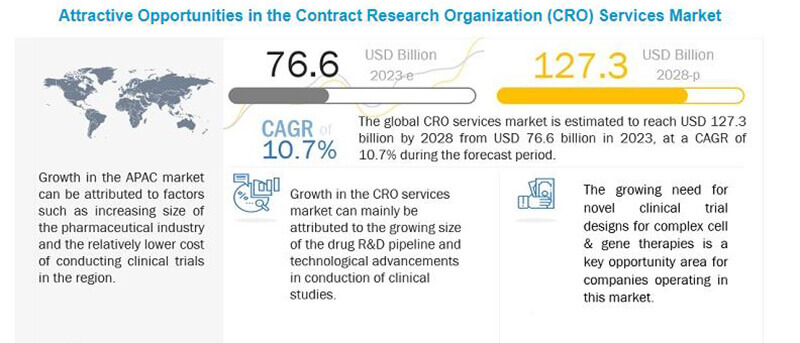
Source: Markets&Markets
Unveiling the Best 10 Clinical Research Organizations in the Field
Join us as we embark on this insightful journey, where we shine a spotlight on the ten biggest CROs shaping the pharmaceutical landscape. –
Source: Lablcorp
Labcorp, a leading provider of comprehensive drug development solutions for various industries, achieved a significant milestone in 2015 with its acquisition of Covance for an impressive $6 billion. This strategic union brought together Covance's expertise in drug development and Labcorp's unparalleled medical testing capabilities, propelling the company to become the world's foremost healthcare diagnostics company.
Over the past decade, Labcorp has continued to expand its influence through a series of strategic acquisitions. Notable additions to their portfolio include LipoScience, Inc., Bode Technology Group, Sequenom, MNG Laboratories, and Personal Genome Diagnostics (PGDx). These acquisitions have further strengthened Labcorp's position in the market, broadening its scope and enhancing its ability to provide cutting-edge solutions.
Looking forward to 2023, Labcorp unveiled its plans to spin off its Contract Research Organization (CRO) segment, creating a separate, independent publicly traded entity known as Fortrea.
Source: IQVIA
In 2016, two industry giants, Quintiles and IMS Health, joined forces and rebranded as IQVIA, establishing the largest Contract Research Organization (CRO) globally. With a presence in over 100 countries, IQVIA brings together cutting-edge advancements in data science, technology, and human science expertise, offering clients a comprehensive end-to-end clinical and commercial service. Through a series of strategic acquisitions of smaller specialist companies, IQVIA has strengthened its position and continues to lead the way in the world of CROs.
In the year 2022, IQVIA demonstrated remarkable growth and success, generating revenues of $14.494b . This impressive growth, amounting to 3.29% on a reported basis and 7.8% at constant currency , highlighted the company's continued upward trajectory. Despite uncertain market conditions, IQVIA achieved record bookings and surpassed the goals set in their Vision 22 plan, underscoring their commitment to excellence and innovation.
Source: Parexel
Parexel continues to hold its position as one of the largest CROs globally. Specializing in Phase I to IV clinical development services, Parexel plays a vital role in expediting and ensuring the smooth progress of the drug approval process. With a comprehensive array of service offerings, the company caters to nearly every aspect of clinical trial management, supporting sponsors in conducting successful clinical studies.
In a significant development in November 2021, Parexel was acquired by EQT Private Equity and Goldman Sachs for a remarkable $8.5 billion . Despite this transition, the commitment to putting patients first remains unwavering, ensuring that Parexel maintains its patient-centric trajectory. The newly constituted board of directors brings together a wealth of experience in the life sciences industry, further strengthening Parexel's position as a leader in the field.
4. Pharmaceutical Product Development (PPD)
Source: PPD
Pharmaceutical Product Development (PPD) stands as a leading global Contract Research Organization (CRO) with an extensive workforce of over 30,000 professionals worldwide.
The acquisition of Evidera in 2016 marked a significant milestone for PPD, solidifying their position as a leader in real-world research. Leveraging Evidera's expertise in real-world evidence, PPD has enhanced its capabilities in providing life science companies with a crucial element of the clinical development process, maintaining a competitive edge in the industry. Subsequent strategic acquisitions of Synexus (now Accelerated Enrollment Solutions) and Bioclinica, both patient recruitment and clinical research site businesses, respectively, have further augmented PPD's offerings. These collective advancements have propelled PPD's core organic growth to "high teens" in the segment, as the company achieved remarkable full-year 2022 results. With over $7.00 billion in revenue and a significant contribution of over $2.00 to adjusted earnings per share, PPD remains a dominant force in the CRO landscape, upholding their mission to facilitate groundbreaking clinical research and development.
5. Syneos Health
Source: Syneos Health
Founded in 1999, Syneos Health has its headquarters in Morrisville, North Carolina. With an impressive annual revenue of $5,213 million and a workforce of 28,000 employees, Syneos Health is a leading international Contract Research Organization (CRO) that offers a wide range of services spanning all aspects of bringing new therapies and products to the market.
While Syneos Health provides clinical development services across all stages of drug development, it has particularly specialized expertise in assisting healthcare organizations during the late stages of clinical trials. Some of them include Early phase trials, Late phase trials, Decentralized clinical trial solutions, Clinical data management, Pharmaceutical trial services, Medical device diagnostics trial services, and more.
Source: ICON
Operating across 46 locations worldwide, ICON is a top-tier Contract Research Organization (CRO) that provides a comprehensive range of services, including consulting, clinical development, and commercialization services.
In a pivotal move in 2016, ICON formed strategic partnerships with Genomics England for the UK's ambitious 100,000 Genomes Project and IBM Watson for oncology research support. These collaborations expanded ICON's service offerings and strengthened their presence in the fields of genomic science and oncology research, creating new opportunities for clinical research jobs in these cutting-edge sectors.
Additionally, the year 2022 marked a significant milestone for ICON, as it achieved impressive financial results. With full-year revenues amounting to US$7.7 billion , the company witnessed substantial growth of 41.2% compared to the previous year. On a constant currency basis, the revenue surge reached an outstanding 45%, reflecting ICON's continued success in advancing clinical research and serving as a leading partner for the life sciences industry.
7. Charles River Laboratories
Source: Charles River Lab.
Since its inception in 1947, this company has grown into an exceptional industry leader, specializing in cutting-edge cell and gene therapies. Not only does it provide vital lab services to the pharmaceutical, medical device, and biotech industries, but it also stands out with its impressive global presence. With a robust network spanning over 110+ facilities scattered robust network spanning over 110+ facilities scattered across 20+ countries, the company offers unmatched accessibility and support to clients worldwide, solidifying its reputation as a trailblazer in the field.
As of the latest data available in 2023, the company boasts an impressive annual revenue of 4.092B, a 12.73% increase year-over-year , reflecting its strong position in the market and significant contributions to the life sciences industry.
Source: CTI
CTI Clinical Trial and Consulting Services has been making waves in the industry since its establishment in 1999. With a remarkable track record of driving over 150 new drug and medical device approvals, the company has firmly established itself as a leader in the field.
Boasting a vast international footprint, CTI operates offices in more than 60 locations across the globe, ensuring its presence and support in key regions such as North America, Europe, Latin America, Middle-East, Africa, and Asia-Pacific. In a strategic move in 2021, CTI further bolstered its position by announcing the acquisition of Dynakin, a European-based strategic consulting company. This acquisition aimed to fortify CTI's standing as a comprehensive and dynamic full-service clinical research organization, ready to take on the challenges of a rapidly evolving healthcare landscape.

Source: Medpace
MEDPACE is a global organization with a workforce of over 5,000 employees spread across 40 countries. In 2022, Medpace reported an impressive full-year revenue of $1.46 billion, reflecting a substantial 27.8% increase from the previous year. In an earnings call, Jesse Geiger, the President at Medpace, highlighted the company's ability to grow its workforce by 15.8% despite facing challenges in a competitive labor environment. In 2023, Medpace places significant emphasis on employee retention and ongoing recruitment as top priorities to support its future business endeavors.
10. Fisher Clinical Services
Source: Thermo Fisher
Fisher Clinical Services is a vital division of Thermo Fisher Scientific with a rich history of over two decades in supply chain management. The company's core focus centers on efficiently handling the distribution and packaging requirements for clinical trials conducted worldwide.
With an impressive annual revenue of $20 billion and a workforce of 70,000 employees, Fisher Clinical Services is dedicated to delivering high-value products that align with the rigorous standards of pharmaceutical companies, ensuring reliability, sustainability, and optimal performance. The company plays a crucial role in supporting the success of clinical trials across the globe.
In conclusion, the best Clinical Research Companies in the USA serve as indispensable partners for pharmaceutical, biotech, and medical device industries embarking on new medicine and medical device development. With the complexities of clinical trials escalating, CROs play a pivotal role in managing and leading these trials efficiently amidst rising prices, stringent regulations, and tighter deadlines.
As the pharmaceutical landscape continues to evolve, pharmaceutical companies are increasingly turning to Clinical Research Organizations for their R&D activities to maintain competitiveness, flexibility, and profitability. With their proven track record, comprehensive services, and commitment to excellence, the best Clinical Research Companies in the USA stand as crucial allies in the quest to advance medical science and improve patient outcomes. Through fruitful partnerships with CROs, the pharmaceutical, biotech, and medical device industries continue to push the boundaries of innovation and deliver life-changing products to those in need.
Healthcare Databases
- Healthcare Email List
Doctors Email List
- Nurses Email List
- Medical Executives Email List
- Physician Email List
- Dentist Email List
- Pharmacist Email List
- Healthcare SIC Code
- Healthcare NAICS Code
- Chiropractors Email List
- Veterinarian Email List
- Radiologists Email List
- Psychiatrists Email List
- Pediatrician Email List
- Cardiologists Email List
- Neurologist Email List
- Dermatologist Email List
- Oncologists Email List
Medical Services
- Hospitals Email List
- Pharmacy Email Database
- Nursing Homes Email List
- X-Ray Laboratories Mailing List
- Pharmaceutical Email List
- Assisted Living Email List
- Dental Clinics Email List
- Clinics Email List
- Physical Fitness Facilities List
HealthcareMailing
- +1 (786) 408 5757
- [email protected]

- Privacy Policy
We are a leading global provider of CRO services, and our company’s Mission is to enable our customers to make the world healthier, cleaner and safer.
In planning, in strategy, in recruitment. In every aspect of inclusive research, we’re all in.
Explore the latest utilization and growth projection of FSP partnerships in clinical research and the future of FSP strategies and models.
Our colleagues in clinical research services are at the forefront of getting therapies to market. Explore opportunities to join our global team.
PPD is a leading global contract research organization focused on delivering life-changing therapies

Clinical development
Laboratories, ppd careers, connect with us, ppd offers proven solutions, from early development to phase ii-iii to pharmacovigilance to peri- and post-approval services, to ensure your product’s success.

PPD offers a comprehensive set of lab services – from bioanalytical to vaccine sciences.
Offering proven solutions, ranging from early development to pharmacovigilance to post-approval services, to ensure your product’s success.
Clinical trial therapeutic expertise
Learn about our expertise in areas like oncology, cardiovascular, rare diseases and more.
Fuel your drug development more efficiently
Here are four key areas to consider when choosing the right FSP partner for you.
130k+ colleagues globally connected by our mission to enable our customers to make the world healthier, cleaner and safer.

Top 10 largest clinical research organizations
We take a look at the 10 biggest contract research organizations in the pharma sector in 2022.

The drug discovery process is complex. The clinical stage is particularly resource-intensive, something that has led to the rise in demand for contract research organizations (CROs) that can support drug manufacturers at each stage of the process, from discovery to approval. The core activities a CRO can provide include (but are not limited to) clinical trial management, data research and project management.
Despite an initial downturn at the start of the pandemic, Covid-19 created a spike in the number of clinical trials taking place due to the need for vaccines and drugs to tackle the virus. In the coming years, we expect the rise of technologies that enable decentralized clinical trials to help expand the market share of CROs.
Currently the global CRO services market is projected to grow from US$73.38 bn to $163.48 bn by 2029. Here Pharma IQ takes a look at the 10 biggest CROs in pharma today.
Founded in 1982, IQVIA is an American multinational company formed through the merger of Quintiles, a leading provider of product development and integrated healthcare services, and IMS Health, a global information and technology services company. The latter has enabled company to have a strong focus on digital solutions and analytics. In 2017, Quintiles IMS rebranded to IQVIA.
Laboratory Corporation of America Holdings, also known as Labcorp, is an American company that operates one of the largest clinical laboratory networks in the world. In an average week Labcorp processes tests on more than 3 million patient specimens. In 2020 the company earned revenue in excess of $14 bn.
Syneos Health
Founded in 1999, Syneos Health was created following the merger of two biopharmaceutical companies: INC Research and inVentive health. Today it has offices in more than 110 countries and offers services as a CRO as well as consultancy.
Founded in 1985 as a one-person consultancy firm, Pharmaceutical Product Development (PPD) is a global contract research organization that provides drug development, lab and lifecycle management services. In 2020 the company made US$4.7bn and the following year it became part of Thermo Fisher Scientific.
Headquartered in Dublin, Icon provides strategic management and support for clinical development from the compound selection stage through to clinical trials. Its services include clinical trials management, biometric activities, investigator recruitment and outcomes research.
Parexel was founded in 1982 and acquired by private equity firm Pamplona Capital in 2017 in a deal worth $5 bn. In 2021 it was bought by EQT Private Equity and Goldman Sachs Asset Management. The company conducts clinical trials and operates in more than 50 countries. It makes around $3 bn in revenue annually.
Charles River Laboratories
Founded in 1947, today this company specializes in cell and gene therapies as well as lab services for the pharmaceutical, medical device and biotech industries. As of 2021 it operates more than 90 facilities in 20 countries and has an annual revenue of $3.54 bn.
Award-winning MedPace has offices on six continents, with headquarters in Ohio where it has a clinical research campus and a number of clinical and bioanalytical laboratories. It provides clinical trial services for Phase I-IV studies in the biotech, pharma and medical device industries. Its revenue has been growing steadily year- on-year and is currently almost $1 bn.
CTI Clinical Trial and Consulting Services
This organization was founded in 1999 to provide clinical trial services and bring new drugs to market. It operates in more than 60 countries and since its inception has contributed to the approval of more than 150 new drugs and medical devices around the world.
WuXi AppTec
Founded in 2000 in Shanghai, WuXi AppTec is the newest company in our top 10. It provides services across the entire development cycle including small molecule R&D and manufacturing, biologics R&D and manufacturing, cell and gene therapy. It currently operates in 18 locations across China, Iceland and the US.
Quick links
- How to improve the success rate of clinical trials
- Webinar: The technology enabling decentralized clinical trials
- The challenges of handling and delivering highly potent oncology drugs
Get exclusive access to member-only articles, reports, videos, interviews, webinars and other premium content from industry experts and thought leaders by signing up to Pharma IQ here .
Upcoming Events
Digit pharma & health 2024.
September 10 - 12, 2024 Düsseldorf, Germany

Temperature Control and Logistics North America Summit
September 10 - 12, 2024 Falls Church, VA

AI for Pharma & Healthcare
24 - 26 September, 2024 Amsterdam Marriott Hotel

SmartLabs Europe
February 25 - 26, 2025 Novotel Amsterdam City, Netherlands

SmartLab USA 2025
07 - 08 April, 2025 Le Méridien, Fort Lauderdale, Florida, USA

Pharma Contract Manufacturing
08 - 10 April, 2025 Berlin

Subscribe to our Free Newsletter
Insights from the world’s foremost thought leaders delivered to your inbox.
Latest Webinars
Pharma iq's power list 2022: in conversation with pharma's top leaders.
2022-10-18 02:00 PM - 03:00 PM BST

A post-pandemic 3D view of the patient and supply journey
2022-06-01 04:30 PM - 05:30 PM CET

Discover how targeted radiotherapy induced toxicity can be identified with imaging
2022-04-28 01:00 PM - 02:00 PM EST

RECOMMENDED

FIND CONTENT BY TYPE
- Infographics
Pharma IQ COMMUNITY
- Advertise With Us
- User Agreement
- Cookie Policy
- Become a Member Today
- Pharma IQ App
ADVERTISE WITH US
Reach Pharmaceuticals & Biotechnology professionals through cost-effective marketing opportunities to deliver your message, position yourself as a thought leader, and introduce new products, techniques and strategies to the market.
JOIN THE Pharma IQ COMMUNITY
Join Pharma & Biotech today and interact with a vibrant network of professionals, keeping up to date with the industry by accessing our wealth of articles, videos, live conferences and more.

Pharma IQ, a division of IQPC
Careers With IQPC | Contact Us | About Us | Cookie Policy
Become a Member today!
PLEASE ENTER YOUR EMAIL TO JOIN FOR FREE
Already an IQPC Community Member? Sign in Here or Forgot Password Sign up now and get FREE access to our extensive library of reports, infographics, whitepapers, webinars and online events from the world’s foremost thought leaders.
We respect your privacy, by clicking 'Subscribe' you will receive our e-newsletter, including information on Podcasts, Webinars, event discounts, online learning opportunities and agree to our User Agreement. You have the right to object. For further information on how we process and monitor your personal data click here . You can unsubscribe at any time.

Recognized for Excellence.
Find out why we were the highest-rated ph i service provider for overall user preference and the highest-rated ph ii/iii cro for overall performance., discover why your cro search is over.

You and your work matter here.
Accessible partners. Shared commitment. Personalized for you.

Discover why it’s a world of difference at Worldwide.
Clinical research isn’t a straight path. let’s navigate it together..
At Worldwide, we believe a personalized approach is the only way to unlock the power of everyone working on a study – from operational and therapeutic experts to site partners and scientists. That means you’ll always have direct access to our experts. You’ll be able to tap into more than 30 years of therapeutic experience on a global scale. And your mission is our mission – we are dedicated to working with you to meet your goals.
White Paper
Biomarkers in Oncology Studies
The power of personalization amid the changing cro landscape.
Press Release

Worldwide Clinical Trials Recognized with Coveted 2024 CRO Leadership Awards Based on Customer Feedback for 11th Consecutive Year
Want a cro partner who gives you their full attention you’re in the right place..
Michael Murphy, MD, PhD Chief Medical and Scientific Officer
Your extended team…always within reach.
We were founded on an unwavering commitment to therapeutic excellence and personalized attention. And today, that’s more important than ever. That means you have access to our senior-level experts who will work directly with you. It means we listen to you, understand your study’s needs, and actively seek customized solutions that are tailored to your specific project – even if we need to pivot and adapt along the way.
Every study is different. Every path forward is different. And we will partner with you every step of the way.

Aman Khera, MBA, FTOPRA, FRAPS Vice President, Regulatory Science, Strategy and Innovation
Flexible? Check. Responsive and nimble? Absolutely.
We understand that no clinical trial looks the same. Your trials deserve solutions backed by decades of therapeutically relevant and adaptable expertise. That’s exactly what we bring to the table.
When you partner with Worldwide, we’ll stay by your side through the entire lifecycle of product development with nimble, solutions that are never “just off the shelf” but customized for you and your specific needs.
Let’s work together to bring clarity to complex drug development.
Schedule a consultation.
Want to see what’s possible? Connect with us!
Request a Proposal
Ready to work with a CRO partner who gives you our full attention?
Meet Us at an Event
Where in the world is Worldwide? If you’ll be at an upcoming event, let’s connect! Here’s where we’ll be:
Outsourcing Clinical Trials UK & Ireland
July 9 Event with Suvoda
Clinical Trials in Oncology East Coast
2024 ESMO Congress
Send Us a Message
A member of the team is online now to help address your needs.
Explore Open Careers
Cookies on the worldwide clinical trials website.
We use cookies to improve your browsing experience and help us improve our websites. We have carefully selected third parties that use cookies to achieve purposes illustrated in the cookie policy. For more information. Read more about online privacy statement . By closing this banner or continuing to browse our website, you agree to our use of such cookies.
Therapeutic Expertise
Participate

Explore end-to-end solutions throughout development — from portfolio optimization and regulatory strategy, to Phase I-IV clinical trials, market access planning, and more.
- Portfolio management and asset valuation
- Early development and innovation
- Integrated clinical development
- Approval and access
- Value substantiation lifecycle management
HOW WE DO IT
- Delivery models
- Operational excellence
- Building patient insights into assets, profile and claims
- Portfolio optimization
- Asset valuation and indication prioritization
- Early evidence review
- Model-based drug development
- Integrated development strategy and planning
- Phase I Clinical Trials
- Proof of Concept Studies: Phase IB-IIA
- Patient Engagement Strategy and Enrollment Solutions
- Patient Inclusion
- Site Alliance Network and KOL Engagement
- Protocol Optimization
- Regulatory Strategy
- Market Access Strategy and Delivery
- Biomarker and Genomic Medicine Strategy
- Clinical Trial Supply & Logistics
- Medical Communications
- Phase IIB-IV Clinical Trials
- Real World Evidence
- Protocol-Driven, Customized Site Solution Strategy
- Regulatory Strategy, Submissions, Compliance, and Outsourcing
- Clinical Development Technology Optimization
- Global Regulatory Submissions and Outsourcing
- Compliance and Risk Management
- Real-World Evidence, Market Access Strategy and Planning
- Regulatory Compliance, Drug Safety and Pharmacovigilance
- Lifecycle Optimization
- Gain an advantage through FSP
- Leveraging AI and digital in clinical development

Parexel Biotech provides the end-to-end capabilities you’ll need to succeed. We're fast, flexible, and dedicated to impacting patient lives.

Utilize our expertise across therapeutic areas, combining innovative trial designs, leading clinical and regulatory expertise, global reach, and a passion for changing patient lives.
- Neuroscience
- General medicine
- Infectious disease & vaccines
- Inflammation & immunology
Cross-Therapeutic Expertise
- Cell & gene therapies
- Rare diseases

Our experts help you stay at the forefront of the industry - and ahead of change.
New Medicines, Novel Insights
- Advancing precision oncology
- Advancing rare disease drug development
- Accelerating development of cell and gene therapies
- Achieving patient-guided drug development
Discussions on Diversity
- Chapter 1 Bridging the Gap
- Chapter 2 Beyond the Binary
The Regulatory Navigator
- Pursuing alternative approaches to animal models in drug development: EMA innovation task force and scientific advice opportunities
- EU-CTR transitions: To meet the deadline, act now
- Potency assurance for CGT products: FDA's new draft guidance
Latest Report

New Medicines, Novel Insights: Advancing precision oncology

Thinking about joining a clinical trial? Learn the drug development process, what it’s like to participate, how to find a trial, and answers to frequently asked questions.
INTERESTED IN PARTICIPATING?
- Healthy Volunteers
- Patient Volunteers
- Patient Advocacy Groups
HEAR FROM REAL PATIENTS
- Patient Stories
TRIAL SITES

Want to collaborate with us to offer clinical trials at your site? We would welcome the opportunity to discuss.
We are one of the largest CROs in the world, speeding life-changing medicine to market by engaging patients With Heart ™. Learn about who we are, what we do, and what we believe.
- Leadership team
- Global reach
- Our DE&I strategy
- Compliance, tax & privacy
- Meet us at an event
- Board of Directors
- Our ESG strategy
- Compliance & Ethics
- Tax Strategy
- Privacy Policy
- Supplier Data Privacy Requirements
- Terms of Use
- French Gender Pay Data
- UK Gender Pay Data
- UK Modern Slavery Act Statement
- Data Privacy Framework
- Supplier Diversity Policy
What can we help you find today?
The perspectives and opinions expressed in this material represent those of the patient advocate only and should not be considered a solicitation, promotion or advertisement for any services of Parexel, or any drugs or therapies, including those under development. Participating in clinical trials for investigational medicines offers patients potential benefits, such as access to cutting-edge treatments and expert medical care, while contributing to medical research. However, risks may include side effects, unpredictable outcomes, and time commitment. Careful assessment of these factors helps patients make informed decisions. The content of this material, including graphics, images and text, is provided for informational purposes only and does not constitute medical advice, diagnosis or treatment. Please consult your healthcare professional for medical advice. The patient advocate has provided their consent for the use and distribution of this content.
Patient Story
Rare Diseases
- Inflammation & Immunology
Cell & Gene Therapy
While running a triathlon, andrea stumbled and realized something was wrong..
Her condition worsened. Within months, she was forced to walk with a cane.
Then, finally, came the diagnosis.
She thought it was just an injury. Her hamstrings were tight, and her body felt stiff and slow.
She had als, a condition affecting more than 200,000 people around the world., her life expectancy was shortened to just two to five years — but she was determined to make the most of it., andrea continued to join triathlons, even as she underwent treatment in a clinical trial, inspiring many and raising $1,000,000 for als research., today, alive beyond her original prognosis, she serves on our patient advisory council, using her experiences to help improve our neuroscience trials., lives can change when you design neuroscience trials with speed and precision..
- Utilize the right experts, with the right specialization
- Find the patients you need and earn their trust
- Satisfy global regulations to get your treatment to patients safely and quickly
- Design studies and endpoints with market access in mind
What we do, we do
Our Experts
Our neuroscience specialists collaborate to get your treatment to patients faster.
MORE EXPERTS
Antal János, M.D., M.B.A.
Vice President, Glob...
Karla Kanis, M.D.
Mohit Verma, M.D.
Global TA Section He...

Andreas Lysandropoulos, M.D., Ph.D.
Senior Vice President, Global Neuroscience Therapeutic Area Head
With 15+ years of experience as a clinical neurologist, principal investigator, and medical leader, Andreas works with you to create an effective development strategy. Once the plan is in place, he and his team lead all aspects of execution — ensuring your assets are approved quickly and efficiently.
"For me, ‘With Heart’ means with passion and empathy. It is my mission to gather people around a common cause and inspire everybody to reflect on and be passionate about what we do together because it matters to patients who rely on us all for a better life."
Our diverse neuroscience experiences ensure you get the expertise you need across all indications of neurology and psychiatry.

Innovative trial designs allow us to optimize your trials for maximum impact.
Neuroscience sites around the world allow us to accelerate study startups.
- North America
- South America
- Middle East & Africa

Advanced modeling and simulation allow us to predict drug effects ahead of time, saving time, money, and resources.
And our patient-first approach results in deeper, more relevant insights for trial design and execution.
BENEFITS MAY INCLUDE
What can we do to help you change patient lives?
See neuroscience capabilities Visit all therapeutic areas Explore neuroscience careers
Discover other patient stories
Sara's only symptom was fatigue. But after some routine tests, she was diagnosed with breast cancer.
When Austin was 3 years old, his parents realized something was wrong. Multiple falls, concussions, and a broken arm led to a diagnosis.
Tina had lost her father to Crohn's disease. Now she faced her own diagnosis — and it derailed her life.
One night, while watching TV with her husband, Robyn felt a lump in her neck. As a doctor, she knew the risk.
We focus on patients, because they inspire us to deliver better trials, faster than ever . So we can make a difference for more patients everywhere.
Who we are,, parexel is proudly among the world’s largest clinical research organizations.
A dedicated CRO providing the full range of Phase I to IV clinical development services and leveraging the breadth of our clinical, regulatory and therapeutic expertise , our team of more than 21,000 global professionals works in partnership with biopharmaceutical leaders and sites to design and deliver clinical trials with patients in mind , to make clinical research a care option for anyone, anywhere.

- Find a Study
- Salt Lake City, UT
- San Antonio, TX
- What is Clinical Research?
- Why Participation is Important?
- Refer a Friend
- ICH GCP (De)
- ICH GCP (En)
- ICH GCP (Es)
- ICH GCP (Fr)
- ICH GCP (It)
- ICH GCP (Pt)
- ICH GCP (Ru)
- AUSTRALIA (NHMRC)
- JAPAN (PMDA)
- US Clinical Trials Registry
- EU Clinical Trials Registry
- Pharmaceutical Companies
- Clinical Research Labs
- Service Companies
- Clinical Research Events
- Publications
- Researchers
Top 15 Contract Research Organizations (CROs) in 2024

Post-pandemic, as drug development businesses recover and grow, CROs around the world continue to undergo profound transformation. As in previous years, 2023 will be all about mergers and acquisitions, with big players getting even bigger and smaller companies surviving thanks to niche advantages such as technological know-how or brilliant scientists among their employees.
The top 15 CROs globally would be in the following order based on 2023 revenue :
- LabCorp (Covance) , with 15.05 billion USD (2023) and 75000 employees ( Labcorp , 2024). In February 2015, Labcorp completes its $6 billion purchase of Covance, Inc., creating the world’s leading health care diagnostics company. The combination of Covance’s drug development leadership and Labcorp’s medical testing expertise builds the market leader in central laboratory and bioanalysis services. Labcorp’s clinical trials companies, Labcorp Clinical Trials and Tandem Laboratories, align under the Covance brand.
- IQVIA , with 14.85 billion USD (2023) and 86000 employees (IQVIA, 2022). IQVIA, formerly Quintiles and IMS Health, Inc., is an American Fortune 500 and S&P 500 multinational company serving the combined industries of health information technology and clinical research. IQVIA (NYSE:IQV) is a leading global provider of advanced analytics, technology solutions, and clinical research services to the life sciences industry. With approximately 86,000 employees, IQVIA conducts operations in more than 100 countries.
- ICON (PRA) , with 7.74 billion USD (2022) and 41160 employees (ICON, 2023). From a small team of 5 people in 1990, ICON now employs over 41,100 people. ICON has been recognised as one of the world’s leading Contract Research Organisations through a number of high-profile industry awards .
- Thermo Fisher Scientific (PPD) , with 7.02 billion USD (2023) and 35000 employees (Businesswire, 2023). Recognized as a global industry leader in accelerating promising medicines from early development through regulatory approval and market access, we serve pharma, biotech, medical device and government organizations with custom-tailored solutions, including full-service partnerships and functional service partnerships. As a strategic partner in clinical development and analytical services, we apply cutting edge technologies, therapeutic expertise and a firm commitment to quality to help customers deliver life-changing therapies.
- Lonza - 6.5 billion USD, 17896 employees. Though being a contract development and manufacturing organization (CDMO), rather than a CRO, takes the 10th position with the 5.9 billion USD in sales and over 17500 employees worldwide (2023), offering “proprietary line of in silico and in vitro services for manufacturability, immunogenicity, potency assessment, humanization and protein engineering” ( https://pharma.lonza.com , 2022). Founded in 1897 in the Swiss Alps, today, Lonza operates across five continents. With approximately 17,500 full-time employees, it comprises high-performing teams and individual talent that make a meaningful difference to our own business, as well as to the communities in which we operate. The business benefits from global supply chains, but we have worked to maintain the agility to address marketplace needs on a local level. CDMOs become reliable partners for Pharma companies as their “services reduce R&D costs while improving productivity and are essential to providing safe and effective treatment to patients” ( https://pharma.lonza.com , 2022.
- Wuxi AppTec with 5.8 billion USD revenue in 2021 and 44360 employees (Companiesmarketcap, 2022). As a global company with operations across Asia, Europe, and North America, WuXi AppTec provides a broad portfolio of R&D and manufacturing services that enable the pharmaceutical and healthcare industry around the world to advance discoveries and deliver groundbreaking treatments to patients. Through its unique business models, WuXi AppTec’s integrated, end-to-end services include chemistry drug CRDMO (Contract Research, Development and Manufacturing Organization), biology discovery, preclinical testing and clinical research services, and cell and gene therapies CTDMO (Contract Testing, Development and Manufacturing Organization), helping customers improve the productivity of advancing healthcare products through cost-effective and efficient solutions. WuXi AppTec received AA ESG rating from MSCI in 2023 and its open-access platform is enabling more than 6,000 customers from over 30 countries to improve the health of those in need – and to realize the vision that "every drug can be made and every disease can be treated."
Syneos Health® is a leading fully integrated biopharmaceutical solutions organization built to accelerate customer success. We translate unique clinical, medical affairs and commercial insights into outcomes to address modern market realities.
We bring together a talented team of professionals, who work across more than 110 countries, with a deep understanding of patient and physician behaviors and market dynamics. Together we share insights, use the latest technologies and apply advanced business practices to speed our customers’ delivery of important therapies to patients.
Cell and Gene Therapy CDMO Solutions has supported the development of 11 FDA approved cell and gene therapies and have conducted more than 900 studies in this field over the past year. Unsurpassed end-to-end offering results in enhanced access to scientific and regulatory expertise via multidisciplinary bench of experts to help you problem solve.
Parexel (EQT Private Equity and Goldman Sachs Asset Management) with 3.8 billion USD in 2022 and 21000 employees (Parexel, 2023). Parexel is among the world’s largest clinical research organizations (CROs), providing the full range of Phase I to IV clinical development services to help lifesaving treatments reach patients faster. Leveraging the breadth of our clinical, regulatory, and therapeutic expertise, our team of more than 21,000 global professionals works in partnership with biopharmaceutical leaders, emerging innovators, and sites to design and deliver clinical trials with patients in mind, increasing access and participation to make clinical research a care option for anyone, anywhere.
- Medpace Holdings, Inc. with 1.135 billion USD revenue in 2021 and 5400 employees (Medpace, 2023). Integrating core clinical trial services delivers efficient and streamlined execution and higher quality results. Through its wholly-owned subsidiaries, Medpace offers clinical pharmacology, as well as supporting laboratory services including central labs, bioanalytical lab, ECG core lab, and imaging core labs.
Worldwide Clinical Trials – 0.6 billion USD , 3000 employees, 60 countries.
Worldwide Clinical Trials started out more than 30 years ago as a small science team with a big goal: To always provide authentic, personalized attention to our partners. And as the industry has grown, CROs have merged, and trials have grown increasingly complex, we haven’t forgotten what truly matters: People.
With more than 3,000 employees in more than 60 countries around the world, we’re still staying true to those roots. Our team is dedicated to staying accessible, flexible, and solution-focused to ensure our partners not only have the best possible outcomes – but they also know their Worldwide team is only a call away.
Allucent (CATO SMS and Pharm-Olam) – 0.36 billion USD, 1200+ employees .
Allucent originated with CATO SMS, itself created by the merger of Cato Research and SMS-oncology in 2019. Cato Research, founded in 1988, was known for its ability to design and execute successful development strategies and guide creative new products through the regulatory process.
Previous acquisitions by CATO SMS included Array Biostatistics, a full-service biostatistical and statistical programming CRO, and Nuventra Pharma Sciences, one of the industry’s leading providers of clinical pharmacology science and services. With these acquisitions, CATO SMS expanded its services to offer biostatistical consulting, analysis, programming, and cutting-edge modeling and simulation techniques to inform clinical trial designs and predict trial outcomes.
In early 2022, CATO SMS merged with Pharm-Olam, a global clinical research organization delivering clinical trial services to organizations around the world.
KCR – 0.15 billion USD, 700 employees
KCR is a clinical development solutions provider creating value for biotechnology and pharmaceutical organizations.
Founded in 1997, our expert teams support clients with full-service clinical development capabilities across three main areas: Trial Execution, Consulting, and Placement. KCR operates across North America, Europe, and Australia, with main office locations in Boston, US, Berlin, Germany, and Warsaw, Poland.
Our geographical coverage across 25+ countries, cutting-edge technical capabilities and tailored offerings allow for the optimized delivery of solutions to develop life-changing therapies. KCR offers access to an estimated population of 1.1 billion people.
Advanced Clinical – 0.1 billion USD , 1100+ employees
Today, Advanced Clinical has grown organically, with coverage across North America, Eastern Europe, Western Europe and Asia-Pacific, providing contract research organization (CRO), FSP and strategic resourcing solutions. A research services and strategic resourcing organization committed to providing a better clinical experience by delivering comprehensive Contract Research Organization (CRO), Functional Service Provider (FSP) and Strategic Resourcing services that optimize effectiveness for both patients and sponsors throughout the clinical research journey.
Using our decades of experience, we strive to improve the lives of everyone touched by clinical research and help our clients achieve better outcomes through candid conversations, foresight, resilience and innovative solutions.
CTI Clinical Trial and Consulting Services – 0.08 billion USD, 750 employees . CTI has grown consistently and significantly, becoming a global organization with associates in more than 60 countries across the world. We have worked on more than 10,000 projects, worked on every continent except Antarctica, and have contributed to more than 150 new drug and device approvals through global regulatory agencies such as the FDA, EMA, and others. Currently, we work with approximately 250 pharmaceutical and emerging biotechnology and medical device companies.
Among recent reasons for mergers and the whole new approach to conducting clinical research, apart from pursuit of new clients, talents, expertise, products, and markets, there have been notorious coronavirus crisis and military conflicts worldwide. We faced the advent of globalization, decentralization, remote monitoring, and personalized approach to drugs R&D. But we are now looking into the era of technology in clinical research. Processes that allow remote audits and monitoring, previously considered too risky, are now risk mitigators (Bahls, Christine, 2021), at least from that point of view. Clinical trial participants enrollment, and even participation itself, constantly moves into tech dimension.
Only few among the leading CROs escaped M&As for now, but this seems to be the matter of time. Savlovschi – Wicks, Theodora (2022) estimated that “in 2021, the global CRO market is expected to reach an impressive US$88 billion by 2028”. According to IQVIA , only in biopharma sector the total transaction value grew by 70% to reach 152 billion USD by the end of 2023, featuring prominent $43Bn Pfizer-Seagen deal, and other considerable M&As for BMS-Karuna ($14Bn), Merck-Prometheus ($10.2Bn), AbbVie-Immunogen ($10.1Bn), AbbVie-Cerevel ($8.7Bn), Biogen-Reata ($7.3Bn) and Roche-Telavant ($7.1Bn).
Among the CRO businesses the following M&As took place lately:
- Charles River Labs acquires MPI Research (2018), Citoxlab (2019), HemaCare (2020) and Cognate BioServices (2021)
- IQVIA (formerly Quintiles and IMS Health) acquired Clintec and 40% of Q2 Solutions (2021)
- ICON purchased PRA Health Services (2021)
- Thermo Fisher Scientific bought PPD (2021)
- Labcorp purchased Covance and Chiltern
- Inotiv acquired Envigo (2021)
- INC Research and inVentiv Health merged to later become Syneos Health
- Parexel become the property of EQT Private Equity and Goldman Sachs Asset Management
- CATO SMS and Pharm-Olam merged in 2022
- Triley Bidco acquired Clinigen (2022)
The article by Nataliia Vietchinkina, MS in Clinical Research Administration
University of Liverpool.
References:
- Christine Bahls (2021) ‘The Post-Pandemic CRO Landscape’, Applied Clinical Trials, Applied Clinical Trials-09-01-2021, Volume 30, Issue 9 [online] Available from: https://www.appliedclinicaltrialsonline.com/view/the-post-pandemic-cro-landscape
- Savlovschi – Wicks, Theodora (2022) ‘Top 10 CROs to watch in 2022’ [online]. Available from: https://www.proclinical.com/blogs/2022-3/top-10-cros-to-watch-in-2022
- Labcorp (2022) Labcorp Announces 2021 Fourth Quarter and Full-Year Results, 10 February 2022 [online]. Available from” https://ir.labcorp.com/news-releases/news-release-details/labcorp-announces-2021-fourth-quarter-and-full-year-results#:~:text=Revenue%20was%20%2416.12%20billion%20%2C%20an,by%20divestitures%20of%20(0.1%25)
- IQVIA (2022) IQVIA Reports Fourth-Quarter and Full-Year 2021 Results; Raises Full-Year 2022 Profit Guidance, 15 February 2022 [online]. Available from: https://ir.iqvia.com/press-releases/press-release-details/2022/IQVIA-Reports-Fourth-Quarter-and-Full-Year-2021-Results-Raises-Full-Year-2022-Profit-Guidance/default.aspx#:~:text=Revenue%20of%20%2413%2C874%20million%20for,12.4%20percent%20at%20constant%20currency
- ICON (2022) ICON Reports Fourth Quarter and Full Year 2021 Results, 2022 [online]. Available from: https://www.iconplc.com/news-events/press-releases/icon-reports-fourth-quarter-and-full-year-2021-results/
- Businesswire (2021) ‘PPD Reports Fourth Quarter and Full Year 2020 Results’, 23 February 2021 [online]. Available from: https://www.businesswire.com/news/home/20210223006094/en/PPD-Reports-Fourth-Quarter-and-Full-Year-2020-Results
- Macrotrends (2022) ‘Syneos Health Revenue 2012-2021 | SYNH’ [online]. Available from: https://www.macrotrends.net/stocks/charts/SYNH/syneos-health/revenue
- https://companiesmarketcap.com/ (2022) ‘Revenue for Charles River Laboratories (CRL)’ [online]. Available from: https://companiesmarketcap.com/charles-river-laboratories/revenue/
- Vinluan, Frank (2021) ‘CRO Parexel changes private equity hands again, this time for $8.5B’ [online]. Available from: https://medcitynews.com/2021/07/cro-parexel-changes-private-equity-hands-again-this-time-for-8-5b/
- Medpace (2021) ‘Medpace Holdings, Inc. Reports Third Quarter 2021 Results’ [online]. Available from: https://investor.medpace.com/news-releases/news-release-details/medpace-holdings-inc-reports-third-quarter-2021-results#:~:text=The%20Company%20forecasts%202021%20revenue,%24176.0%20million%20to%20%24180.0%20million%20 .
- Companiesmarketcap (2022) ‘Revenue for WuXi AppTec (2359.HK) Revenue in 2021 (TTM)’ [online]. Available from: https://companiesmarketcap.com/wuxi-apptec/revenue/
- https://pharma.lonza.com (2022) ‘Design, assess and optimize for clinical success’ [online]. Available from: https://pharma.lonza.com/offerings/early-development-services .
Clinical Research News
Upcoming clinical trials.
- Gulab Devi Hospital Recruiting Comparison Between Low Pressure Pneumoperitoneum With High Pressure Pneumoperitoneum in Post-operative Pain, Shoulder Tip Pain and Common Bile Duct Injuries in Patients Undergoing Laparoscopic Cholecystectomy Pneumoperitoneum | Pain, Postoperative | Cholecystectomy Pakistan
- Spyre Therapeutics, Inc. Altasciences Company Inc. Recruiting A Study of SPY001-001 in Healthy Volunteers Healthy United States, Canada
- Kocaeli City Hospital Recruiting Initiation of Airway Code: The Role of the Airway Team in Unexpected Difficult Airways Airway Management | Intubation;Difficult | Unexpected Difficult Airway Turkey
- Karolinska Institutet Not yet recruiting Early Double Sequential Defibrillation in Out-of-hospital Cardiac Arrest (DOUBLE-D) Cardiac Arrest | Sudden Cardiac Death | Sudden Cardiac Arrest | Fibrillation, Ventricular
- Shanghai Jiaolian Drug Research and Development... Shanghai Pharmaceuticals Holding Co., Ltd Not yet recruiting A Clinical Study to Evaluate Safety, Tolerability and Pharmacokinetics of SV001 in Chinese Healthy Adult Volunteers. Generalized Myasthenia Gravis China
- Obstetrics & Gynecology Hospital of Fudan University Recruiting Development of an Imaging Prediction Model for Pelvic Lymph Node Metastasis of Cervical Cancer Using Artificial Intelligence Techniques. Cervical Cancer | Artificial Intelligence | Lymph Node Metastasis China
- AstraZeneca Not yet recruiting Volrustomig Priming Regimens Exploratory Phase II Platform Study (eVOLVE-01) Non-small Cell Lung Cancer
- Fujian Medical University Recruiting One-Year Trajectory of Cognitive Changes in Patients With Late-life Depression in the Short Term Late Life Depression China
- BioMérieux Recruiting VIDAS® TBI Performance in Subjects With Mild Traumatic Brain Injury (mTBI) Mild Traumatic Brain Injury United States
- Ataturk University Not yet recruiting The Effect of Mindfulness Compassionate Living Practice on Hemodialysis Patients Hemodialysis Patients
- Edward Via Virginia College of Osteopathic Medicine Recruiting Effects of Spencer's Technique on Shoulder Function Shoulder United States
- University Hospital, Montpellier Not yet recruiting Identification and Characterization of Circulating Tumor Cells Before and After Treatment (Surgery and Radiotherapy) in a Cohort of Patients Treated for Resectable Stage I-III Merkel Cell Carcinoma (MERCCALIBI) Merkel Cell Carcinoma
Clinical Research Jobs
Sponsors and Collaborators
- Assiut University
- GlaxoSmithKline
- National Cancer Institute (NCI)
- Cairo University
- AstraZeneca
- Assistance Publique - Hôpitaux de Paris
- Mayo Clinic
- M.D. Anderson Cancer Center
- Novartis Pharmaceuticals
- National Institute of Allergy and Infectious Diseases (NIAID)
- Massachusetts General Hospital
- National Taiwan University Hospital
- Boehringer Ingelheim
- Hoffmann-La Roche
- Merck Sharp & Dohme LLC
- Stanford University
- University of California, San Francisco
- Eli Lilly and Company
- Duke University
- View Full List
Medical Conditions
- Breast Cancer
- Hypertension
- Prostate Cancer
- HIV Infections
- Coronary Artery Disease
- Heart Failure
- Diabetes Mellitus, Type 2
- Colorectal Cancer
- Lung Cancer
- Schizophrenia
- Cardiovascular Diseases
- Atrial Fibrillation
Drug Interventions
- Cyclophosphamide
- Carboplatin
- Dexamethasone
- Gemcitabine
- Capecitabine
- Bevacizumab
- Pembrolizumab
- Methotrexate
- Oxaliplatin
- Dexmedetomidine
- Fludarabine
10 Best Clinical Research Organizations In The USA
With the growing population, diseases affecting mankind are also increasing. The old medications are being obsolete for certain diseases like bacterial infections, and new effective medicines are urgently needed. The clinical research institutions facilitate drug manufacture by running preclinical and clinical tests on the drugs. In addition, they also study safety, efficacy, and effectiveness of medical devices, diagnostic products, and treatment regimens intended for human health. The healthcare industry is booming with a lot of clinical research institutions in the US and all around the world.
Table of Contents
Top 10 American CROs
The United States of America has some of the top clinical research organizations in the world. Amongst them, here is the list of the 10 Best Clinical Research Organizations In The USA.
Annual Revenue: $9.6B Headquarters: Princeton, New Jersey Number of Employees: 50,000+ Total Funding: $0 Status: Public, Subsidiary of LabCorp Founded: 1996
In 2014, LabCorp acquired Covance for $6.1B. Covance, a global contract research organization under LabCorp, is the world’s leading healthcare diagnostics company, providing comprehensive clinical laboratory services and end-to-end solutions for drug and diagnostics development and commercialization. With its mission to bring new and innovative medicines to patients sooner, it has worked on more than 50 drugs available in the market today. It provides drug development services across all phases of development and multiple therapeutic areas.
Annual Revenue: $9.74B Headquarters: Durham, North Carolina Number of Employees: 55,000 Operating Income: $1.33B Status: Public, Independent Company Founded: 1982
Working to enhance healthcare by integrating health information technologies and clinical research, IQVIA explores a new path to better health outcomes via Human Data Science. It integrates the study of human science with breakthroughs in data science and technology to advance our understanding of human health, and help everyone make better, more insightful decisions.
3. Syneos Health
4. parexel international.
Annual Revenue: $2.4B Headquarters: Waltham, Massachusetts Number of Employees: 18,660 Total Funding: $0 Status: Public, Subsidiary of Pamplona Capital Management LLP Founded: 1982
5. PRA Health Sciences
Annual Revenue: $2.7B Headquarters: Raleigh, North Carolina Number of Employees: 16,400 Total Funding: $305.6M Status: Public, Subsidiary of Kohlberg Kravis Roberts & Co. L. P Founded: 1976
PRA participated in the pivotal or supportive trials and/or key NDA support services that led to the approval of 85 important products currently on the market. Its aim is to improve the drug development process for getting better medicines to market sooner.
6. Pharmaceutical Product Development
Annual Revenue: $1.9B Headquarters: Wilmington, North Carolina Number of Employees: 18,583 Total Funding: $0 Status: Private, Independent Company Founded: 1985
PPD applies innovative technologies, therapeutic expertise, and a firm commitment to provide comprehensive, integrated drug development, laboratory, and lifecycle management services. Its clients and partners include pharmaceutical, biotechnology, medical device, academic, and government organizations. It helps to bend the cost and time curve of drug development and optimize value in delivering life-changing therapies to improve health.
7. Charles River Laboratories
Annual Revenue: $2.27B Headquarters: Wilmington, Massachusetts Number of Employees: 14,700 Total Funding: $224M Status: Public, Independent Company Founded: 1947
Icon helps its clients to accelerate the development of drugs and devices that save lives and improve quality of life. It offers a full range of consulting, development, and commercialization services from a global network of offices in 37 countries.
9. WuXi AppTec
10. medpace holdings.
Annual Revenue: $704.6M Headquarters: Cincinnati, Ohio Number of Employees: 2,900 Total Funding: $161M Status: Public, Subsidiary of Cinven Limited Founded: 1992
Medpace provides services for Phase I-IV of drug and medical device development services, including regulatory services and central laboratory services. It combines scientific guidance, efficient clinical trial management, global regulatory leadership, and innovative technologies to deliver high-quality results.
Related Posts
Top 10 medical device companies in the world, top 10 jobs you can get in the biotechnology sector, top 10 biotechnology universities in the world.

Why Catalyst Clinical Research?
You need someone to listen to your needs. You need a catalyst for your drug development needs.
Catalyst provides highly customizable clinical trial and drug development solutions to the global biotechnology and biopharmaceutical industries through our Catalyst Oncology and Catalyst Flex. Through these two solutions, Catalyst offers a full-service oncology CRO and multi-therapeutic functional service provider services.
With Catalyst, you can always count on us. We are a catalyst.
Get the latest news and press releases from Catalyst Clinical Research

- Privacy Overview
- Strictly Necessary Cookies
- 3rd Party Cookies
- Additional Cookies
This website uses cookies so that we can provide you with the best user experience possible. Cookie information is stored in your browser and performs functions such as recognising you when you return to our website and helping our team to understand which sections of the website you find most interesting and useful.
Strictly Necessary Cookie should be enabled at all times so that we can save your preferences for cookie settings.
If you disable this cookie, we will not be able to save your preferences. This means that every time you visit this website you will need to enable or disable cookies again.
This website uses Google Analytics to collect anonymous information such as the number of visitors to the site, and the most popular pages.
Keeping this cookie enabled helps us to improve our website.
Please enable Strictly Necessary Cookies first so that we can save your preferences!
This website uses the following additional cookies:
(List the cookies that you are using on the website here.)

Clinical Research Services in USA
Global team and resources to serve you better, executive support assigned to your program, latest news.
Meet us at 2024 ASCO Annual Meeting

Message from the Managing Director’s Desk
As we continue to navigate the ever-changing clinical research industry, I am pleased to share some perspectives and developments within our company that will drive our future growth strategies.
We have made a paradigm shift in our operations in the US and Canada, with consolidation of Lambda Therapeutic Research’s Toronto, Canada clinic and bioanalytical laboratory into Novum operations. Canada’s state-of-the-art facility offers niche Pharmacokinetics (PK), Late Phase and Complex Bioanalytical services, which we feel would be a comprehensive value-add in your Drug development programs. By embracing the Novum brand, with its nearly fifty year history, as across our North American operations, we will deliver an enhanced, more seamless suite of services to our clients.
In the past year, we have maintained an impeccable regulatory track record, successfully clearing various international inspections across our global units. We have also further strengthened our global clinical trial management capabilities by strategically investing in scientific talent and expanding our global network of clinical sites. Our commitment to exceeding client expectations remains unwavering, and we continue to offer a comprehensive portfolio of early and late-phase services.
We are confident that our positive energy, unwavering integrity, and unwavering commitment will ensure we continue to meet and exceed client expectations, always .
Bindi Chudgar Managing Director
Turnkey Clinical Trial Management solution for YOUR drug development programs
Guided by a senior management team with expertise in science, operations, quality assurance, medical affairs and project management, Novum offers the highest levels of integrity and reliability.

Phase I Early Clinical Development

Phase II-IV Clinical Trial Management

Scientific Affairs

Bioanalytical

Nearly fifty years of high-quality research outcomes.
Novum is a full-service CRO with an impeccable performance record and numerous FDA approvals in new drug, 505(b)(2) and ANDA submissions. We manage complex studies across numerous therapeutic areas and dosage forms, spanning early phase to late phase trials. With the full integration of our bioanalytical unit in Toronto, Canada in 2020, Novum is poised to continue its longstanding tradition of serving clients with safety, quality, and scientific leadership.
There are plenty of ways to get in touch. Just select the one that is best for you.
Novum prides itself on its talented and dedicated team of employees. The Novum team is passionate about customer satisfaction, safety and study integrity. All Novum clinical staff members receive extensive training designed to continually hone and increase the skills of each employee.

Want to participate in a study?
Last year we paid over $6.6 million to participants of our research studies.
This website stores cookies on your computer. These cookies are used to collect information about how you interact with our website and allow us to remember you. We use this information in order to improve and customize your browsing experience and for analytics and metrics about our visitors both on this website and other media. To find out more about the cookies we use, see our Privacy Policy.
If you decline, your information won’t be tracked when you visit this website. A single cookie will be used in your browser to remember your preference not to be tracked.
- Company Insights
- New Registration
- Update Registration
- Phase 1 to 4 Clinical Trials
- Medical Monitoring
- Project Management
- Regulatory Affairs
- Site Selection
- Vendor Management
- Clinical Safety Services
- Clinical Operations
- Investigator Site Selection
- Site Selection and Management
- Patient Recruitment & Retention
- Data Management & Statistics
- Medical Writing
- Literature Services
- Signal detection and Report management
- Adverse Event Intake
- Case Processing
- Development and Implementation
Phase 1 to 4 Clinical Trial
Medical device clinical evaluation, software as a medical device clinical evaluation, bioequivalence clinical trials, biological clinical trials, herbal and nutraceutical clinical trials, in vitro diagnostics clinical evaluation, new drug and repurposed drug clinical trials, personalized medicine clinical trials, drug safety and pharmacovigilance services for us fda, drug safety and pharmacovigilance services for ema, drug safety and pharmacovigilance services for cdsco.
- CNS – Including Pain & Psychiatry
- Autoimmune / Inflammation
- Cardiovascular
- Opthalmology
- Endocrinology / Diabetology
Clinical Research Organization in US & India
Welcome to prorelix research cro, your drug development arm.
ProRelix Research is a Clinical Research Organization (CRO) with offices in the USA and India along with site networks in Australia, Thailand, Africa, and Europe supporting the clinical development of innovative new healthcare inventions and innovations to improve the health of patients. Every project is equally important to us. We first listen to you and strategize, aiming to a long-term partnership. We understand each project is unique on its own, and conscious clinical development, where efficiency is utmost and accuracy is supreme, is valuable and demanding to long-term success.
We provide services to help pharmaceutical, medical device, biopharmaceutical and nutraceutical/ herbal companies to transform scientific discoveries into new treatments. From clinical trial services, clinical data management services, pharmacovigilance services, medical writing services, drug regulatory affairs services, decentralized clinical trial services to leveraging real world insights, our therapeutic, technical, & operational ability is underpinned by a great conviction in what we do.
ProRelix Research has a global network of distinguished scientific leaders who are experts in a diverse range of therapeutic areas. We specialize in chronic diseases clinical trial services including oncology, respiratory, ophthalmology, endocrinology, immunology, dermatology, etc. Our reputation in clinical research services differentiates us from conventional clinical research organizations (CRO). We leverage our scientific leadership to establish and maintain the engagement of unique investigator networks for clinical research services.
Our Portfolio
Clinical trials play a pivotal role in the approval of new drugs as they most accurately reflect the effect of the drug in the human body. Since drug development is a costly and laborious process with the clinical trial phase...
Medical devices are an integral part of our lives from a simple thermometer to a life-saving pacemaker. Devising medical device clinical studies is complex and requires knowledge and experience-our experts at ProRelix Research can help you design studies depending upon...
The use of software to inform healthcare decisions is ubiquitous and is being utilized by patients, clinicians, and healthcare providers. Proper understanding, planning, and design of SaMD is essential for approval. ProRelix Research provides customized solutions and helps you adapt...
The success of a generic drug or biosimilar often hinges on the results of bioequivalence studies making it essential to partner with an experienced and reliable CRO to design and conduct bioequivalence studies. ProRelix Research offers full range service for...
ProRelix Research offers customized, end-to-end services for clinical trials of a broad range of biological products to help bring both innovator biologics and biosimilars to the market in a timely and cost-effective manner. We ensure compliance with regulatory requirements in...
The exponential growth in the natural product and dietary supplements field requires clinical trials to support safety and efficacy. ProRelix Research lends support at every step of your clinical trial journey to help you carve a niche in the dietary...
Diagnosis of diseases by clinicians using suitable tests guides treatment decisions and is necessary for improving and prolonging the health and well-being of patients. ProRelix Research partners with you to provide clinical trial support and ensure your device meets regulatory,...
ProRelix Research provides tailored end-to-end management of clinical trials keeping cost and patient safety at the forefront. We help you navigate the complex path of clinical trials by offering study support from patient recruitment to statistical interpretation to help you...
At ProRelix Research we understand that realization of the full potential of the emerging field of personalized medicine is dependent on the design and execution of clinical trials. We can help you circumvent the unique challenges associated with conducting precision...
ProRelix Research understands the utmost importance of protecting patient safety from clinical trials to real-world use of a new drug, biological or medical device. We help you develop appropriate pharmacovigilance plans and adverse event reporting systems in compliance with US...
Our experts at ProRelix research provide customized and comprehensive pharmacovigilance and safety monitoring programs in accordance with European Union (EU) regulations with an emphasis on patient well-being from research and development to post-market authorisation.
Patient safety and well-being lie at the heart of all our pharmacovigilance operations at ProRelix Research. We are with you every step of the way-from development of a pharmacovigilance system to regular reporting of safety and adverse events to regulatory...
How ProRelix Research can help you?
The successful execution and completion of a clinical trial are determined by several interconnected cross-functional expertise teams, including Medical Writing, Regulatory Affairs, Clinical Project Management, Clinical Data Management, Site Management, and Patient Recruitment, Faster Ethics Committee Approval, Pharmacovigilance, and much more.
A Quality driven Global Clinical Research Organization (CRO) that can truly understand your needs and help you to achieve your goals by minimizing the complexity across functional teams. We at ProRelix Research strive to bring forth the best clinical research services managed by therapeutically sound experts and professionals who are having over a decade of international regulatory experience in conducting complex clinical trials into simple ones.
Also, our extensive knowledge, quality work, and global recognition make us an ideal choice for the rising demand for conducting holistic clinical trials which include New/ Repurposed Pharmaceutical Products, Biological, Herbal, Nutraceutical, In Vitro Diagnostic Devices, Therapeutic Medical Devices, and more.
We are dedicated to follow the best ethical and clinical practices when conducting clinical studies while keeping the safety of all of our participant at core. Our motto is to provide our clients with prompt clinical study management, reliable clinical data with integrity, and ethical conduction of the clinical trials. The uniqueness of our services and functionality is that we not only design the ideal mechanism for better functionality of your trials but we also provide you a lending hand in all those processes and take you through them from the start till the end to reach your goal.
Our service portfolio includes Clinical Trial Project management, Phase 1 to 4 Clinical Trials, Drug Regulatory Affairs, Medical Writing, Clinical Data Management, Pharmacovigilance, Clinical Operations, etc . We are open to clients from all over the world and have a strong presence in USA and India and strategic partners in Europe, Australia. Let’s commit ourselves to better healthcare systems all around the world by ethical, prompt, and reliable clinical research studies!
Our Highlights
- Clinical Research Organization with Global Services Capability in USA, India, Europe, and Australia
- Phase 1, 2, 3, and 4 clinical trial services for (i) Pharmaceutical (ii) Medical Device (iii) Herbal/ Neutraceutical
- Organized Structure and Reputation
- Experienced and Best Service Portfolio
- Adaptability and Flexibility with Sponsor/Study Requirements
- Quality Assurance and Transparency
CRO Services
Clinical research services.
Our clinical research service focuses on the Quality, Ethics, and Well-being of the clinical trial subject… Read More
Data Management & Statistics Services
At ProRelix Research we are a team of experienced people who are well aware… Read More
Medical Writing Services
ProRelix Research’s medical writing service capabilities range from Clinical Research, Regulatory… Read More
Regulatory Affairs Services
ProRelix Research’s Regulatory Affairs services provide expert support in drug development programs. Read More
Recent Posts
Requirements to conduct first-in-human (fih) clinical trials for biopharmaceuticals.
The transition of a drug, medical device, or biopharmaceutical from the laboratory bench to bedside involves a long series of steps starting from drug discovery, computational chemistry, in vitro and […]
The Promise of Clinical Trials in Advancing Solid Tumour Cancer Treatments
According to the National Cancer Institute (NCI), a solid tumour is a mass of tissue that usually does not contain cysts or liquid areas such as sarcomas (arising from connective […]
Investigator Initiated Trial (IIT) and Proof of Concept Study: Collaborative Model in Industry-Acade …
Explore the dynamic synergy between industry and academia in groundbreaking medical research through Investigator Initiated Trials (IITs) and Proof of Concept Studies. Uncover the collaborative model shaping innovative partnerships, driving […]
Testimonials
I would highly recommend Prorelix Research as a client‐focused company with a strong patient database that delivers on its promise to deal with patient recruitment.
Global Pharma Company
We selected the Prorelix Research because of its comprehensive nature, flexibility and reliability. We were happy as ProRelix Research proved to be a best solution to our clinical trial operations .
Indian Pharma Company
We are very happy to have found Prorelix Research, during our relationship, ProRelix Research has delivered best services for our medical device study project..
Global Medical Device Company
The quality of the service provided by ProRelix Research matches the sophistication of their services platform. Their services have helped us thoroughly and promptly.
Privacy Overview
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |
Select Services Clinical Research Medical Writing Data Management & Statistics Pharmacovigilance Regulatory Affairs
Register Now
All the fields are mandatory
Applying AI to manage the risks and costs of postmarketing requirements
Translating intent to impact.
The triad of trust: Navigating real-world healthcare data integration
Ensuring the validity of clinical outcomes assessment data.
The value of rater training
Advancements in Artificial Intelligence for site selection
Using human-enabled AI to enhance decision-making and minimise risk.
A multifaceted risk factor: Addressing obesity's impact across the disease spectrum
Considerations for clinical trial design focused on obesity treatments.
Optimising biotech funding whitepaper series
Navigating biotech's challenges and embracing a promising tomorrow.
2023 biotech sector survey
Featured solutions.
Blended solutions
Bespoke, seamless solutions to meet unique sponsor challenges.
Digital Health Technologies
ICON acquires HumanFirst, a cloud-based technology company for life sciences supporting precision measurement in patient centred clinical research.
ICON provides full service outsourcing and flexible support for biotech specific needs such as due diligence and asset valuation.
Cardiac Safety Solutions
End-to-end cardiac safety solutions, including ECG, event monitoring, BPM, long-term Holter monitoring, ECHO and MUGA studies.
Early Clinical and Bioanalytical Solutions
Innovative early clinical solutions that will advance your drug development strategy.
Site & Patient Solutions
Transforming recruitment through patient-centric trials and real-world, real-time data.
Market Access
Expertise in mission-critical pricing, market access, and reimbursement.
Unlocking precision medicine: An overview of Illumina’s TSO500
17 June 2024. Register today.
Ensuring patient recruitment success at a site level
9 July 2024. Register today.
Practical guidance for successful global regulatory submissions: Understanding FDA and PMDA data standards requirements
Watch the webinar.
ICON Insights
- 06 Jun 2024
The next frontier: Digital health and patient safety
- 04 Jun 2024
ICON at SCDM 2024: Connecting the dots of clinical data management
Navigating regulatory landscapes: A guide to global submission standards
- 10 May 2024
Integrating Performance Studies of In-Vitro Diagnostics into Clinical Trials: A Complex Challenge
SDoH data analysis for proactive outcome improvement: A multilayered approach
- 09 May 2024
Navigating the complexities of healthcare data types
- 01 May 2024
Cross-border initiative to empower rare disease researchers
- 26 Apr 2024
How AI could transform literature surveillance for pharmacovigilance
- 25 Apr 2024
Complexities of running clinical trials in retinal disorders and ways to overcome them
What’s happening in icon, how can we help.
- Cell and Gene Therapies
- Early Clinical
- Medical Device
- Rare & Orphan Diseases
- Real World Evidence
- Site & Patient Recruitment
- Strategic Solutions
Top Clinical Research Organizations (CRO) in the United States
Patricio ledesma.
Contract Research Organization
6 January, 2020

Contact us at [email protected] if you need CRO services in the United States.
Which are the top clinical research organizations (CRO) in the United States?
The top clinical research organizations (CRO) in the United States are Covance (LabCorp), IQVIA, Syneos Health, PPD, PRA Health Sciences, and Parexel.
Covance (LabCorp)
Covance (CEO Paul Kirchgraber) is a CRO offering preclinical, clinical, and commercialization services for the pharma and biotech industries. Founded in 1996 and with headquarters in Princeton, New Jersey, Covance is a subsidiary of LabCorp and employs more than 50,000 people in over 60 nations. LabCorp’s revenue in 2018 was $11.3 billion.
IQVIA (CEO Ari Bousbib), formerly known as Quintiles, is a multinational CRO providing biopharmaceutical development, laboratory, analytical, and consulting services for phase I-IV clinical trials. Founded in 1982, and with headquarters in Durham, North Carolina, IQVIA has more than 58,000 employees in more than 100 countries. IQVIA’s revenue in 2018 was $10.4 billion.
Syneos Health
Syneos Health (CEO Alistair Macdonald) is a global CRO created through the merger of INC Research and inVentiv Health. Founded in 1984 and based in Morrisville, North Carolina, Syneos Health provides integrated biopharmaceutical solutions bringing together nearly 24,000 employees in more than 110 countries. Syneos Health’s revenue in 2018 was $4.39 billion.
PPD (CEO David Simmons) is a leading global CRO founded in 1985, providing comprehensive drug development, laboratory, and lifecycle management services. PPD’s central offices are located in Wilmington, North Carolina, and has around 23,000 employees around the world. PPD’s annual revenue in 2018 was $2.8 billion.
PRA Health Sciences
PRA Health Sciences (CEO Colin Shannon) is a multinational CRO serving the biopharmaceutical industry. With headquarters in Raleigh, North Carolina, the company was founded in 1982 and by the end of 2019 employed 15,800 people approximately. PRA Health Sciences’ annual revenue in 2018 was $2.8 billion.
Parexel (CEO Jamie Macdonald) is a global CRO offering biopharmaceutical and clinical trial management services for the clinical research sector. Parexel, founded in 1982, has central offices in Waltham, Massachusetts, and employs around 20,000 professionals operating in more than 51 countries. Parexel’s revenue in 2018 was $2.4 billion.
Request for Information
Privacy Policy

Patricio Ledesma ( B.Eng Concordia University, Montreal, Canada and Master’s Degree in Clinical Trials, University of Seville, Spain) is the Head of Clinical Operations and Founder at Sofpromed CRO. Patricio is a professional consultant providing comprehensive, one-stop expert advice and guidance to biotechnology and pharmaceutical companies worldwide in the field of clinical trials and drug development. He is personally and enthusiastically devoted to helping biotech Chief Executive, Operations, Scientific, Medical, and Regulatory Officers in the planning and execution of phase I-IV clinical trials across North America, Europe, Asia-Pacific, Latin America, and Middle East regions. You can contact Patricio at: +34 607 939 266 [email protected]
To our articles and updates
- Follow Follow
You may also be interested…

List of Spanish CROs Managing Clinical Trials in Spain
Please contact us at [email protected] if you need a Spanish CRO to conduct a clinical trial in Spain Spain is a world leader in clinical research and in conducting clinical trials, currently positioned within the top 4 European countries with more clinical studies...

What Are Community-Based Clinical Trials?
Community-Based Clinical Trials
If you are a Sponsor seeking to run a community-based clinical trial in underserved populations, please contact us at [email protected] Clinical trials are fundamental to advance medical knowledge and improve patient care. However, one significant challenge in...

Clinical Research Site Networks: Accelerating Clinical Trials
Site Networks
If you are a Sponsor seeking to run a clinical trial through a clinical research site network, please contact us at [email protected] Clinical research plays a central role in advancing medical treatments and improving healthcare outcomes. To ensure the smooth...

How Community-Based Clinical Trials Are Advancing Healthcare in Underserved Populations
If you are a Sponsor interested in running a community-based clinical trial in underserved populations, please contact us at [email protected] Clinical trials are instrumental in advancing healthcare by evaluating the safety and effectiveness of new treatments and...

Advantages of Clinical Research Site Networks in North Carolina
If you are a Sponsor interested in running a clinical trial through a clinical research site network in North Carolina, please contact us at [email protected] Clinical research plays a pivotal role in advancing medical knowledge, improving patient care, and driving...

Benefits of Clinical Research Site Networks in Illinois
If you are a Sponsor interested in running a clinical trial through a clinical research site network in Illinois, please contact us at [email protected] Clinical site networks play a central role in advancing medical research and improving patient care. In this...

Advantages of Clinical Research Site Networks in Pennsylvania
If you are a Sponsor interested in running a clinical trial through a clinical research site network, please contact us at [email protected] Pennsylvania is a hub for clinical research, with numerous reputable clinical site networks offering a wide range of trials to...

Benefits of Clinical Research Site Networks in New York
If you are a Sponsor interested in running a clinical trial through a clinical research site network in New York, please contact us at [email protected] New York, with its vibrant healthcare landscape, is home to several prominent clinical research site networks.In...
Patricio Ledesma ( B.Eng Concordia University, Montreal, Canada and Master’s Degree in Clinical Trials, University of Seville, Spain) is the Head of Clinical Operations and Founder at Sofpromed CRO. Patricio is a professional consultant providing comprehensive, one-stop expert advice and guidance to biotechnology and pharmaceutical companies worldwide in the field of clinical trials and drug development. He is personally and enthusiastically devoted to helping biotech Chief Executive, Operations, Scientific, Medical, and Regulatory Officers in the planning and execution of phase I-IV clinical trials across North America, Europe, Asia-Pacific, Latin America, and Middle East regions. You can contact Patricio at: +34 607 939 266 [email protected]
A CRO Built for Innovation in Medical Devices, Drugs, and Diagnostics.
About proxima., case studies.

Our Services
Recent news, frequently asked questions, tools &templates.

An official website of the United States government
Here's how you know
Official websites use .gov A .gov website belongs to an official government organization in the United States.
Secure .gov websites use HTTPS A lock ( Lock A locked padlock ) or https:// means you’ve safely connected to the .gov website. Share sensitive information only on official, secure websites.

The Modernized ClinicalTrials.gov Will Become the Singular Website Experience
Classic website to retire on june 25, 2024.
Starting June 25, 2024, the National Library of Medicine’s (NLM) modernized ClinicalTrials.gov website will become the singular website experience for all users. As part of its commitment to enhance the user experience and deliver improved functionality for searching, viewing, and downloading information about clinical trials, NLM is retiring the classic version of the website and its application programming interface. Attempts to use the classic version of the ClinicalTrials.gov website ( https://classic.clinicaltrials.gov/ ), including some frequently used links, will automatically be redirected to the modernized ClinicalTrials.gov website (www.clinicaltrials.gov) where information and documents continue to be available.
ClinicalTrials.gov is the world’s largest clinical trial and results registry. This web-based resource provides patients and their advocates, health care providers, researchers, and the public with easy access to information on publicly and privately supported clinical research studies covering a wide range of diseases and conditions. Managed by NLM on behalf of the National Institutes of Health (NIH), ClinicalTrials.gov lists records for more than 490,000 clinical trials, observational studies, and expanded access programs.
As previously announced , users have had nearly a year to explore the modernized ClinicalTrials.gov website and take advantage of its benefits, which include web components that improve navigation and make important information easier to find. Register to learn more about preparing for the final transition to the new ClinicalTrials.gov, with NLM’s free webinar on June 6, 2024, at 1 p.m. EDT.
For more information, see Modernization Transition Top Questions . To learn more about the modernization effort and stay informed about future updates, visit the ClinicalTrials.gov Modernization webpage and the latest Release Notes .
- Fierce Pharma
- Fierce Biotech
- Fierce Healthcare
- Fierce Life Sciences Events
- Cell & Gene Therapy
- Clinical Data
- Venture Capital
- Diagnostics
- AI and Machine Learning
- Special Reports
- Special Report
- Awards Gala
- Fierce Events
- Industry Events
- Whitepapers
NIH blends primary care with clinical research in $30M pilot to reach underserved patients
A $30 million, two-year pilot program from the National Institutes of Health (NIH) will set up clinical research at primary care facilities in communities with underserved patient populations. The goal is to eventually give them the means to conduct research projects on issues unique to these patients.
Communities Advancing Research Equity for Health, or CARE for Health, was launched June 6. The program’s first sites will be in rural communities and will work with established clinical research networks funded by the NIH to set up on-site research capabilities. The agency will also collaborate with organizations that serve rural communities to get the program started. Funding awards for these partners will be handed down in fall 2024, a press release from the NIH said.
“To better understand relationships between the genes we inherit and the environmental and societal factors that surround us and to deliver more evidence-driven healthcare, research must be integrated into clinical care and community settings, reaching patients from all walks of life,” Monica Bertagnolli, M.D., director of the NIH, wrote in an editorial in Science.
“Unlike most research settings, primary care is patient- and community-centered and emphasizes health maintenance and preventive care alongside treatment and rehabilitation in a location as convenient as possible for the patient,” Bertagnolli said.
Blending primary care with clinical research creates a system in which “research informs clinical practice and clinical data informs research,” Tara Schwetz, Ph.D., NIH deputy director for program coordination, planning and strategic initiatives, said in the press release. Program sites will select their own studies from a menu of options rather than have them handed down to them by central researchers, so they can choose those that apply most to issues faced by their communities. Eventually, primary care providers and communities will work together to design their own so they can address their unique healthcare needs.
The network’s research studies will include topics like disease prevention as well as trials on making healthcare delivery more efficient, Bertagnolli wrote. In addition to assistance from the NIH's existing research networks, primary care providers will receive funding and study designs that integrate research into care workflows and make study data collection part of electronic health records.
"The initiative will support [primary care providers] in the way that they need to meet the goals of the research they choose to participate in," Renate Myles, acting director for communications and public liaison for the NIH, told Fierce Biotech Research in an email.
The program is part of a broader strategy to improve access to healthcare for people on the outskirts of the medical system as well as to open up clinical research to historically underrepresented racial and social groups. Better access and research participation is badly needed in rural areas, which have fewer providers —particularly specialists—but more residents who are elderly, poor or uninsured than urban ones. They’re also sicker: Rural areas suffer from greater medical burdens in virtually every category, from maternal health to mental illness.
These challenges are compounded for Black, Indigenous and Hispanic or Latinx populations, who, on account of racism, face unique barriers to quality healthcare on top of broader issues. Some of these populations suffer from higher death rates than people of the same ethnicity in urban areas, an analysis published by Penn State in early 2024 found.
Clinical drug trials have made their way into community settings, too, with pharma giants like Eli Lilly extolling the value of decentralized designs that meet patients where they are rather than requiring them to go to a single site at an academic research center. That’s also true for recruitment efforts: Trial sponsors are traveling to less-than-traditional places, like barber shops, to meet patients out in the community.
“Clinical trials should reflect the diversity of Americans—because we know that delivers the best results,” Xavier Becerra, U.S. secretary of the Department of Health and Human Services, said in the press release about CARE for Health. “We are taking a big step towards ensuring communities that are historically underrepresented in clinical research are fully included and have the same access to the best available results and analysis. There has never been more potential for progress than we have today.”

American Psychological Association

The impact of transgender legislation
Psychological science points to an increased risk of suicide and poor mental health amid a record number of bills aimed at restricting the rights of the LGBTQ+ population
APA policy supporting transgender, gender diverse, nonbinary individuals
Membership in APA

APA Community
A new exclusive destination tailored for APA members

Membership benefits
Unlock the tools, discounts, and services included with your membership

Renew your membership
Keep your benefits and access to leading psychological information
Psychology topics spotlight

Misinformation and disinformation

Resources to navigate trauma

Tips to foster a healthy workplace
Science and practice of psychology

Ethics Code

Continuing Education

Grants, Awards, and Funding

Standards and Guidelines
Networks and communities

Network with peers, enhance your professional development, expand your personal growth, and more

APA Divisions

High school teachers

Undergraduate educators

Graduate students

Early career psychologists

Managing your career
Resources to help you throughout your career in psychology, including finding a job, salary data, finances and money management, mentoring and supervision, and training and professional development

Explore career paths

Psychologist profiles

How did you get that job?

Events and training
Featured jobs
Apa publications and products.

Write with clarity, precision, and inclusion
Children’s books
Monitor on Psychology
Newsletters
Reports and surveys
Continuing education
Merchandise

Real Siblings

Jacob's Missing Book

Harper Becomes a Big Sister
Attachment-Based Family Therapy for Sexual and Gender Minority Young Adults and Their Non-Accepting Parents
Dismantling Everyday Discrimination
APA Services

Learn how you can help APA advocate for psychology-informed federal policy and legislation, and support psychological research

APA Services, Inc.
A companion professional organization to APA, serving all members and advocating for psychology

- Research Overview
Research Activities
- Research Resources
Training & Education
- Collaborate With NCATS
- Funding Overview
- Open Funding Opportunities
- Small Business Programs
- Funding Policy & Operating Guidelines
- Grant Information
- Challenges and Prize Competitions
- Scientific Review
- Conference Grant Information
Resources for You
- Resources for Researchers
- Resources for Trainees
- Resources for Rare Disease Patients and Advocates
- News & Events Overview
- Newsletters
- Scientific Publications
- NCATS Overview
- About Translational Science
- Advisory Committees
- Careers at NCATS
Director's Corner
- Divisions & Offices
More Treatments for All People More Quickly
We conduct research and support activities that address long-standing challenges in translational research so that new treatments and other health solutions reach people faster.
Latest News & Events
Read our latest research news or follow coverage of our translational science activities through national and local media.

Paving the Way for Ultrarare Bone Disease Treatment Trial
May 21, 2024 - NCATS News
- Our Impact on Drug Discovery and Development
- Therapeutics for Rare and Neglected Diseases (TRND)
Clinicians at the NIH Clinical Center will soon start testing an investigative drug for an ultrarare bone disease.
Doctors Advise Caution as Energy Drinks May Trigger Life-Threatening Cardiac Arrhythmias in Patients With Genetic Heart Diseases
June 6, 2024 - Grantee/Partner News
- Clinical and Translational Science Awards (CTSA) Program
Cannabis Use Common Among Patients, With Most Using It to Manage a Symptom or Health Condition
June 5, 2024 - Grantee/Partner News
iTHRIV Scholars Program Announces 2024 Cohort
June 4, 2024 - Grantee/Partner News
Advancing Translational Science

Learn how our work speeds research and improves health. Key areas include big data , clinical trials and rare diseases .

Explore our research labs, projects and collaborations that are advancing translational science. Our largest program is the Clinical and Translational Science Awards (CTSA) Program .

Enhance your knowledge of translational science through programs, activities and resources offered by NCATS and our partner institutions .
Whether you're a researcher, an aspiring translational scientist or an interested member of the community, find information and materials important to you.
For Researchers
Learn about our latest opportunities for funding and collaboration and connect with our staff.
For Trainees
Learn about the resources and support available to help you grow as a translational scientist.
For Patients and Advocates
Learn about the patient-focused research and resources available through NCATS and NIH.

Joni L. Rutter, Ph.D.
Communicating About Rare Diseases
February 28, 2024
NCATS Director Joni Rutter penned a guest post for NIH’s Science, Health, and Public Trust blog on why communicating about rare diseases research is important and what to consider when sharing information.
About the NCATS Director
Joni L. Rutter, Ph.D., is the director of NCATS at NIH. Rutter oversees the planning and execution of NCATS complex, multifaceted programs.
Previous Director's Messages
Search all Director's Messages by topic and year.
Provide Feedback on the Draft NCATS Strategic Plan
We invite you to review the draft NCATS strategic plan and email feedback by June 14.
Last updated on May 14, 2024
- Health Tech
- Health Insurance
- Medical Devices
- Gene Therapy
- Neuroscience
- H5N1 Bird Flu
- Health Disparities
- Infectious Disease
- Mental Health
- Cardiovascular Disease
- Chronic Disease
- Alzheimer's
- Coercive Care
- The Obesity Revolution
- The War on Recovery
- Adam Feuerstein
- Matthew Herper
- Jennifer Adaeze Okwerekwu
- Ed Silverman
- CRISPR Tracker
- Breakthrough Device Tracker
- Generative AI Tracker
- Obesity Drug Tracker
- 2024 STAT Summit
- Wunderkinds Nomination
- STAT Madness
- STAT Brand Studio
Don't miss out
Subscribe to STAT+ today, for the best life sciences journalism in the industry
NIH will bring clinical research into primary care offices with $30 million pilot
By Annalisa Merelli June 7, 2024

F or many Americans, health care means going to a local primary care office. But the vast majority of clinical research is conducted inside the walls of large, specialized academic health centers. Millions of patients are left out of those studies, which often fail to capture the population in all its diversity.
Now, for the first time, the National Institutes of Health is investing in the creation of a national primary care research network to try to address this issue. Its $30 million pilot program, called Communities Advancing Research Equity for Health and announced on Thursday, will fund and support a small number of primary care sites as they participate in a range of clinical trials.
advertisement
“This is an incredibly exciting opportunity,” said Diane Harper, a primary care physician and professor in family medicine at the University of Michigan, Ann Arbor. Instead of running individual, siloed research projects out of specialized sites, the program will allow network members to participate in several trials — choosing between 20 to 30 studies — to match their local population’s health needs.
“A person is not a disease, and most of the NIH is organized around diseases,” said Harper. “This is the first time that NIH is recognizing that people are complex, and have many factors that pertain to their health care … that cannot be teased out and separated to be studied in a single, particular line.”
Related: NIH-funded clinical trials often miss racial, gender diversity enrollment goals, report finds
The pilot is an opportunity to better understand how research can serve the needs of patients outside traditional settings, said NIH Director Monica Bertagnolli. “We know that every community is different, and we cannot just assume that a rural community in Alabama is going to be the same as a rural community in Montana or that their health issues are the same,” she said. “What we want to do is to be able to offer any community a whole bunch of different research opportunities,” learning through experience what they find most meaningful.
The research projects will go beyond drug trials to emphasize issues that are acutely experienced by communities facing health disparities, including substance use, mental health disorders, and obesity.
The timeline is tight: The NIH hopes to award funding very soon, and hold its first investigator meeting by the winter. The focus on a speedy start, said Harper, is not giving primary care sites sufficient time to apply for the funding deadline, which is set for next week. As a result, she worries the first round of research will be led by academic networks with links to primary care. “These are not primary care networks,” she said. “These are Ph.D.s with ideas about what it means to be in primary care, it’s not the people who practice.”
The accelerated timeline was put in place to support a launch by the end of fiscal year 2024, said NIH spokesperson Renate Myles, while still allowing the application period to be open for about six weeks. “We expect more opportunities in future years for potential applicants who were unable to participate this year,” she said.
This isn’t the first experiment with primary care research networks. “There’s many folks who’ve worked for a long time to help the NIH get to this point where they’re ready to take their research out of the academic medical center into rural and frontier and underserved community practices,” said Jack Westfall, a rural primary care physician and retired professor at the University of Colorado.
In the late 1990s, Westfall helped establish the High Plains Research Network, a research network of all the primary care practices in eastern, rural, and frontier Colorado towns — most with just a few thousand residents each. He’s found that community-based studies can pay dividends for researchers, too.
“The NIH research needs to move outside of the academic medical centers, out into the community, out into the primary care practices, both for finding study subjects, but also for finding research ideas,” said Westfall. “Many times there are clinical questions that come up out of the community, from the patients, from their interactions with their physicians that could generate ongoing ideas for research.”
Related: Inside a push to create an NIH office for post-infection chronic illness
The NIH also sees the program as an opportunity to gain trust from communities that are skeptical of the medical establishment. “We are here to understand what people need and to earn their trust by delivering for them,” said Bertagnolli. “Trust is not automatic, it has to be earned.”
To build that trust, the network will have to be careful not to treat patients and their local providers like cogs in a machine. “The risk is that this will just be extractive, not collaborative,” said Westfall. “We want to make sure that this is not just an extraction of study subjects out of primary care and NIH, but a bidirectional flow of resources, of ideas, of topics, of power.”
“Making studies available closer to where people are actually getting treated, that’s the first step,” said Andrew Trister, chief medical and scientific officer at Verily, an Alphabet company that builds tools for clinical research. But it raises a number of important questions, he said: “What’s the chain of the trust chain? Who is trusted in the community? Who could be able to help people understand more about what the clinical study is about? Why participate in research?”
Down the line, the network could help primary care sites to mobilize more effectively in case of national health emergencies. “The Covid pandemic did not use primary care at all efficiently,” said Harper. But if the pilot is successful and expands to create a nationwide network, it could eventually enable a faster, more effective public health response — and clinical trials, to boot.
About the Author Reprints
Annalisa merelli.
General Assignment Reporter
Annalisa (Nalis) Merelli is a general assignment reporter at STAT. Her interests are ever-expanding, but she is especially drawn to the coverage of reproductive and maternal health, and their intersection with health equity.
Clinical trials
STAT encourages you to share your voice. We welcome your commentary, criticism, and expertise on our subscriber-only platform, STAT+ Connect
To submit a correction request, please visit our Contact Us page .

Recommended

Recommended Stories

Lab experiment shows exactly how heat waves can put hearts into ‘oxygen debt’

STAT Plus: FDA advisers unanimously recommend approval of Eli Lilly’s Alzheimer’s disease treatment

STAT Plus: The inside story of how Lykos’ MDMA research went awry

STAT Plus: FDA advisory panel votes overwhelmingly against MDMA therapy for PTSD

STAT Plus: Elevance PBM’s president out as customers complain of prescription chaos
First Cancer TIL Therapy Gets FDA Approval for Advanced Melanoma
March 5, 2024 , by Carmen Phillips

FDA has approved the first treatment for cancer that uses tumor-infiltrating lymphocytes, or TILs.
In an event more than three decades in the making, the Food and Drug Administration (FDA) has approved lifileucel (Amtagvi), the first treatment for cancer that uses immune cells called tumor-infiltrating lymphocytes, or TILs. Announced on February 16, the agency’s decision also makes lifileucel the first cellular therapy to be approved for a solid tumor , the skin cancer melanoma.
The agency’s accelerated approval covers the use of lifileucel for people with advanced melanoma that has gotten worse after treatment with certain immunotherapy drugs or targeted therapies.
As is the case with CAR T-cell therapy , another type of cellular therapy, lifileucel is made using a patient’s own T cells . And with both therapies, the cells are collected at the hospital where the patient is being treated but are sent away to be manufactured into the final treatment.
But there’s a key difference between TILs and CAR T-cell therapies.
For the approved CAR T-cell therapies, the T cells are collected from a patient’s circulating blood. For TIL therapy, by contrast, the T cells are collected from the patient’s tumor.
Lifileucel’s approval was based on findings from a clinical trial sponsored by Iovance Biotherapeutics. Among more than 70 participants who were treated with the lifileucel dose ultimately approved by FDA, nearly one-third had at least some reduction in the size of their tumors (a tumor response), with tumors disappearing completely (a complete response ) in several participants.
Moreover, about 40% of those whose cancer responded to lifileucel still had no progression of their cancer a year after receiving the one-time infusion treatment.
TIL therapy was pioneered by Steven Rosenberg, M.D., and his colleagues in NCI’s Surgery Branch . In the late 1980s, Dr. Rosenberg led the first-ever clinical trials of TIL therapy, showing that it could shrink tumors in people with very advanced melanoma. (Dr. Rosenberg was recently given the nation’s highest award for technology and innovation for his work on immunotherapy , including the development of TIL therapy.)
Over the ensuing years, the NCI researchers further refined the process for manufacturing and delivering TIL therapy. In 2011, NCI entered into a cooperative research agreement with Iovance to further develop this particular TIL therapy, including conducting larger clinical trials and developing a manufacturing infrastructure, paving the way for FDA approval.
It’s been a long time coming, Dr. Rosenberg acknowledged. The new approval “is a big step,” he said, one which demonstrates that “cellular therapy is joining the mainstream of cancer treatment.”
TILs that just need a little help
Six different CAR T-cell therapies have been approved by FDA, all of which are used to treat blood cancers like leukemia and lymphoma.
With CAR T-cell therapy, the collected immune cells undergo genetic engineering in the laboratory to give them the ability to recognize cancer cells and enhance their ability to kill them. Next, they are grown, or expanded, into the hundreds of millions, and then infused into the patient.
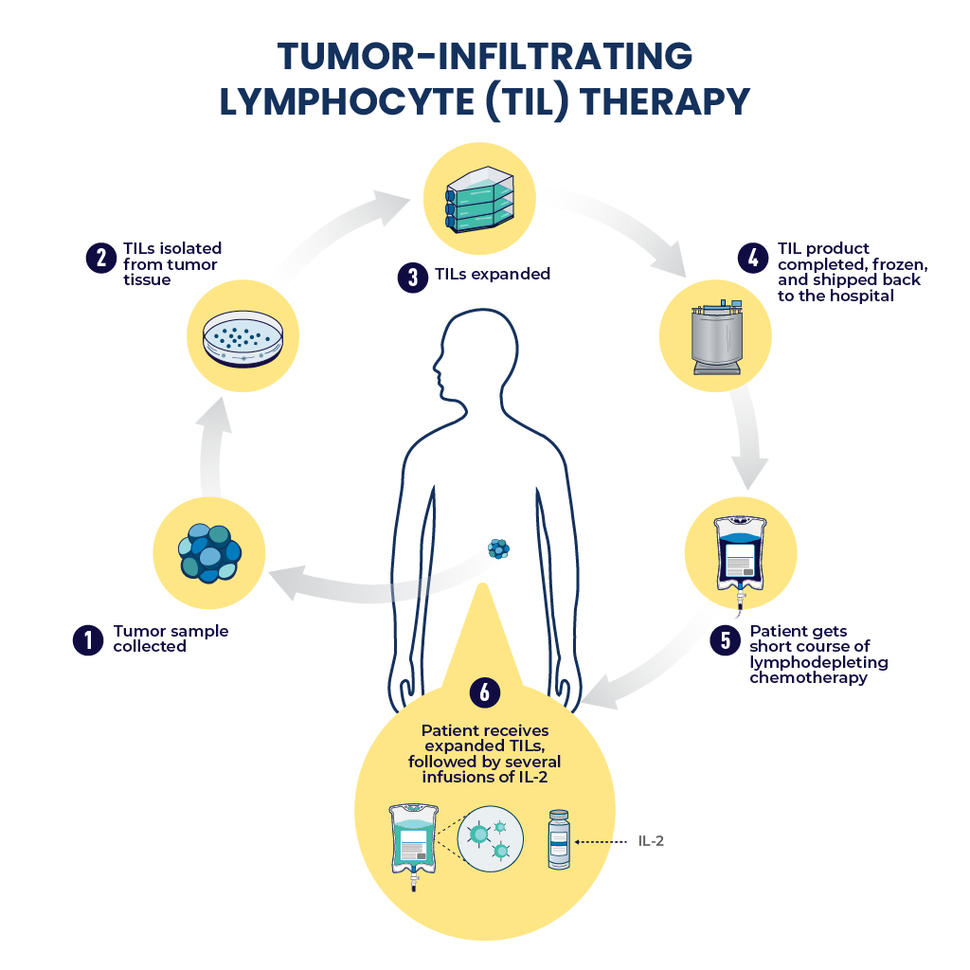
Tumor-infiltrating lymphocytes, or TILs, are T cells collected from a patient's own tumor. Once isolated from the tumor sample, the TILs are expanded into the billions and infused back into the patient.
With lifileucel, the collected T cells are not engineered before being expanded. That’s because, having been collected from samples of tumor tissue, they’ve already proven that they can recognize and navigate to tumors, Dr. Rosenberg explained. That recognition relies on the presence of specific abnormal proteins , or antigens , on the surface of the tumor cells.
But, like a robber who sneaks into a bank but doesn’t have the tools to open the safe, the collected TILs lack the ability or support required to complete the job. What the TILs need are reinforcements—a lot of them—and some encouragement.
So, although the TILs collected from a patient’s tumor are not genetically changed, there are steps taken during the manufacturing process—including mixing them with a drug called IL-2 —to help them expand into billions of cancer-fighting immune cells.
In addition, as part of lifileucel treatment, two measures are taken to enhance the infused TILs ability to attack the cancer and their overall potency. First, in the days prior to receiving lifileucel, patients undergo a few rounds of high-dose “lymphodepleting” chemotherapy. And, second, shortly after the lifileucel infusion, they are given several doses of IL-2.
Long-lasting tumor responses to lifileucel
FDA’s approval was based on data from a group of 73 patients in the trial whose cancer had gotten worse despite treatment with a PD-1 / PD-L1 -targeted immune checkpoint inhibitor or a BRAF inhibitor and whose lifileucel dose was at least 7.5 billion cells (the dose specified in the approval).
Longer-term findings from a larger group of 153 patients in the trial were reported in December 2023 at the European Society of Medical Oncology’s (ESMO) 2023 Immuno-Oncology Congress, and they largely mirror the findings from these 73 patients.
Of the 48 people (31.5%) whose cancer responded to treatment with lifileucel, more than half have lived at least a year without any evidence of their cancer getting worse . About 10% of these responses are ongoing—that is, there continues to be no evidence that their cancer is getting worse—and nearly all have lasted several years.
The longest lasting response is nearly 5 years, one of the trial’s lead researchers, Martin Wermke, M.D., Ph.D., of the University of Dresden in Germany, reported at the ESMO meeting.
The researchers plan to follow all patients for 5 years from the time of their lifileucel infusion, Dr. Wermke said. Based on the results thus far, he continued, the researchers anticipate that cancer will not get worse or come back in any participants with ongoing responses.
Who will respond to lifileucel? What about side effects?
There are some hints from the results in the larger patient group that might help identify who is most likely to respond to lifileucel, Dr. Wermke explained.
For example, few people whose cancer had spread to the brain or liver, which was the case for about half of the 153 patients, responded. Responses were also unlikely in people who had larger tumor burdens—that is, a substantial amount of cancer in their body.
These findings, he noted, suggest that lifileucel might be most effective if it’s given before the cancer has spread to many other parts of the body or individual tumors have become quite large.
All participants in the trial had side effects caused by the treatment, Dr. Wermke reported. Most, however, were not dangerous and were largely caused by the chemotherapy given before the lifileucel infusion and IL-2 given afterward. The most common included anemia , high fevers, and substantial drops in levels of platelets and certain white blood cells .
TIL therapy does not appear to cause the often serious immune-related side effects commonly seen in people treated with CAR T-cell therapy, including cytokine release syndrome and neurologic effects, Dr. Wermke said.
He also stressed that the side effects seen in the trial had a consistent and familiar pattern.
“They’re all occurring during the first 2 or 3 weeks” after someone receives the treatment, he said. “There are hardly any new [side effects] occurring in the long term,” he said.
From clinical trials to using TILs in everyday cancer care
Iovance announced that it has approved more than 30 treatment centers across the United States to collect tumor samples for making lifileucel and administering the final treatment to patients. There are expected to be 50 such centers by the end of May.
Initially, all treatments will be manufactured at an Iovance facility or a contracting facility, both in Philadelphia.
One CAR T-Cell Therapy for Blood Cancers?
Using CRISPR, researchers have developed a potential “universal” immunotherapy for blood cancers like leukemia and lymphoma.
According to the company, a single treatment with lifileucel will cost $515,000. During a company webcast held after the approval, Iovance officials said they expect that many insurance companies will cover the treatment at the same levels as they have for CAR T-cell therapies, which also cost hundreds of thousands of dollars. The company has also established a financial assistance program for patients.
William Sharfman, M.D., a co-director of the melanoma program at the Johns Hopkins Sidney Kimmel Cancer Center, said the approval is “very exciting” news for people with advanced melanoma.
But, Dr. Sharfman cautioned, “there are several limiting factors” for using lifileucel in patients.
Among them, he said, is that patients need to be relatively healthy to receive it, which isn’t always the case for people with advanced cancer who have undergone other treatments.
“Patients really need to have normal heart and lung function,” he said.
Alexander Shoushtari, M.D., of Memorial Sloan Kettering Cancer Center, who studies cellular therapies, agreed that practical considerations may limit lifileucel’s use.
“The relatively high doses of … chemotherapy given before treatment and the high dose Interleukin-2 after [lifileucel] infusion currently limit the types of people who can undergo it,” Dr. Shoushtari said.
After lifileucel approval, what comes next?
FDA’s approval for melanoma is only the first step for lifileucel. Iovance has already started enrolling participants in a large trial combining lifileucel with the immunotherapy drug pembrolizumab (Keytruda) as an initial treatment for advanced melanoma.
Lifileucel is also well along in testing as a treatment for other cancers, including showing promising tumor responses in people with advanced lung cancer , as well as ovarian and head and neck cancers.
Dr. Rosenberg’s lab and others are testing newer forms of TIL-based therapies in clinical trials, and not just for melanoma, but for other solid cancers.
“There are clear examples where TIL therapy has caused regression of [other solid] tumors,” Dr. Rosenberg said. That includes complete eradication of tumors in individual patients with advanced colon cancer and advanced breast cancer treated in clinical trials at the NIH Clinical Center.
With this “landmark approval” in hand, there is a lot of optimism surrounding TIL therapy and cellular therapy more broadly, Dr. Shoushtari said. “I expect that this [approval] will be the tip of the iceberg for cellular-based immune therapy for solid tumors,” he added.
The first-generation TILs like lifileucel are “an important advance and proof of concept” that these treatments can be highly effective, he said. “But much remains to be done to broaden [their] use, both for other cancer types and to a broader range of people with melanoma.”
Dr. Sharfman agreed. “That is the challenge: to design better TILs.”
Researchers are already taking up this challenge, include finding ways to make TILs that are more potent and that can be used in a broader range of solid tumors. Dr. Rosenberg’s group at NCI, for example, has developed a process for identifying TILs that strongly recognize a tumor and are most likely to attack it.
Researchers have also begun genetic engineering of TILs, with the goal of increasing the number of people who respond to the treatment or eliminating the need for the pre-infusion chemotherapy or post-infusion IL-2.
Dr. Rosenberg stressed that the decades of research on cancer cellular therapies by his and other groups has paved the way for rapid advances.
“We now have the knowledge of how to help the immune system target tumors,” particularly the solid tumors “that are responsible for the vast majority of cancer deaths,” he said. “There is a world of opportunities out there.”
Featured Posts
March 27, 2024, by Edward Winstead
March 21, 2024, by Elia Ben-Ari
March 5, 2024, by Carmen Phillips
- Biology of Cancer
- Cancer Risk
- Childhood Cancer
- Clinical Trial Results
- Disparities
- FDA Approvals
- Global Health
- Leadership & Expert Views
- Screening & Early Detection
- Survivorship & Supportive Care
- February (6)
- January (6)
- December (7)
- November (6)
- October (7)
- September (7)
- February (7)
- November (7)
- October (5)
- September (6)
- November (4)
- September (9)
- February (5)
- October (8)
- January (7)
- December (6)
- September (8)
- February (9)
- December (9)
- November (9)
- October (9)
- September (11)
- February (11)
- January (10)

An official website of the United States government
Here’s how you know
Official websites use .gov A .gov website belongs to an official government organization in the United States.
Secure .gov websites use HTTPS A lock ( Lock A locked padlock ) or https:// means you’ve safely connected to the .gov website. Share sensitive information only on official, secure websites.
Project NextGen Selects Initial Vaccine Candidates and Awards Over $500 Million to Advance Development of Vaccines and Therapeutics
Vaccine selection brings products closer to clinical trials slated to begin as early as this winter
Today, the U.S. Department of Health and Human Services (HHS), through the Administration for Strategic Preparedness and Response (ASPR), is announcing the selection of initial next-generation vaccine candidates and more than $500 million in awards for Project NextGen – kick-starting planning for Phase 2b clinical trials and technologies that advance innovative next-generation vaccine and therapeutics platforms.
“The Biden-Harris Administration is committed to keeping people safe from COVID-19,” said HHS Secretary Xavier Becerra. “By investing in next-generation vaccines and treatments, we can improve our ability to respond to new variants, reduce transmission, stop infections, and save lives. Through Project NextGen, we are combining research and development expertise at HHS with the lessons learned throughout the pandemic to protect our nation from COVID-19.”
The over $500 million announced today builds on the over $1.4 billion awarded in August – accelerating products toward clinical trials and potential commercial availability.
“The vaccine selections and funding announced today are important steps forward for Project NextGen – with vaccine and therapeutics candidates moving quickly to clinical trials that will start in the coming months,” said Assistant Secretary for Preparedness and Response Dawn O’Connell. “The technologies that BARDA is investing in, from intranasal vaccines to self-amplifying mRNA, will bolster our protection against COVID-19 for years to come.”
All three next-generation vaccine selections announced today are distinct from each other, targeting stronger, broader, or longer-lasting immune responses. Intranasal vaccines have the potential to stop viruses at the site of infection and self-amplifying mRNA and additional antigens may generate a stronger immune response than current vaccine technologies. The awards announced today will support companies as they prepare their vaccine candidates for Phase 2b clinical evaluation:
- $8.5 million to CastleVax for a vector-based intranasal vaccine candidate.
- $10 million to Codagenix for a live-attenuated intranasal vaccine candidate.
- $10 million to Gritstone Bio for a self-amplifying mRNA vaccine candidate.
To support these and all future Project NextGen clinical studies, BARDA is awarding over $240 million, including investments in cold-chain sample management, genomic sequencing, and increased central laboratory capacity for sample testing:
- $87 million to American Type Culture Collection (ATCC) to provide cold-chain and specimen lifecycle management.
- $4 million to BioInfoExperts to provide genomic sequencing of COVID-19.
- $23.2 million to Meso Scale Diagnostics (MSD) to develop and manufacture multiplexed COVID-19 antibody test kits.
- $126.5 million to PPD to provide a central laboratory capacity that analyzes immune responses from samples collected during Project NextGen vaccine clinical trials.
Finally, BARDA is awarding over $241 million to support new technologies that will improve national preparedness for future COVID-19 outbreaks and patient access. These technologies have the potential to enable shorter development timelines for monoclonal antibodies through mRNA, increased efficiency in virus testing, and alternate routes for vaccine administration:
- $40 million to Vir Biotechnology to support the development of mRNA-expressed monoclonal antibodies.
- $100 million to Luminary Labs to administer two competitions focused on incentivizing the development of patches for vaccine delivery and reducing the number of vaccine doses needed for an effective immune response.
- $59 million to ModeX Therapeutics to develop a monoclonal antibody for COVID-19 pre-exposure prophylaxis that can bind to multiple sites on the virus.
- $10.5 million to Charles Stark Draper Laboratory (Draper Labs) to advance the development of tissue chip platform technology.
- $6.1 million to Battelle and $5.8 million to the UK Health Security Agency to advance human lung and airway tissue chip platform technology.
- $20 million to Evidation to test the use of commercial wearables for early respiratory illness detection and a home specimen sampling capability.
To view the full list of awards and learn more about Project NextGen, please visit medicalcountermeasures.gov/nextgen/ .
Sign Up for Email Updates
Receive the latest updates from the Secretary, Blogs, and News Releases
Subscribe to RSS
Receive latest updates

Related News Releases
Covid-19 vaccination public education campaign saved thousands of lives, billions of dollars, hhs, dhs, and fema announce completion of multi-year review of state efforts to provide language access during the covid-19 public health emergency, readout of hhs secretary becerra’s meeting with pharmacy ceos on covid-19 therapeutics commercialization, related blog posts.

Calling innovators! HHS Long COVID Healthathon launches with $25K in prizes
Reflections from the deputy director of the hhs office of long covid research and practice.

Driving Long COVID Innovation with Health+ Human-Centered Design
Media inquiries.
For general media inquiries, please contact [email protected] .
- Skip to main content
- Skip to FDA Search
- Skip to in this section menu
- Skip to footer links

The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you're on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
U.S. Food and Drug Administration
- Search
- Menu
- For Patients
Clinical Trials: What Patients Need to Know
Learn more about clinical trials and find a trial that might be right for you. Clinical trials are voluntary research studies conducted in people and designed to answer specific questions about the safety or effectiveness of drugs, vaccines, other therapies, or new ways of using existing treatments. It is important to remember that the FDA does not conduct Clinical Trials.
Search for a Clinical Trial
Enter a word or phrase, such as the name of a medical condition or intervention. Example: Cancer AND Los Angeles or expanded access AND compassionate use
Learn More About Clinical Trials
Clinical Research Versus Medical Treatment Understand the differences between clinical research and medical treatment and what those differences mean for you. Find answers to your questions about clinical trials, such as why they are done, who should consider participating, and issues to consider before joining a trial.

What are the Different Types of Clinical Research Understand the different types of research and the four clinical trial phases, such as their purpose and how many people participate in each of the phases.
Informed Consent for Clinical Trials Understand what informed consent is and the questions you need to know before signing informed consent.
Diversity in Clinical Trial Participation It is important to test drugs and medical products in the people they are meant to help. Learn about FDASIA 907 and how FDA works to make sure that people of different ages, races, ethnic groups, and genders are included in clinical trials.
What is an Institutional Review Board Understand what Institutional Review Boards are, who is on them and who they protect.
Sexual and Gender Minorities The NIH Sexual and Gender Minority Research Office (SGMRO) coordinates sexual and gender minority (SGM)--related research and activities by working directly with the NIH Institutes, Centers, and Offices. The office was officially established in September 2015 within the NIH Division of Program Coordination, Planning, and Strategic Initiatives (DPCPSI).
Resources For You
- NIH Clinical Research Trials and You
- Good Clinical Practice
- HHS Office of Human Research Protections - About Research Participation
- Interactive Patient Education Tutorial On Clinical Trials
- Protecting America's Health Through Human Drugs
- Protection of Human Subjects of Research
- About ClinicalTrials.gov

IMAGES
VIDEO
COMMENTS
1. IQVIA. Formerly known as Quintiles and IMS Health, IQVIA is one of the largest CROs in the world, with a large range of service offerings to help advance clinical research. The company was founded in North Carolina in 1982, and has since grown to over 88,000 employees in more than 100 countries.
Medelis is a full service oncology clinical research company. We provide sponsors with a highly-skilled and proactive oncology clinical trial management team to design and manage phase I through phase III oncology trials in North America and Europe. ... Global Contract Research Organizations in United States. 54gene's Clinical Programs Group ...
Founded: 1982 Headquarters: Waltham, Massachusetts Annual Revenue: $3B Employee Size: 18,000 Parexel is another important name in the list of Clinical Research Companies In USA that specializes on behalf of its pharmaceutical partners in carrying out clinical studies to boost and make certain that the medicine approval process takes place smoothly. It has a wide range of offerings that spreads ...
Here is a quick overview of the best companies of clinical research: IQVIA: Best for data-driven insights and advanced analytics in healthcare research. ICON: Best for comprehensive clinical development services and therapeutic expertise. Parexel: Best for global biopharmaceutical services, emphasizing regulatory and clinical trial excellence.
Parexel is a clinical research organization that provides biopharmaceutical services and solutions. They have expertise in clinical development, translational medicine, and early phase research. Parexel has over 10,000 employees in 50 countries. Parexel has been recognized by Frost & Sullivan with the 2022 Global Customer Value Leadership Award.
Top 10 Leading Clinical Research Companies In The USA. The landscape of drug discovery and development is intricate, demanding significant resources at each stage of the process. As pharmaceutical companies strive to bring groundbreaking therapies to market, they increasingly rely on the expertise of Contract Research Organizations (CROs).
Who we are. Parexel is among the world's largest clinical research organizations (CROs), providing the full range of Phase I to IV clinical development services to help lifesaving treatments reach patients faster. Leveraging the breadth of our clinical, regulatory, and therapeutic expertise, our team of more than 21,000 global professionals ...
PPD offers a comprehensive set of lab services - from bioanalytical to vaccine sciences. Offering proven solutions, ranging from early development to pharmacovigilance to post-approval services, to ensure your product's success. Find out how we help.
Laboratory Corporation of America Holdings, also known as Labcorp, is an American company that operates one of the largest clinical laboratory networks in the world. In an average week Labcorp processes tests on more than 3 million patient specimens. In 2020 the company earned revenue in excess of $14 bn. Syneos Health.
At Worldwide, we believe a personalized approach is the only way to unlock the power of everyone working on a study - from operational and therapeutic experts to site partners and scientists. That means you'll always have direct access to our experts. You'll be able to tap into more than 30 years of therapeutic experience on a global scale.
Parexel is proudly among the world's largestclinical research organizations. A dedicated CRO providing the full range of Phase I to IV clinical development services and leveraging the breadth of our clinical, regulatory and therapeutic expertise, our team of more than 21,000 global professionals works in partnership with biopharmaceutical ...
In 2022 PharmaTimes named us Clinical Research Company of the Year—The Americas, an honor we have received many times over the last few years. We were also named PharmaTimes International Clinical Research Company of the Year. At ICON, we know the power of research can change lives for the better. That's why we're dedicated to the future ...
A research services and strategic resourcing organization committed to providing a better clinical experience by delivering comprehensive Contract Research Organization (CRO), Functional Service Provider (FSP) and Strategic Resourcing services that optimize effectiveness for both patients and sponsors throughout the clinical research journey.
The top 15 CROs globally would be in the following order based on 2023 revenue: LabCorp (Covance), with 15.05 billion USD (2023) and 75000 employees ( Labcorp, 2024). In February 2015, Labcorp completes its $6 billion purchase of Covance, Inc., creating the world's leading health care diagnostics company.
The United States of America has some of the top clinical research organizations in the world. Amongst them, here is the list of the 10 Best Clinical Research Organizations In The USA. 1. Covance. Source. Annual Revenue: $9.6B. Headquarters: Princeton, New Jersey. Number of Employees: 50,000+. Total Funding: $0.
Catalyst Flex is a provider of specialized, multi-therapeutic functional solutions delivered by experts in clinical operations, data management, biostatistics and programming, safety and medical writing. We leverage experienced clinical research professionals to partner and address your study needs. Learn About Catalyst Flex.
List of clinical research organizations in the US. 1. IQVIA. IQVIA is a globally renowned medical research companies in USA that stands out for its exceptional contributions to the healthcare and pharmaceutical industries. As one of the leading providers of advanced analytics, technology solutions, and contract research services, IQVIA plays a ...
As we continue to navigate the ever-changing clinical research industry, I am pleased to share some perspectives and developments within our company that will drive our future growth strategies. We have made a paradigm shift in our operations in the US and Canada, with consolidation of Lambda Therapeutic Research's Toronto, Canada clinic and ...
ProRelix Research is a Clinical Research Organization (CRO) with offices in the USA and India along with site networks in Australia, Thailand, Africa, and Europe supporting the clinical development of innovative new healthcare inventions and innovations to improve the health of patients. Every project is equally important to us. We first listen to you and strategize, aiming to a long-term ...
ICON is the world's leading clinical research organisation, providing outsourced clincal development and commercialisation services to the pharmaceutical, ... ICON acquires HumanFirst, a cloud-based technology company for life sciences supporting precision measurement in patient centred clinical research.
PPD (CEO David Simmons) is a leading global CRO founded in 1985, providing comprehensive drug development, laboratory, and lifecycle management services. PPD's central offices are located in Wilmington, North Carolina, and has around 23,000 employees around the world. PPD's annual revenue in 2018 was $2.8 billion.
Proxima provides regulatory, quality & clinical trial life science solutions to the new class of rising stars in drugs, devices, diagnostics & digital therapeutics By clicking "Accept", you agree to the storing of cookies on your device to enhance site navigation, analyze site usage, and assist in our marketing efforts.
ClinicalTrials.gov is the world's largest clinical trial and results registry. This web-based resource provides patients and their advocates, health care providers, researchers, and the public with easy access to information on publicly and privately supported clinical research studies covering a wide range of diseases and conditions.
A $30 million, two-year pilot program from the National Institutes of Health (NIH) will set up clinical research at primary care facilities in communities with underserved patient populations.
The American Psychological Association (APA) is a scientific and professional organization that represents psychologists in the United States. APA educates the public about psychology, behavioral science and mental health; promotes psychological science and practice; fosters the education and training of psychological scientists, practitioners and educators; advocates for psychological ...
Research Activities. Explore our research labs, projects and collaborations that are advancing translational science. Our largest program is the Clinical and Translational Science Awards (CTSA) Program.
In the late 1990s, Westfall helped establish the High Plains Research Network, a research network of all the primary care practices in eastern, rural, and frontier Colorado towns — most with ...
From clinical trials to using TILs in everyday cancer care. Iovance announced that it has approved more than 30 treatment centers across the United States to collect tumor samples for making lifileucel and administering the final treatment to patients. There are expected to be 50 such centers by the end of May. ... According to the company, a ...
Through Project NextGen, we are combining research and development expertise at HHS with the lessons learned throughout the pandemic to protect our nation from COVID-19." The over $500 million announced today builds on the over $1.4 billion awarded in August - accelerating products toward clinical trials and potential commercial availability.
Clinical trials are voluntary research studies conducted in people and designed to answer specific questions about the safety or effectiveness of drugs, vaccines, other therapies, or new ways of ...